Technology Readiness Levels (TRL) in Forensic Science: A Complete Guide from Concept to Courtroom
This article provides a comprehensive guide to the Technology Readiness Level (TRL) scale, a systematic metric for assessing technological maturity, with a specific focus on its critical application in forensic...
Technology Readiness Levels (TRL) in Forensic Science: A Complete Guide from Concept to Courtroom
Abstract
This article provides a comprehensive guide to the Technology Readiness Level (TRL) scale, a systematic metric for assessing technological maturity, with a specific focus on its critical application in forensic science. Tailored for researchers, scientists, and drug development professionals, it explores the TRL framework from foundational principles to advanced implementation. Readers will gain insights into navigating the unique challenges of forensic technology development, including methodological adaptations for techniques like comprehensive two-dimensional gas chromatography (GC×GC), strategies for overcoming funding and validation hurdles, and meeting the rigorous standards for legal admissibility. The content synthesizes current research and strategic priorities to offer a practical roadmap for advancing forensic technologies from the laboratory into validated, court-ready tools.
What is the TRL Scale? Defining the Framework from NASA to Modern Forensic Labs
The Origins and History of Technology Readiness Levels
Technology Readiness Levels (TRLs) represent a systematic metric for assessing the maturity of a particular technology. Originally developed by the National Aeronautics and Space Administration (NASA) in the 1970s, the TRL framework has evolved from a specialized aerospace assessment tool into a globally adopted methodology across diverse sectors, including forensic science research. This whitepaper traces the historical development of TRLs, examines their standardized definitions, and explores their critical application in forensic science for evaluating the maturity of analytical techniques such as comprehensive two-dimensional gas chromatography (GC×GC) against legal admissibility standards. By providing a structured approach to technology assessment, TRLs enable forensic researchers, scientists, and drug development professionals to effectively gauge technical maturity and navigate the pathway from fundamental research to court-admissible evidence.
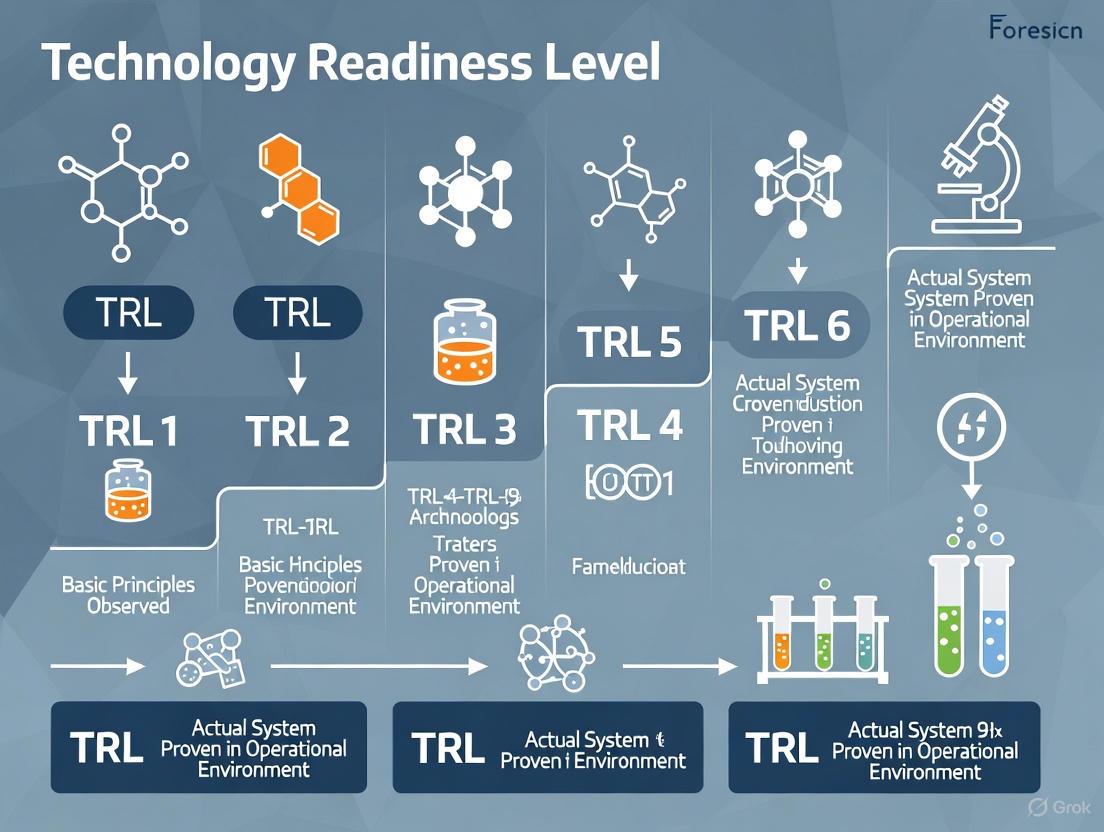
Technology Readiness Levels (TRLs) are a type of measurement system used to assess the maturity level of a particular technology during the acquisition phase of a program [1]. Each technology project is evaluated against parameters for each technology level and is then assigned a TRL rating based on the project's progress, with TRL 1 being the lowest (basic principles observed) and TRL 9 being the highest (actual system proven through successful mission operations) [1]. TRLs enable consistent and uniform discussions of technical maturity across different types of technology, providing a common language for engineers, managers, and investors to gauge technological readiness for deployment [2].
The fundamental purpose of using TRLs is to help management in making decisions concerning the development and transitioning of technology [2]. Among their key advantages are providing a common understanding of technology status, facilitating risk management, informing decisions concerning technology funding, and guiding the transition of technology from research to operational use [2]. The primary purpose of using technology readiness levels is to help management in making decisions concerning the development and transitioning of technology, serving as one of several tools needed to manage the progress of research and development activity within an organization [2].
Table 1: The Nine Technology Readiness Levels
| TRL | Description | Key Characteristics |
|---|---|---|
| TRL 1 | Basic principles observed and reported | Scientific research begins translation into applied R&D; basic properties studied [3] |
| TRL 2 | Technology concept formulated | Practical applications invented; limited to analytic studies [3] |
| TRL 3 | Analytical and experimental critical function proof-of-concept | Active R&D initiated; laboratory studies; proof-of-concept model constructed [1] |
| TRL 4 | Component validation in laboratory environment | Basic technological components integrated and tested in laboratory [3] |
| TRL 5 | Component validation in relevant environment | Technology tested in simulated realistic environment; breadboard technology [1] |
| TRL 6 | System/subsystem model demonstration in relevant environment | Fully functional prototype tested in realistic environment [3] |
| TRL 7 | System prototype demonstration in operational environment | Working model demonstrated in space environment [1] |
| TRL 8 | Actual system completed and qualified | System "flight qualified" through test and demonstration [1] |
| TRL 9 | Actual system proven through successful operations | Actual system "flight proven" through successful mission operations [1] |
Historical Development of TRLs
Origins at NASA
Technology Readiness Levels were conceived at NASA in 1974 and formally defined in 1989 [2]. The methodology was originated by Stan Sadin at NASA Headquarters in 1974 [2]. The original definition included seven levels, but in the 1990s, NASA adopted the nine-level scale that subsequently gained widespread acceptance [2]. The initial development was driven by NASA's need for a structured way to assess and manage the progression of various technologies relevant to its space shuttle missions, which involved substantial costs and risks [4].
Ray Chase, then the JPL Propulsion Division representative on the Jupiter Orbiter design team, played a crucial role in early TRL application at NASA. At the suggestion of Stan Sadin, Chase used this methodology to assess the technology readiness of the proposed JPL Jupiter Orbiter spacecraft design [2]. Chase later spent a year at NASA Headquarters helping Sadin institutionalize the TRL methodology, and after joining ANSER in 1978, he used the TRL methodology to evaluate the technology readiness of proposed Air Force development programs [2].
Table 2: Historical Timeline of TRL Development and Adoption
| Time Period | Key Development | Significant Event |
|---|---|---|
| 1970s | Conceptual Origin | TRL methodology conceived by Stan Sadin at NASA in 1974 [2] |
| 1989 | Formal Definition | NASA formally defines TRL scale with seven levels [2] |
| 1990s | Scale Expansion | NASA adopts nine-level TRL scale [2] |
| 1990s | Early Adoption | U.S. Air Force and Department of Defense begin using TRLs [2] |
| 1995 | Conceptual Expansion | John C. Mankins of NASA publishes paper extending scale descriptions [2] |
| 1999 | Policy Influence | U.S. GAO report recommends wider DOD use of TRLs [2] |
| Early 2000s | Government Mandate | DOD adopts TRLs for procurement; Deputy Under Secretary of Defense issues memorandum endorsing TRL use [2] |
| 2003 | Standardization | ISO publishes ISO 16290:2013 standard incorporating TRLs [2] |
| 2008 | International Adoption | European Space Agency adopts TRL scale [2] |
| 2010 | EU Implementation | European Commission advises EU-funded projects to adopt TRL scale [2] |
| 2013-2014 | Program Integration | TRLs used in EU Horizon 2020 program [2] |
| 2020s | Sector Expansion | Widespread adoption across sectors including forensic science [5] |
Evolution and Refinement
In 1995, John C. Mankins of NASA wrote a seminal paper that discussed NASA's use of TRLs, extended the scale, and proposed expanded descriptions for each TRL level [2]. This work represented a significant refinement of the original NASA definitions and helped facilitate broader adoption beyond aerospace applications.
A pivotal moment in TRL history came in 1999 when the United States General Accounting Office (now Government Accountability Office) produced an influential report that examined differences in technology transition between the DOD and private industry [2]. The GAO concluded that the DOD took greater risks and attempted to transition emerging technologies at lesser degrees of maturity than private industry, increasing overall program risk. The report recommended that the DOD make wider use of technology readiness levels as a means of assessing technology maturity prior to transition, significantly accelerating TRL adoption across government agencies [2].
Government Adoption and Standardization
In 2001, the Deputy Under Secretary of Defense for Science and Technology issued a memorandum that formally endorsed the use of TRLs in new major programs [2]. This was followed by the development of detailed guidance for using TRLs in the 2003 DOD Technology Readiness Assessment Deskbook, marking the formal institutionalization of TRLs within defense acquisition.
The international standardization of TRLs occurred in 2013 with the publication of the ISO 16290:2013 standard by the International Organization for Standardization, which canonized the TRL scale [2]. This represented the culmination of a multi-decade evolution from a NASA-specific assessment tool to a globally recognized methodology.
Diagram 1: TRL Adoption History
TRL Scale Definitions and Comparative Analysis
The original NASA TRL scale has been adapted by various organizations worldwide, with slight modifications to suit different contexts and applications. The core concept remains consistent across these adaptations, maintaining the fundamental progression from basic research to proven operational capability.
Original NASA Definitions
NASA's original TRL definitions, developed during the 1970s and formally defined in 1989, established the foundational framework that subsequent adopters would reference [2]. These definitions were specifically tailored to the needs of space technology development, with higher levels (7-9) explicitly referencing space environments and flight qualification.
Comparative Framework Adaptations
As TRLs gained international acceptance, various organizations developed their own nuanced definitions while maintaining the core nine-level structure. The European Union, through its Horizon 2020 program (and subsequent Horizon Europe program), implemented a TRL scale that adapted NASA's original definitions for broader research and innovation contexts [2]. Similarly, the U.S. Department of Energy and other federal agencies have developed variations with small modifications to certain levels [6].
Table 3: Comparative TRL Definitions Across Organizations
| TRL | NASA Definitions [2] | European Union Definitions [2] | Common Application in Forensic Science |
|---|---|---|---|
| 1 | Basic principles observed and reported | Basic principles observed | Initial observation of analytical principles or techniques |
| 2 | Technology concept and/or application formulated | Technology concept formulated | Formulation of forensic application concept |
| 3 | Analytical and experimental critical function and/or characteristic proof-of-concept | Experimental proof of concept | Laboratory proof-of-concept for forensic analysis |
| 4 | Component and/or breadboard validation in laboratory environment | Technology validated in lab | Basic forensic components validated in controlled lab |
| 5 | Component and/or breadboard validation in relevant environment | Technology validated in relevant environment | Forensic technology tested in simulated operational setting |
| 6 | System/subsystem model or prototype demonstration in a relevant environment (ground or space) | Technology demonstrated in relevant environment | Full prototype demonstrated in simulated forensic lab |
| 7 | System prototype demonstration in a space environment | System prototype demonstration in operational environment | Prototype demonstrated in operational forensic laboratory |
| 8 | Actual system completed and "flight qualified" through test and demonstration (ground or space) | System complete and qualified | System complete and meets forensic laboratory standards |
| 9 | Actual system "flight proven" through successful mission operations | Actual system proven in operational environment | System proven through successful casework application |
The "Valley of Death" in TRL Progression
One of the most critical concepts in TRL implementation is the technological "Valley of Death" - the gap between technology development and successful implementation [7]. This valley is most commonly associated with the transition from TRL 5-6 to TRL 7, when a technology must move from demonstration in a relevant environment to demonstration in a true operational environment [7].
A NASA study noted that the expense and effort required to advance a technology increase dramatically at higher TRLs. The cost of advancing from TRL 5 to TRL 6 can be multiple times higher than all the work from TRL 1 to 5 combined, and moving from TRL 6 to TRL 7 represents an even more substantial leap [7]. This "Valley of Death" is where many promising technologies falter due to technical issues, budget constraints, or difficulties in securing appropriate testing opportunities.
TRLs in Forensic Science Research
Application to Forensic Methodologies
In forensic science research, TRLs provide a structured framework for evaluating the maturity of analytical techniques and methodologies before they can be adopted into routine casework and accepted as evidence in legal proceedings [5]. The implementation of new analytical methods in forensic laboratories requires meeting rigorous analytical standards while also adhering to legal admissibility standards including the Frye Standard, Daubert Standard, Federal Rule of Evidence 702 in the United States, and the Mohan Criteria in Canada [5].
Recent research has applied TRL assessment to comprehensive two-dimensional gas chromatography (GC×GC) techniques for various forensic applications, including illicit drug analysis, fingerprint residue characterization, chemical/biological/nuclear/radioactive (CBNR) substance identification, toxicological evidence, odor decomposition analysis, and petroleum analysis for arson investigations and oil spill tracing [5]. This assessment enables systematic evaluation of both analytical readiness and legal readiness for courtroom implementation.
Experimental Protocols for TRL Assessment in Forensic Science
The progression of forensic technologies through TRL stages requires specific experimental protocols and validation methodologies at each maturity level. For analytical techniques like GC×GC, this involves increasingly rigorous testing environments and validation criteria.
TRL 3-4 Protocol: Laboratory Proof-of-Concept and Validation
- Objective: Establish analytical feasibility and initial laboratory validation
- Methodology: Controlled laboratory studies using standard reference materials and simulated forensic samples
- Validation Parameters: Retention time reproducibility (±0.5 min), peak capacity (>500), signal-to-noise ratio (>10:1), linear dynamic range (≥2 orders of magnitude)
- Quality Controls: Internal standards, calibration verification, system suitability tests
- Documentation: Standard operating procedures, method development reports, initial validation data
TRL 5-6 Protocol: Simulated and Representative Environment Testing
- Objective: Validate performance in simulated operational environments
- Methodology: Testing in mock forensic laboratory settings using authentic case-type samples
- Validation Parameters: Accuracy (±15% of true value), precision (<15% RSD), limit of detection (matrix-specific), selectivity (no interferences from common contaminants)
- Quality Controls: Blind samples, proficiency testing, comparison with established methods
- Documentation: Complete validation reports, uncertainty measurements, comparison studies
TRL 7-8 Protocol: Operational Environment Demonstration and Qualification
- Objective: Demonstrate reliability in operational forensic laboratories and achieve qualification
- Methodology: Implementation in accredited forensic laboratories following quality assurance standards
- Validation Parameters: Ruggedness (different analysts, instruments), reproducibility (inter-laboratory studies), error rate determination, measurement uncertainty
- Quality Controls: Continuous monitoring, external quality assessment, adherence to ISO 17025 standards
- Documentation: Quality manual entries, technical procedures, validation packages for accreditation
Research Reagent Solutions for Forensic TRL Assessment
Table 4: Essential Research Reagents for Forensic Technology Development
| Reagent/Category | Function in TRL Assessment | Application Examples |
|---|---|---|
| Certified Reference Materials | Method validation and calibration at all TRL levels | Drug standards, explosive compounds, ignitable liquid mixtures |
| Internal Standards | Quantification quality control and method performance monitoring | Deuterated analogs, stable isotope-labeled compounds |
| Quality Control Materials | Inter-laboratory reproducibility studies (TRL 6-8) | Proficiency test samples, authentic case-type materials |
| Matrix Materials | Specificity and interference testing (TRL 4-7) | Blood, urine, soil, fire debris, explosive residues |
| System Suitability Mixtures | Instrument performance verification across TRLs | Retention index standards, sensitivity mixtures, resolution mixtures |
Legal Admissibility and TRL Progression
For forensic technologies, progression to higher TRLs (8-9) requires not only analytical validation but also demonstration of reliability under legal standards. The Daubert Standard, followed by U.S. federal courts, requires assessment of whether the technique has been tested, has been subjected to peer review and publication, has a known error rate, and has gained general acceptance in the relevant scientific community [5]. Each of these criteria directly correlates with specific TRL milestones.
Diagram 2: Forensic TRL Progression
Current Applications and Future Directions
Contemporary Implementation
The use of TRLs has expanded significantly beyond their original NASA context. Currently, TRLs are widely employed across diverse sectors including aerospace, defense, energy, healthcare, and forensic science [4]. The U.S. National Institutes of Health has developed three distinct TRL scales specifically tailored for diagnostics, therapeutics, and medical devices, demonstrating the framework's adaptability to different technological domains [4].
In forensic science, recent research has categorized GC×GC applications by TRL to assess their readiness for implementation. As of 2024, techniques for oil spill forensics and decomposition odor analysis have reached the highest maturity levels (TRL 7-8), with 30+ published works each, demonstrating growing acceptance and validation [5]. Other applications, including illicit drug analysis and fingerprint chemistry, are typically assessed at TRL 4-6, indicating ongoing development and validation stages [5].
Limitations and Criticisms
Despite widespread adoption, the TRL framework faces several criticisms. The linear progression inherent to the TRL scale may oversimplify the complex, iterative nature of technological innovation [8]. The framework primarily focuses on technical readiness while potentially overlooking critical external factors such as market dynamics, user adoption barriers, and regulatory requirements [8].
In forensic science, additional limitations emerge regarding the framework's ability to adequately address legal admissibility requirements. The transition from TRL 8 to TRL 9 requires not only technical validation but also successful navigation of legal standards, which may involve considerations beyond purely technical maturity [5]. Furthermore, the absence of complete standardization across forensic disciplines can lead to inconsistencies in TRL assessment [6].
Emerging Adaptations
Recent research has proposed various adaptations to address TRL limitations. The European Association of Research and Technology Organisations (EARTO) has published comprehensive recommendations for using the TRL scale as a research and innovation policy tool [2]. Some researchers have suggested additional levels beyond TRL 9 to account for continuous technology improvement and application in new contexts, though these have not been widely standardized [6].
In forensic science, specialized assessment frameworks are emerging that integrate TRLs with legal admissibility criteria, creating more comprehensive evaluation tools specifically designed for forensic technologies [5]. These integrated approaches assess simultaneous progression along technical, operational, and legal dimensions, providing a more complete readiness assessment for forensic applications.
The Technology Readiness Level framework has evolved significantly from its origins at NASA in the 1970s to become a globally recognized methodology for assessing technological maturity. In forensic science research, TRLs provide an essential structured approach for evaluating the development of analytical techniques from basic principles through to court-admissible evidence. By enabling systematic assessment against both analytical and legal standards, the TRL framework supports forensic researchers, scientists, and drug development professionals in navigating the complex pathway from innovative concept to forensically validated methodology. As the framework continues to evolve through specialized adaptations and integration with legal standards, its utility in forensic science is expected to grow, supporting the development of robust, reliable, and legally admissible analytical technologies.
The Technology Readiness Level (TRL) scale is a systematic metric used to assess the maturity of a particular technology. It provides a common framework for engineers, project managers, and researchers to have a consistent understanding of a technology's evolution, regardless of their technical background [9]. Developed by the National Aeronautics and Space Administration (NASA) in the 1970s, the TRL scale has since been adopted by numerous organizations worldwide, including the European Union (EU), the U.S. Department of Defense, and various industries, to guide research funding, manage project risk, and make critical decisions concerning technology development and transition [2] [9].
The scale consists of nine levels, with TRL 1 being the lowest, representing basic principle observation, and TRL 9 being the highest, indicating a technology that has been proven in its operational environment [1]. For forensic science research, which operates at the intersection of cutting-edge science and the stringent demands of the legal system, understanding and applying the TRL scale is paramount. It helps bridge the gap between innovative research and its practical, court-admissible application, ensuring that new methods are not only scientifically sound but also legally robust [5].
The 9 Technology Readiness Levels: Detailed Breakdown
The following table provides a detailed breakdown of each of the nine TRLs, including their core definition and a representative example from a forensic science context.
Table 1: The 9-Level Technology Readiness Level (TRL) Scale
| TRL | Description | Stage of Development | Forensic Science Example |
|---|---|---|---|
| TRL 1 | Basic principles observed and reported [1] [3]. | Basic Research | Initial scientific research on the interaction of Laser-Induced Breakdown Spectroscopy (LIBS) with explosive residues, establishing theoretical foundations [10]. |
| TRL 2 | Technology concept and/or application formulated [1] [3]. | Basic Research | Formulating a practical application for Next-Generation Sequencing (NGS) to predict physical characteristics (phenotyping) from degraded DNA samples [11]. |
| TRL 3 | Analytical and experimental critical function and/or characteristic proof-of-concept [1] [2]. | Proof of Concept | Laboratory studies demonstrating that Attenuated Total Reflectance Fourier Transform Infrared (ATR FT-IR) spectroscopy can estimate the age of a bloodstain under controlled conditions [10]. |
| TRL 4 | Component and/or breadboard validation in a laboratory environment [1] [2]. | Technology Validation | Basic components of a portable LIBS sensor are integrated and tested in a laboratory, confirming it can detect and classify trace elements from forensic samples [10]. |
| TRL 5 | Component and/or breadboard validation in a relevant environment [1] [2]. | Technology Validation | A prototype handheld X-ray fluorescence (XRF) spectrometer is tested in a simulated crime scene setting to verify it can distinguish between ash from different tobacco brands [10]. |
| TRL 6 | System/subsystem model or prototype demonstration in a relevant environment [1] [2]. | Demonstration | A fully functional Forensic Bullet Comparison Visualizer (FBCV) prototype is demonstrated in a ballistics laboratory, using advanced algorithms to compare bullet striations with high objectivity [11]. |
| TRL 7 | System prototype demonstration in an operational environment [1] [2]. | System Demonstration | A Next Generation Identification (NGI) System prototype, integrating palm prints, facial recognition, and iris scans, is piloted in a select law enforcement agency for real-time identification [11]. |
| TRL 8 | Actual system completed and qualified through tests and demonstrations [1] [2]. | System Qualified | The Integrated Ballistic Identification System (IBIS) is completed, "flight qualified," and integrated into the workflows of national law enforcement agencies for routine comparison of ballistic evidence [11]. |
| TRL 9 | Actual system proven through successful deployment in an operational setting [1] [3]. | Mission Proven | Traditional 1D Gas Chromatography-Mass Spectrometry (GC-MS) is used successfully for years in forensic laboratories worldwide, with its results routinely accepted as evidence in court [5]. |
The progression through the TRL scale is a logical and sequential process. The following diagram visualizes this pathway from basic research to operational deployment.
Figure 1: The sequential progression of a technology through the nine Technology Readiness Levels, grouped into three major phases.
The Scientist's Toolkit: Key Reagents and Technologies in Modern Forensic Research
The advancement of forensic technologies relies on a suite of sophisticated reagents, instruments, and computational tools. The following table details several key solutions that are central to contemporary forensic science research and development.
Table 2: Key Research Reagent Solutions in Modern Forensic Science
| Tool/Technology | Primary Function in Forensic Research |
|---|---|
| Next-Generation Sequencing (NGS) | Provides high-precision analysis of entire genomes or specific regions from minute, damaged, or complex DNA mixtures, enabling enhanced identification and phenotypic prediction [11]. |
| Comprehensive Two-Dimensional Gas Chromatography (GC×GC) | Separates complex mixtures of volatile and semi-volatile compounds (e.g., drugs, ignitable liquids, decomposition odors) with significantly higher peak capacity and sensitivity than traditional 1D GC [5]. |
| Raman Spectroscopy | Provides non-destructive, molecular-level analysis of evidence such as inks, pigments, and pharmaceuticals through measurement of vibrational energy, with advancements in portability for crime scene use [10]. |
| Isotope Ratio Analysis | Determines the geographical origin of materials (e.g., water, drugs, human remains) by measuring the unique signature of stable isotopes within a sample [11]. |
| Artificial Intelligence (AI) & Machine Learning | Used to analyze complex datasets, including mixed DNA profiles, fingerprint data, and photograph comparisons, to draw objective conclusions and assist in pattern recognition [11]. |
| Omics Techniques (Genomics, Proteomics, Metabolomics) | Allow for a comprehensive, systematic study of biological samples for species identification, phylogenetics, and interpreting forensically relevant behavioral characteristics at the genetic level [11]. |
| Carbon Dot Powders | Applied to latent fingerprints to make them fluorescent under UV light, overcoming challenges of low sensitivity and contrast for enhanced visualization and analysis [11]. |
| Immunochromatography Test Strips | Enable rapid, on-site presumptive testing for the presence of specific substances, such as drugs or medications, in bodily fluids [11]. |
Experimental Protocols for TRL Assessment in Forensic Science
To illustrate the practical assessment of technology maturity, below is a detailed experimental methodology for a specific forensic technique as it progresses through mid-level TRLs.
Case Study: Estimating Bloodstain Age via ATR FT-IR Spectroscopy
Determining the time since deposition (TSD) of a bloodstain is a critical yet challenging task in forensic investigations. The following protocol outlines the key experiments for validating ATR FT-IR spectroscopy combined with chemometrics for this purpose, corresponding to TRLs 3 through 5 [10].
- Objective: To develop and validate a non-destructive, spectroscopic method for accurately estimating the age of bloodstains at crime scenes.
- Primary Instrumentation: Attenuated Total Reflectance Fourier Transform Infrared (ATR FT-IR) Spectrometer. This instrument measures the absorption of infrared light by the sample, creating a molecular "fingerprint" without destroying the evidence.
Workflow Overview: The experimental process for developing this method involves a structured workflow from sample preparation to data analysis, with increasing environmental relevance at each stage.
Figure 2: Experimental workflow for developing a bloodstain age estimation method using ATR FT-IR spectroscopy.
Key Experimental Steps:
- Sample Preparation (TRL 3): Fresh human blood samples are deposited onto a range of forensically relevant substrates (e.g., glass, cotton, wood). These samples are stored under tightly controlled laboratory conditions (constant temperature, humidity, and light exposure) and aged for predetermined time intervals [10].
- Data Acquisition and Proof of Concept (TRL 3): Using the ATR FT-IR spectrometer, spectra are collected from each bloodstain at each aging interval. The initial goal is to analytically observe and report systematic changes in the infrared spectrum (e.g., changes in amide bands, oxidation markers) that correlate with the passage of time.
- Chemometric Model Building: The collected spectral data is analyzed using chemometric techniques (e.g., principal component analysis, partial least squares regression). The objective is to develop a statistical calibration model that correlates specific spectral features with the known age of the stain.
- Laboratory Validation (TRL 4): The developed model is tested against a new, independent set of laboratory-created bloodstains. This step validates that the integrated system (spectrometer + model) can accurately and reliably estimate the age of an "unknown" stain within the laboratory environment.
- Relevant Environment Validation (TRL 5): The technology is tested in a simulated operational environment. Bloodstains are created and exposed to a range of variable, but still monitored, conditions (e.g., fluctuating temperature and humidity, different substrate textures) to assess the robustness and accuracy of the method outside ideal lab conditions.
The Critical Bridge: From Research to Courtroom Admissibility
For a forensic technology to progress to TRL 8 or 9, it must overcome a significant hurdle beyond technical validation: legal admissibility. In the United States, the Daubert Standard (and its predecessor, the Frye Standard) governs the admission of expert scientific testimony in federal courts [5]. This standard requires judges to act as gatekeepers, assessing whether:
- The theory or technique has been or can be tested.
- It has been subjected to peer review and publication.
- It has a known or potential error rate.
- It is generally accepted in the relevant scientific community [5].
This legal framework directly impacts the TRL pathway. A technology like GC×GC-MS, while analytically superior to traditional GC-MS in separating complex mixtures, is primarily used in forensic research and is not yet routine in casework [5]. To advance to higher TRLs, research must focus not only on technical improvements but also on intra- and inter-laboratory validation, establishing known error rates, and achieving standardization to meet the stringent Daubert criteria [5]. This underscores that in forensic science, a technology is not truly "mission proven" (TRL 9) until it has been successfully deployed and its results routinely upheld in a court of law.
The Technology Readiness Level scale provides an indispensable, structured framework for guiding the development of forensic technologies from theoretical concepts to court-admissible tools. For researchers and scientists in the field, a clear understanding of this scale is crucial for strategic planning, risk management, and securing funding. The journey from TRL 1 to TRL 9 is not merely a technical challenge but also a legal and procedural one, requiring rigorous validation and standardization to meet the exacting demands of the justice system. By systematically advancing technologies through each TRL, the forensic science community can ensure that innovative research translates into reliable, effective, and legally robust solutions that uphold the integrity of criminal investigations.
First conceived by NASA in the 1970s, the Technology Readiness Level (TRL) scale is a systematic metric used to assess the maturity of a particular technology [2]. It employs a unified scale from 1 to 9, where TRL 1 is the lowest level of maturity and TRL 9 is the highest, representing a technology proven in its operational environment [1]. This guide details the TRL scale's critical role in establishing a common language for technical maturity, its application in risk management and decision-making, and its specific implications for forensic science research and development. For forensic technologies, reaching high TRLs is intricately linked to meeting stringent legal admissibility standards, making the scale an indispensable tool for managing the journey from basic research to courtroom-admissible evidence.
The TRL Scale: A Standardized Metric for Technology Maturity
The TRL scale provides a consistent framework for discussing technical maturity across different types of technology, enabling clearer communication among researchers, project managers, and funding agencies [2]. The scale was originally developed at NASA during the 1970s and formally defined in 1989, and has since been adopted by the U.S. Department of Defense, the European Union, and other organizations worldwide [2]. The scale categorizes the technology development process into four broad phases: Basic Research (TRL 1-3), Applied Research (TRL 4-5), Development (TRL 6-8), and Implementation (TRL 9) [12].
The table below provides a consolidated view of the definitions for each TRL.
Table 1: Technology Readiness Level (TRL) Definitions
| TRL | Description | Key Activities & Evidence |
|---|---|---|
| 1 | Basic principles observed and reported [3] | Translation of basic scientific research into applied R&D; paper studies of basic properties [13]. |
| 2 | Technology concept and/or application formulated [3] | Invention begins; practical applications are conceived based on observed principles. Applications are speculative [13]. |
| 3 | Analytical and experimental critical function and/or proof of concept [3] | Active R&D initiated; analytical and laboratory studies validate proof-of-concept; non-integrated components [1] [13]. |
| 4 | Component and/or breadboard validation in a laboratory environment [3] | Basic technological components are integrated and tested in a laboratory to establish they work together ("low-fidelity" integration) [13]. |
| 5 | Component and/or breadboard validation in a relevant environment [3] | Basic technological components are integrated with realistic supporting elements and tested in a simulated environment ["high-fidelity" laboratory integration] [13]. |
| 6 | System/subsystem model or prototype demonstration in a relevant environment [3] | A representative model or prototype is tested in a relevant environment (e.g., high-fidelity laboratory or simulated operational environment) [13]. |
| 7 | System prototype demonstration in an operational environment [3] | A system prototype is demonstrated in its operational environment (e.g., real-world conditions) [2] [13]. |
| 8 | Actual system completed and qualified through tests and demonstrations [3] | The technology is proven to work in its final form and under expected conditions [13]. |
| 9 | Actual system proven through successful deployment in an operational setting [3] | The actual technology is proven through successful operation in its real-life mission conditions [13]. |
This progression is visualized in the following workflow, which maps the technology development pathway from basic principles to operational deployment.
The Critical Role of TRLs in Risk Management and Decision-Making
The primary purpose of using TRLs is to assist management in making critical decisions concerning the development and transitioning of technology [2]. It serves as one of several essential tools for managing the progress of research and development within an organization.
Appropriate Uses of the TRL Scale
The TRL scale has several key applications in project and risk management [12]:
- Identifying Technical Gaps: It helps pinpoint technical gaps that must be filled to advance the technology to the next readiness level.
- Portfolio Analysis: It allows for a rough portfolio analysis in terms of technology maturity, helping organizations balance their R&D investments.
- Internal Communication: It serves as an effective "shorthand" for discussing project status internally and externally among technical and non-technical stakeholders.
- Structuring Assessments: It provides a guide for technical experts and program managers to structure discussions about the state of development of a single technology, leading to a shared understanding [12].
Limitations and Inappropriate Uses
While powerful, the TRL scale has limitations and should not be used as a sole indicator for all project decisions [2] [12]. It is not designed to:
- Evaluate the difficulty or investment required to advance a technology to the next level.
- Measure the potential impact, benefit, or market for a technology.
- Serve as the single indicator for whether a project should be continued or terminated.
- Effectively evaluate projects designed for the implementation of an existing technology or projects with multiple subprojects that have different user communities or underlying technologies [12].
TRLs in Forensic Science Research: Bridging the Lab and the Courtroom
In forensic science, the path from a novel analytical technique to evidence presented in court is particularly rigorous. The TRL framework provides a structured pathway for this transition, ensuring that new methods are not only scientifically sound but also legally admissible.
The Challenge of Legal Admissibility
For any forensic technology to be adopted for routine casework, it must meet rigorous analytical standards and adhere to legal standards for evidence admissibility [5]. In the United States, the Daubert Standard guides the admission of expert testimony, requiring that the technique or theory has been tested, has been peer-reviewed, has a known error rate, and is generally accepted in the relevant scientific community [5]. Similarly, Canada uses the Mohan Criteria, which focus on relevance, necessity, absence of exclusionary rules, and a properly qualified expert [5]. These legal benchmarks create a high bar for forensic technologies, effectively demanding a high TRL (typically 8 or 9) before they can be relied upon in legal proceedings.
TRL Assessment in Forensic Practice: The Case of GC×GC
The application of TRLs in forensic science is exemplified by the development of Comprehensive Two-Dimensional Gas Chromatography (GC×GC). This technique provides superior separation for complex mixtures found in evidence such as illicit drugs, fingerprint residue, and ignitable liquids [5]. A 2024 review categorized the readiness of GC×GC across seven forensic applications using a simplified readiness scale (Levels 1-4), which aligns with the early to mid-stages of the traditional TRL scale [5]. This assessment revealed that while research is active, the technology has not yet reached routine implementation in forensic laboratories for most applications, underscoring the gap between promising research (low TRL) and court-ready methods (high TRL).
Current Operational Requirements and Research Directions
The National Institute of Justice's Forensic Science Research and Development Technology Working Group identifies operational needs that highlight technologies at various TRLs. For instance, current requirements include [14]:
- Development of novel presumptive tests for evidence analysis at the scene and in the lab (aligning with TRL 2-4).
- Machine Learning tools for mixed DNA profile evaluation (aligning with TRL 3-5).
- Improved methods for determining the geographical origin of remains (aligning with TRL 2-4).
These requirements highlight the ongoing need for research and development to advance the technological maturity of forensic tools.
Implementing the TRL Framework: A Guide for Researchers and Scientists
For researchers, particularly in forensic science and drug development, systematically applying the TRL framework can strategically focus efforts and resources.
Key Methodologies for Advancing TRLs
Advancing a technology through the TRL scale requires specific, validated activities at each stage. The table below outlines the progression using GC×GC and Electronic Toll Collection (ETC) as illustrative examples.
Table 2: Experimental Progression and Methodologies for Advancing TRLs
| TRL Range | Development Phase | Key Experimental Methodologies & Activities | Illustrative Example: GC×GC in Forensics [5] | Illustrative Example: Electronic Toll Collection [12] |
|---|---|---|---|---|
| 1-3 | Basic & Applied Research | Paper studies, formulation of practical applications, analytical studies, laboratory experiments to validate proof-of-concept. | Early proof-of-concept studies resolving simple mixtures; foundational theory development. | Basic research on radio transponders; patent applications for automated tolling; conceptual proposal of a system. |
| 4-5 | Technology Formulation & Validation | Integration of components in a lab; testing in a simulated/relevant environment with realistic supporting elements; documentation of user requirements and interfaces. | Testing GC×GC with multiple component pieces; rigorous testing in simulated forensic sample matrices. | Development and validation of RFID tags in laboratory settings for tracking nuclear materials and livestock. |
| 6-7 | System Demonstration | Prototype demonstration in a relevant/operational environment; testing with representative components under real-world conditions. | Testing a fully functional GC×GC-MS prototype on authentic casework samples (e.g., casework-like drug evidence). | Testing ETC prototypes on closed courses and public roads with temporary installations and limited users. |
| 8-9 | System Operation & Qualification | Final system tested and qualified in its operational environment; successful deployment under mission conditions. | Implementation of a fully qualified GC×GC-MS system in a forensic lab, with intra- and inter-laboratory validation and established error rates. | Large-scale deployments of ETC on public toll roads; use by the general public; federal mandate for use in new toll facilities. |
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key reagents, tools, and technologies that are essential for developing and validating forensic technologies, such as GC×GC, through the TRL scale.
Table 3: Key Research Reagents and Technologies for Forensic Method Development
| Item | Function in Research & Development |
|---|---|
| GC×GC System with Modulator | The core hardware for achieving superior separation of complex mixtures; the modulator is critical for transferring effluent from the first to the second dimension [5]. |
| Diverse Stationary Phase Columns | Provides the independent separation mechanisms needed for the two chromatographic dimensions, crucial for achieving high peak capacity [5]. |
| Mass Spectrometry (MS) Detectors | Enables definitive identification of separated analytes; high-resolution and time-of-flight MS are used for advanced detection [5]. |
| Certified Reference Materials | Essential for method validation, calibration, and determining retention factors (Rf values) to compare known and unknown substances [15]. |
| Standardized Data Processing Software | Critical for handling the complex data generated, ensuring analyses are transparent, reproducible, and resistant to cognitive bias [16]. |
| Validated Statistical Models (e.g., Likelihood Ratios) | Provides a logically correct framework for the interpretation of evidence, which is necessary for meeting legal admissibility standards [16]. |
Strategic Planning with the TRL Scale
To effectively use the TRL scale, research teams should:
- Conduct an Initial TRA: Perform a baseline Technology Readiness Assessment to establish a consensus on the current TRL.
- Define TRL Exit Criteria: For each project phase, define specific, measurable criteria that must be met to advance to the next TRL. This is particularly important for TRL 6 and above, where demonstration in relevant and operational environments is required.
- Align with Legal Standards Early: For forensic technologies, integrate the requirements of the Daubert Standard or Mohan Criteria into the development process from TRL 4 onwards. This includes planning for peer-reviewed publication, error rate analysis, and intra-laboratory validation [5].
- Use TRLs for Gap Analysis: Regularly use the TRL framework to identify the most significant technical and validation gaps preventing progression and focus development efforts accordingly.
The Technology Readiness Level scale is far more than a simple checklist; it is a fundamental framework for creating a common language around technological maturity. Its disciplined application enables effective risk management, strategic decision-making, and clear communication among all stakeholders in the research and development process. For forensic scientists and drug development professionals, the journey to a high TRL is synonymous with the journey to producing reliable, defensible, and legally admissible results. By systematically advancing technologies through the TRL scale, researchers can bridge the critical gap between a promising scientific concept and a trusted tool that delivers impact in the real world.
The Technology Readiness Level (TRL) scale is a systematic metric used to assess the maturity of a particular technology. It is a nine-level scale that provides a common understanding of technology status, facilitating communication and decision-making regarding research and development funding and technology transition. The scale was originally developed by NASA in the 1970s and has since been adopted by numerous major organizations worldwide, including the U.S. Department of Defense (DOD) and the European Union (EU) [2]. The TRL framework enables consistent and uniform discussions of technical maturity across different types of technology, helping management assess the progression of technologies from basic principles (TRL 1) to full operational deployment (TRL 9) [1] [2].
Historical Development and Institutional Adoption
NASA: The Originator
The TRL methodology was conceived at NASA in 1974 by Stan Sadin and was formally defined in 1989 [2]. The original NASA scale contained seven levels, which was later expanded to the nine-level scale that has become the standard [2]. NASA's early use of the scale is exemplified by its application to assess the technology readiness of the proposed JPL Jupiter Orbiter spacecraft design [2]. The primary purpose was to help management make informed decisions concerning the development and transitioning of technology within the context of space missions, where reliability and risk mitigation are paramount [1] [2].
U.S. Department of Defense: Adoption for Procurement
The United States Air Force adopted the use of technology readiness levels in the 1990s [2]. This adoption was significantly influenced by a 1999 report by the United States General Accounting Office (now the Government Accountability Office). The report concluded that the DOD took greater risks and attempted to transition emerging technologies at lower maturity levels than private industry, which increased overall program risk. The GAO recommended that the DOD make wider use of technology readiness levels to assess technology maturity prior to transition [2]. In 2001, the Deputy Under Secretary of Defense for Science and Technology formally endorsed the use of TRLs in new major programs [2]. Detailed guidance was subsequently incorporated into the Defense Acquisition Guidebook and the 2003 DOD Technology Readiness Assessment Deskbook [2].
European Union: Policy Integration
The European Space Agency (ESA) adopted the TRL scale in the mid-2000s, with its handbook closely following the NASA definitions [2]. The universal usage of TRL in EU policy was proposed in the final report of the first High Level Expert Group on Key Enabling Technologies [2]. This recommendation was implemented in the EU's Horizon 2020 research and innovation program (2014-2020) and has been retained in subsequent framework programs [2] [17]. The European Commission formally advised EU-funded research and innovation projects to adopt the scale in 2010, making it a standard tool for a vast range of technologies, from space applications to nanotechnology and communication technology [2]. A 2024 European Commission report confirmed the ongoing use of TRLs to analyze technology progression in Horizon Europe, indicating that most projects begin as basic research (TRL 1-3) with nearly half reaching the demonstration stage (TRL 6-8) by the end of the funding period [17].
Table 1: Historical Timeline of TRL Adoption by Major Agencies
| Year | Agency/Entity | Key Adoption/Milestone Event |
|---|---|---|
| 1974 | NASA | TRL methodology conceived by Stan Sadin [2]. |
| 1989 | NASA | Formal definition of a 7-level TRL scale [2]. |
| 1990s | NASA | Adoption of the 9-level TRL scale [2]. |
| 1990s | U.S. Air Force | Early adoption of TRLs [2]. |
| 1999 | U.S. GAO | Influential report recommending wider DOD TRL use [2]. |
| 2001 | U.S. DOD | Deputy USD(S&T) memo endorsing TRL use [2]. |
| 2003 | U.S. DOD | Publication of the DOD TRA Deskbook [2]. |
| Mid-2000s | European Space Agency | Adoption of the TRL scale [2]. |
| 2010 | European Commission | Advice for EU projects to adopt TRL scale [2]. |
| 2014 | European Union | Full integration into Horizon 2020 program [2]. |
| 2024 | European Commission | Report on TRL use in Horizon Europe [17]. |
Comparative Analysis of TRL Definitions Across Agencies
While the core concept of the TRL scale is consistent across major agencies, there are nuanced differences in their specific definitions and applications. The following diagram illustrates the typical technology development pathway mapped to TRL stages.
NASA TRL Definitions
NASA's definitions are grounded in the context of space systems and missions [1] [18]:
- TRL 1: Basic principles observed and reported [18].
- TRL 2: Technology concept and/or application formulated [18].
- TRL 3: Analytical and experimental critical function and/or characteristic proof-of-concept [18].
- TRL 4: Component and/or breadboard validation in laboratory environment [18].
- TRL 5: Component and/or breadboard validation in relevant environment [18].
- TRL 6: System/subsystem model or prototype demonstration in a relevant environment (ground or space) [18].
- TRL 7: System prototype demonstration in a space environment [1] [18].
- TRL 8: Actual system completed and "flight qualified" through test and demonstration [1] [18].
- TRL 9: Actual system "flight proven" through successful mission operations [1] [18].
European Union TRL Definitions
The European Union's definitions, as used in Horizon Europe and other framework programs, are formulated for a broader range of technologies [2]:
- TRL 1: Basic principles observed [2].
- TRL 2: Technology concept formulated [2].
- TRL 3: Experimental proof of concept [2].
- TRL 4: Technology validated in lab [2].
- TRL 5: Technology validated in relevant environment (industrially relevant environment for key enabling technologies) [2].
- TRL 6: Technology demonstrated in relevant environment (industrially relevant environment for key enabling technologies) [2].
- TRL 7: System prototype demonstration in operational environment [2].
- TRL 8: System complete and qualified [2].
- TRL 9: Actual system proven in operational environment (competitive manufacturing for key enabling technologies; or in space) [2].
Table 2: Comparison of TRL Definitions by NASA and the European Union
| TRL | NASA Definition | European Union Definition |
|---|---|---|
| 1 | Basic principles observed and reported [18]. | Basic principles observed [2]. |
| 2 | Technology concept and/or application formulated [18]. | Technology concept formulated [2]. |
| 3 | Analytical and experimental critical function and/or characteristic proof-of-concept [18]. | Experimental proof of concept [2]. |
| 4 | Component and/or breadboard validation in laboratory environment [18]. | Technology validated in lab [2]. |
| 5 | Component and/or breadboard validation in relevant environment [18]. | Technology validated in relevant environment [2]. |
| 6 | System/subsystem model or prototype demonstration in a relevant environment [18]. | Technology demonstrated in relevant environment [2]. |
| 7 | System prototype demonstration in a space environment [18]. | System prototype demonstration in operational environment [2]. |
| 8 | Actual system completed and "flight qualified" [18]. | System complete and qualified [2]. |
| 9 | Actual system "flight proven" through successful mission operations [18]. | Actual system proven in operational environment [2]. |
Department of Defense TRL Practice
The DOD employs TRLs as a key risk management tool within its acquisition process. The definitions are largely aligned with the NASA scale. The DOD has developed specific assessment tools, such as the Technology Readiness Level Calculator by the Air Force Research Laboratory and the Decision Point Tool/Technology Program Management Model (DP/TPMM) adopted by the Defense Acquisition University (DAU) [2]. These tools provide a standardized set of questions and a high-fidelity activity model to assist Technology Managers in planning, managing, and assessing technologies for successful transition [2].
TRLs in Forensic Science Research: A Case Study on GC×GC
The application of the TRL scale extends beyond its aerospace and defense origins. In forensic science, new analytical techniques must undergo rigorous validation before they can be admitted as evidence in courtrooms. The adoption process for Comprehensive Two-Dimensional Gas Chromatography (GC×GC) in forensic applications serves as an illustrative case study [5].
GC×GC is an advanced separation technique that provides significantly higher peak capacity than traditional 1D GC, improving the separation and detectability of analytes in complex mixtures like illicit drugs, toxicological evidence, and decomposition odors [5].
Detailed Experimental Methodology for GC×GC in Forensic Applications:
- Sample Preparation: Forensic evidence (e.g., drug samples, ignitable liquid residues, biological swabs) undergoes appropriate preparation, which may include extraction, purification, and concentration, depending on the sample matrix [5].
- Instrument Configuration:
- Primary Column (1D): The sample is injected onto the first column with a specific stationary phase (e.g., non-polar) [5].
- Modulator: This is the "heart of the GC×GC" system. It captures narrow bands of eluent from the primary column at fixed intervals (modulation period, typically 1–5 seconds) and focuses/re-injects them into the secondary column [5].
- Secondary Column (2D): A much shorter and narrower column with a different stationary phase (e.g., mid-polar or polar) provides a second, rapid separation orthogonal to the first [5].
- Detection: The effluent from the second column is detected, most commonly by Time-of-Flight Mass Spectrometry (TOFMS) or Flame Ionization Detection (FID), which can generate the high data acquisition rates required for the fast separations in the second dimension [5].
- Data Analysis: The result is a two-dimensional chromatogram. Data processing involves peak finding, integration, and compound identification, often using specialized software for nontargeted analysis where a wide range of unknown analytes must be characterized simultaneously [5].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for GC×GC Forensic Applications
| Item/Category | Specific Examples & Functions |
|---|---|
| Chromatography Columns | Primary (1D) Column: e.g., 20-30m length, non-polar phase (DB-5). Provides the first dimension of separation based on volatility [5]. |
| Secondary (2D) Column: e.g., 1-2m length, mid-polar phase (DB-17). Provides the second, rapid separation based on polarity [5]. | |
| Modulator | Thermal or Valve-Based Modulator: Essential component for trapping and re-injecting eluent from the 1D to the 2D column, enabling the two-dimensional separation [5]. |
| Detection Systems | Time-of-Flight Mass Spectrometry (TOFMS): Provides high-speed spectral data for confident compound identification [5]. |
| Flame Ionization Detection (FID): Provides universal, high-speed detection of organic compounds [5]. | |
| Calibration Standards | Alkanes or other Retention Index Markers: Used to standardize retention times in both chromatographic dimensions [5]. |
| Target Analytic Standards: Certified reference materials for qualitative and quantitative analysis of specific forensic targets (e.g., drugs, explosives) [5]. | |
| Data Processing Software | Specialized GC×GC Software: Required for handling the large, complex 2D datasets, including peak deconvolution, alignment, and statistical analysis [5]. |
Legal Readiness and Courtroom Admissibility
For a forensic technology like GC×GC to transition from research to routine casework, it must meet legal standards for the admissibility of scientific evidence, which directly influences its effective TRL [5]. The following diagram visualizes this critical pathway from technology development to legal acceptance.
- Frye Standard (1923): Requires that the scientific technique must be "generally accepted in the relevant scientific community" [5].
- Daubert Standard (1993): Expands on Frye, making the judge a gatekeeper who must consider: (1) whether the theory/technique can be and has been tested; (2) whether it has been subjected to peer review and publication; (3) the known or potential error rate; and (4) the degree of general acceptance within the relevant scientific community [5].
- Federal Rule of Evidence 702: Codifies the Daubert standard, requiring that expert testimony be based on sufficient facts and data, reliable principles and methods, and the reliable application of those methods to the case [5].
- Mohan Criteria (Canada): Requires that expert evidence be relevant, necessary for assisting the trier of fact, absent any exclusionary rule, and presented by a properly qualified expert [5].
Currently, GC×GC research for various forensic applications is at different stages of technology readiness. As of 2024, areas like oil spill forensics and decomposition odor analysis have seen more than 30 publications each, indicating a higher TRL, whereas other applications remain at earlier research stages [5]. For GC×GC to achieve TRL 9 in forensics, future work must focus on intra- and inter-laboratory validation, establishing known error rates, and standardizing methods to meet these legal benchmarks [5].
The TRL scale, pioneered by NASA and subsequently adopted by the Department of Defense and the European Union, has proven to be an enduring and versatile framework for assessing technological maturity. Its value lies in providing a common language for researchers, project managers, and policymakers to communicate status, manage risk, and make critical funding and transition decisions. The case of GC×GC in forensic science demonstrates the scale's applicability beyond its original context, highlighting the intricate journey from basic principle observation to a courtroom-admissible technology. This journey requires not only analytical and experimental validation but also a deliberate process to meet the rigorous standards imposed by legal systems. As research and innovation continue to advance, the TRL scale remains a foundational tool for guiding technologies from concept to proven operational use.
Applying the TRL Scale in Forensic Science: From Instrumentation to Legal Admissibility
The Technology Readiness Level (TRL) scale is a systematic metric used to assess the maturity of a particular technology. Initially developed by NASA in the 1970s, the TRL framework has since been adopted across numerous fields, from aerospace to pharmaceutical development, providing a common language for researchers, funding agencies, and policymakers to evaluate development progress and risk [2]. The scale ranges from TRL 1 (basic principles observed) to TRL 9 (actual system proven in operational settings) [3] [1]. This case study applies the TRL framework to evaluate Comprehensive Two-Dimensional Gas Chromatography (GC×GC), a powerful analytical technique, within the specific context of forensic science research.
GC×GC represents a significant advancement over traditional one-dimensional gas chromatography (1D-GC) by connecting two separate chromatographic columns in series via a modulator, thereby providing vastly increased peak capacity and separation power for analyzing complex mixtures [5] [19]. The technique has gained traction in forensic applications due to its ability to separate and identify compounds in challenging samples such as illicit drugs, ignitable liquid residues, and decomposition odors [5]. This assessment aims to provide researchers, scientists, and drug development professionals with a structured evaluation of GC×GC's maturity across various forensic applications, highlighting both current capabilities and remaining developmental requirements for routine forensic implementation.
Technology Readiness Level Framework
Standard TRL Definitions
The standard TRL scale consists of nine distinct levels, each representing a specific stage of technological maturation. The following table outlines the official definitions from NASA and the European Union, providing a foundational understanding for the subsequent GC×GC assessment.
Table 1: Standard Technology Readiness Level Definitions
| TRL | NASA Definition [1] | European Union Definition [2] |
|---|---|---|
| 1 | Basic principles observed and reported | Basic principles observed |
| 2 | Technology concept and/or application formulated | Technology concept formulated |
| 3 | Analytical and experimental critical function and/or characteristic proof-of-concept | Experimental proof of concept |
| 4 | Component and/or breadboard validation in laboratory environment | Technology validated in lab |
| 5 | Component and/or breadboard validation in relevant environment | Technology validated in relevant environment |
| 6 | System/subsystem model or prototype demonstration in a relevant environment | Technology demonstrated in relevant environment |
| 7 | System prototype demonstration in a space environment | System prototype demonstration in operational environment |
| 8 | Actual system completed and "flight qualified" through test and demonstration | System complete and qualified |
| 9 | Actual system "flight proven" through successful mission operations | Actual system proven in operational environment |
TRL Adaptations for Analytical Techniques and Forensic Science
While the standard TRL scale provides a general framework, specific fields often require tailored interpretations to address domain-specific requirements. In forensic science, analytical techniques must satisfy additional legal standards beyond technical performance, including the Frye Standard, Daubert Standard, and Federal Rule of Evidence 702 in the United States, and the Mohan Criteria in Canada [5]. These legal frameworks require that scientific techniques be generally accepted in the relevant scientific community, have known error rates, and be supported by peer-reviewed publication [5].
For analytical techniques like GC×GC, TRL assessment must consider both technical maturity and legal admissibility. Recent research has proposed a condensed TRL scale (Levels 1-4) specifically for evaluating forensic techniques [5]:
- Level 1: Basic principles and initial proof-of-concept studies published
- Level 2: Experimental validation in controlled laboratory environments
- Level 3: Method optimization and inter-laboratory validation
- Level 4: Technique fully validated and adopted for routine casework
This adapted framework incorporates the stringent requirements of the legal system, providing a more relevant assessment model for forensic technologies.
Fundamental Principles and Technical Advancements
Comprehensive Two-Dimensional Gas Chromatography (GC×GC) is an advanced separation technique that significantly expands upon traditional 1D-GC. The fundamental innovation lies in the connection of two separate chromatographic columns with different stationary phases via a specialized component called a modulator [5] [19]. As Dr. Katelynn Perrault Uptmor explains, "What we do is we collect what's coming out of the primary column, which is the exact same as the column that you'd see in 1D-GC. So you're collecting for short periods of time... and then you use like a fast injection onto a secondary column" [19]. This configuration provides two independent separation mechanisms, dramatically increasing the peak capacity and resolution for complex samples [5].
The modulator, often described as "the heart of GC×GC," functions by collecting effluent from the first dimension column for short periods (typically 1-5 seconds) and then injecting these concentrated packets onto the second dimension column [5]. This process occurs throughout the entire analysis, allowing for comprehensive separation of sample components. The second dimension column typically has a different stationary phase and is much shorter and narrower than the first dimension column, enabling very fast separations that are completed before the next modulation occurs [19]. Detection systems have evolved from standard flame ionization detection (FID) and mass spectrometry (MS) to more advanced methods including high-resolution mass spectrometry (HRMS) and time-of-flight mass spectrometry (TOFMS), as well as dual detection approaches like TOFMS/FID [5].
Key Research Reagent Solutions and Instrumentation
Successful implementation of GC×GC requires specific components and reagents that form the essential toolkit for researchers. The following table details these critical elements and their functions in GC×GC analysis.
Table 2: Essential Research Reagent Solutions and Materials for GC×GC
| Component/Reagent | Function | Application Notes |
|---|---|---|
| Primary Column (1D) | Initial separation based on primary retention mechanism (typically volatility) | Standard GC columns (e.g., 15-30m length, non-polar phase such as DB-5) [5] [19] |
| Secondary Column (2D) | Secondary separation based on different mechanism (typically polarity) | Short, narrow-bore columns (e.g., 1-5m length, polar phase such as PEG) for rapid separation [5] [19] |
| Modulator | Transfers effluent from 1D to 2D column; heart of GC×GC system | Thermal or flow-based modulation; creates pulsed injections for 2D separation [5] |
| Mass Spectrometer Detector | Compound identification and quantification | TOFMS preferred for fast data acquisition; HRMS provides enhanced identification capability [5] |
| Data Processing Software | Handles complex 2D data visualization and analysis | Essential for interpreting contour plots and performing peak deconvolution [19] |
| Standard Reference Materials | Method calibration and quality control | Complex mixtures for system suitability testing (e.g., petroleum products, synthetic mixtures) [5] |
Method Development Workflow
The method development process for GC×GC can be complex, but recent advances have systematized this procedure to enhance accessibility. Dr. Perrault Uptmor and colleagues have developed a simplified workflow that begins with establishing a robust one-dimensional separation, then systematically optimizes key GC×GC parameters including modulation period, oven temperature ramp rates, and temperature offsets [19]. This logical, step-by-step approach minimizes subjective decision-making and enables new users to develop functional methods efficiently. The availability of modeling software to predict chromatographic behavior further streamlines this process, allowing researchers to simulate separations before laboratory implementation [19].
TRL Assessment of GC×GC in Forensic Applications
Current State of GC×GC Across Forensic Domains
GC×GC has been investigated for numerous forensic applications, with varying degrees of maturity across different subfields. The following table provides a comprehensive TRL assessment based on current literature and implementation status as of 2024.
Table 3: TRL Assessment of GC×GC Across Forensic Applications
| Forensic Application | Current TRL (1-9) | Key Demonstrations | Limitations & Research Needs |
|---|---|---|---|
| Illicit Drug Analysis [5] | 4-5 | Non-targeted analysis of complex drug samples; enhanced separation of co-eluting compounds [5] [1] [20] | Limited intra-/inter-laboratory validation; unknown error rates for casework [5] |
| Toxicological Evidence [5] | 4 | Comprehensive screening for unknown substances in biological samples [5] [21] [5] | Lack of standardized methods for complex matrices; need for reference databases [5] |
| Fingerprint Residue Analysis [5] | 3-4 | Chemical profiling of fingermark composition [5] [20] [2] | Preliminary research stage; requires validation for individualization [5] |
| Odor Decomposition Analysis [5] | 4 | Characterization of volatile organic compounds from decomposing remains [5] | Limited quantitative studies; need for standardized sampling protocols [5] |
| CBNR Forensics [5] | 3-4 | Analysis of chemical, biological, nuclear, and radioactive substances [5] | Early development; requires demonstration in realistic scenarios [5] |
| Petroleum Analysis (Arson & Oil Spills) [5] | 5-6 | ASTM methods for fuel analysis; source identification for oil spills [5] | Highest maturity among applications; approaching routine use for specific analyses [5] |
Detailed Experimental Protocols for TRL Advancement
Protocol for Illicit Drug Analysis (Targeting TRL 5-6)
Objective: Develop and validate a GC×GC-MS method for non-targeted screening of novel psychoactive substances in complex mixtures.
Materials and Equipment:
- GC×GC system equipped with liquid nitrogen or quad-jet thermal modulator
- TOF mass spectrometer detector capable of ≥ 50 Hz acquisition rate
- Primary column: Rxi-5Sil MS (30 m × 0.25 mm i.d. × 0.25 μm df)
- Secondary column: Rxi-17Sil MS (1.5 m × 0.18 mm i.d. × 0.18 μm df)
- Certified reference materials of target analytes
- Internal standard solution (e.g., deuterated analogs)
Method Parameters:
- Injector: 280°C, splitless mode (1 μL injection)
- Carrier gas: Helium, constant flow (1.0 mL/min)
- Oven program: 60°C (hold 1 min), then 5°C/min to 320°C (hold 5 min)
- Modulation period: 6 s (1.5 s hot pulse)
- Transfer line temperature: 280°C
- MS source temperature: 230°C
- Mass range: 40-550 m/z
- Acquisition rate: 100 Hz
Validation Procedure:
- Prepare calibration standards (0.1-100 μg/mL) in simulated street drug matrix
- Establish retention time stability in 2D space (RSD < 2%)
- Determine limits of detection and quantification for target compounds
- Assess intra-day and inter-day precision (n=6, RSD < 15%)
- Conduct ruggedness testing across multiple instruments and operators
- Perform comparative analysis with certified reference methods
This protocol addresses key requirements for advancing TRL by incorporating method validation elements necessary for eventual legal admissibility, including determination of error rates and demonstration of reproducibility [5].
Protocol for Ignitable Liquid Residue Analysis (Targeting TRL 6-7)
Objective: Establish standardized GC×GC method for classification and source identification of fire debris extracts.
Materials and Equipment:
- GC×GC system with cryogenic modulator
- FID and TOFMS detectors in parallel configuration
- Primary column: DB-1HT (30 m × 0.25 mm i.d. × 0.25 μm df)
- Secondary column: DB-17 (2 m × 0.18 mm i.d. × 0.18 μm df)
- ASTM Ignitable Liquid Reference Collection
- Passive headspace concentration devices (e.g., activated charcoal strips)
Method Parameters:
- Injector: 250°C, split ratio 50:1
- Oven program: 40°C (hold 2 min), 3°C/min to 280°C (hold 10 min)
- Modulation period: 8 s
- FID data acquisition rate: 100 Hz
- MS acquisition rate: 50 Hz
Standardized Analysis Workflow:
- Extract ignitable liquid residues using passive headspace concentration
- Analyze samples alongside reference collection using standardized conditions
- Apply pattern recognition algorithms (PCA, HCA) to 2D chromatographic data
- Establish match criteria based on retention alignment and peak ratio consistency
- Conduct inter-laboratory study to establish reproducibility metrics
- Develop statistical models for classification confidence estimation
This protocol builds upon existing petroleum applications where GC×GC has achieved higher TRLs, adapting established methodologies specifically for forensic evidentiary standards [5].
Visualizing the TRL Assessment Pathway for GC×GC
The following diagram illustrates the technology development pathway for GC×GC in forensic applications, mapping specific requirements and milestones against TRL progression:
GC×GC TRL Assessment Pathway
This visualization illustrates how most forensic applications of GC×GC currently cluster at TRL 4, with petroleum analysis representing the most advanced application. The diagram highlights the critical pathway through legal admissibility requirements that must be traversed to achieve higher TRLs in forensic contexts.
Comprehensive Two-Dimensional Gas Chromatography represents a technologically advanced analytical platform with significant potential for forensic applications. Current TRL assessments indicate that most forensic uses remain at middle readiness levels (TRL 4-5), characterized by successful laboratory validation but limited implementation in routine casework [5]. Petroleum analysis for arson investigation and oil spill tracing represents the most mature application, approaching TRL 6 in some specific methodologies [5].
The primary barriers to broader adoption and higher TRLs are not primarily technical, but rather relate to validation and standardization requirements necessary for legal admissibility. Future research should prioritize intra- and inter-laboratory validation studies, establishment of standardized methods, determination of error rates, and development of reference databases [5]. Additionally, continued simplification of method development processes, as championed by researchers like Dr. Perrault Uptmor, will enhance accessibility and promote wider adoption in forensic laboratories [19].
For researchers and drug development professionals, GC×GC offers powerful capabilities for non-targeted analysis of complex mixtures, with particular value in novel psychoactive substance identification and herbal medicinal product characterization [21] [5]. Strategic investment in validation studies and standardization efforts will be essential to advance the TRL of GC×GC across forensic applications, ultimately enabling this powerful analytical technique to reach its full potential in legal contexts.
Technology Readiness Levels (TRLs) provide a systematic metric for assessing the maturity of a particular technology. The scale was first developed by NASA and has since been widely adopted across research and industry, including by the European Union's Horizon Europe program and the U.S. Department of Energy [6] [2]. This nine-level scale ranges from TRL 1 (basic principles observed) to TRL 9 (actual system proven in operational environment), offering a common language for researchers, funders, and policymakers to consistently evaluate technological maturity [1]. For forensic science, integrating the TRL framework with strategic research priorities ensures that promising scientific developments methodically advance from fundamental research to validated tools that strengthen the criminal justice system.
The forensic science community faces significant challenges, including the need to strengthen the validity, reliability, and consistency of forensic methods [22]. A disciplined TRL approach provides a structured pathway to address these challenges by quantifying progress and establishing statistically rigorous measures of accuracy [22]. This alignment is essential for transitioning innovative forensic technologies from laboratory concepts to operational tools that deliver actionable intelligence, support examiner conclusions, and ultimately uphold public trust in forensic science.
The TRL Scale: Definitions and Forensic Science Applications
Standard TRL Definitions and Descriptions
The TRL scale provides a standardized method for estimating technology maturity during program acquisition phases [2]. The following table details each level with specific forensic science context and interpretations.
Table 1: Technology Readiness Levels (TRLs) and Corresponding Forensic Science Applications
| TRL | General Definition | Forensic Science Application & Interpretation |
|---|---|---|
| TRL 1 | Basic principles observed and reported [2]. | Initial scientific research begins; fundamental observations of physical, chemical, or biological principles relevant to forensic evidence are documented [1]. |
| TRL 2 | Technology concept and/or application formulated [2]. | Practical forensic application is proposed based on initial principles. Technology remains speculative with no experimental proof of concept [1] [2]. |
| TRL 3 | Experimental proof of concept [2]. | Active R&D begins with analytical and laboratory studies. A proof-of-concept model is constructed to validate the forensic application's feasibility [1]. |
| TRL 4 | Technology validated in laboratory environment [2]. | Multiple component pieces of the forensic technology (e.g., assay, instrument, software) are tested and integrated in a laboratory setting [1]. |
| TRL 5 | Technology validated in relevant environment [2]. | A breadboard technology (e.g., prototype instrument) undergoes rigorous testing in a simulated operational environment, such as a mock laboratory [1]. |
| TRL 6 | Technology demonstrated in relevant environment [2]. | A fully functional prototype or representational model is demonstrated in a relevant forensic environment, such as a controlled laboratory setting [1]. |
| TRL 7 | System prototype demonstration in operational environment [2]. | A working model of the forensic technology is successfully demonstrated in a real-world operational environment, such as an active crime laboratory [1] [2]. |
| TRL 8 | System complete and qualified [2]. | The technology is fully developed, "flight qualified," tested, and ready for implementation into existing forensic laboratory workflows or technology systems [1]. |
| TRL 9 | Actual system proven in operational environment [2]. | The technology has been successfully deployed and proven effective through routine use in casework by forensic service providers [1]. |
TRL Pathway for Forensic Technologies
The following diagram illustrates the logical progression of a forensic technology through the TRL pathway, from basic research to operational deployment.
Aligning Forensic Science Strategic Priorities with the TRL Pathway
The National Institute of Justice (NIJ) and National Institute of Standards and Technology (NIST) have established clear strategic priorities to address critical gaps and opportunities in forensic science [23] [22]. These priorities can be systematically advanced by mapping them to appropriate TRL stages, ensuring research investments effectively mature technologies toward operational implementation.
Foundational Research (TRL 1-3)
Strategic Focus: Support foundational research to assess the fundamental scientific basis of forensic analysis [23]. This aligns with NIST's "grand challenge" to quantify and establish statistically rigorous measures of accuracy and reliability [22].
- Objective II.1: Foundational Validity and Reliability - Research to understand the fundamental scientific basis of forensic disciplines and quantify measurement uncertainty [23].
- Objective II.2: Decision Analysis - Measurement of the accuracy and reliability of forensic examinations through black-box and white-box studies, including research on human factors [23].
- Objective II.4: Stability, Persistence, and Transfer - Study the effects of environmental factors, time, and transfer mechanisms on evidence integrity [23].
Experimental Protocol: Fundamental Study on Evidence Transfer
- Aim: To establish baseline data on the transfer and persistence of a novel trace evidence type (e.g., organic gunshot residue).
- Methodology:
- Sample Collection: Apply controlled quantities of material to varying substrates (cotton, polyester, skin simulant).
- Environmental Exposure: Expose samples to controlled environmental conditions (time, temperature, humidity).
- Analysis: Analyze samples at set time intervals using targeted analytical techniques (e.g., LC-MS/MS, SEM-EDS).
- Data Modeling: Quantify decay rates and model persistence under different conditions using statistical software.
Applied Research and Development (TRL 4-6)
Strategic Focus: Advance applied R&D to meet the practical needs of forensic practitioners [23]. This includes developing new methods and automating tools to support examiners, directly addressing the need for rapid analyses and new insights from complex evidence [22].
- Objective I.2: Novel Technologies and Methods - Investigate nontraditional aspects of evidence (e.g., microbiome, nanomaterials) and develop reliable field-deployable technologies [23].
- Objective I.4: Expediting Actionable Information - Develop methods and workflows to enhance investigations, including expanded triaging tools and technologies for forensic operations at the scene [23].
- Objective I.5: Automated Tools - Develop objective methods to support interpretations, including technology to assist with complex mixture analysis and algorithms for quantitative pattern evidence comparisons [23].
Experimental Protocol: Validation of a Novel Field Screening Device
- Aim: To validate the sensitivity, specificity, and reliability of a new biosensor for detecting fentanyl in mixtures (TRL 4-5).
- Methodology:
- Calibration: Calibrate the biosensor against certified reference standards of fentanyl and common cutting agents.
- Blinded Testing: Perform blinded analysis of prepared samples (n>100) with varying compositions and concentrations.
- Comparison: Compare results with laboratory-grade confirmation analysis (e.g., GC-MS) to determine false positive/negative rates.
- Robustness Testing: Evaluate performance under different environmental conditions (e.g., temperature, humidity) likely to be encountered in the field.
Table 2: Essential Research Reagents and Materials for Forensic R&D
| Category/Item | Specific Example | Function in Forensic R&D |
|---|---|---|
| Certified Reference Materials | Seized Drug Standards, NIST Standard Reference Materials | Provide a known chemical identity and purity for instrument calibration, method validation, and quality control. |
| Biological Sample Collections | Characterized Bloodstain Cards, Saliva Samples | Used for developing and validating methods for body fluid identification, DNA extraction, and analysis. |
| Calibration Standards | ISO/IEC 17025 Accredited Mass Spec Calibrants | Ensure analytical instruments (e.g., GC-MS, LC-MS/MS) provide accurate and reproducible quantitative results. |
| Proteomic & Genomic Reagents | STR Kits, PCR Master Mixes, Proteinase K | Essential reagents for DNA amplification, sequencing, and body fluid proteomics in forensic biology. |
| Trace Evidence Substrates | Certified Fiber Standards, GSR Collection Stubs | Provide consistent materials for testing evidence collection efficiency, transfer studies, and analytical sensitivity. |
System Integration and Operational Implementation (TRL 7-9)
Strategic Focus: Maximize the impact of forensic science R&D by ensuring successful transition from research to practice [23]. This involves rigorous demonstration in operational environments and supporting widespread adoption.
- Objective III.2: Support Implementation - Assist technology transition, demonstrate and test new methods, and pilot implementation into practice [23].
- Objective I.7: Practices and Protocols - Optimize analytical workflows, implement new technologies, and evaluate laboratory quality system effectiveness [23].
- Objective III.4: Examine Role and Value - Evaluate the implementation of new policies and practices and understand the costs and benefits of new forensic science services [23].
Experimental Protocol: Pilot Implementation of an AI-Based Tool for Pattern Evidence
- Aim: To assess the operational impact and practical workflow integration of a software tool for cartridge case comparison (TRL 7-8).
- Methodology:
- Beta Deployment: Install the software in a live, but initially isolated, laboratory environment.
- Procedural Development: Collaborate with examiners to develop standard operating procedures (SOPs) for tool use.
- Parallel Testing: Run a set of known and blind casework samples through both traditional and new automated methods.
- Impact Assessment: Collect data on analysis time, reproducibility, result concordance, and user feedback to perform a cost-benefit analysis.
The following workflow diagram synthesizes the alignment between strategic priorities, research activities, and the TRL pathway.
Aligning forensic science research and development with the Technology Readiness Level pathway provides a disciplined, strategic framework for advancing the field. By mapping the NIJ's strategic priorities—from foundational and applied research to implementation and workforce development—onto the structured progression of the TRL scale, stakeholders can more effectively manage the innovation lifecycle. This alignment ensures that public investments in forensic R&D consistently deliver validated, reliable, and impactful technologies that strengthen the criminal justice system. The ultimate goal is a robust, scientifically sound forensic practice, where new methods are not only proven in laboratory settings but are also effectively integrated into operational environments to deliver justice.
In forensic science research, a significant gap often exists between a technology's technical maturity and its acceptance as admissible evidence in a court of law. While the Technology Readiness Level (TRL) scale provides a systematic framework for assessing technological maturity, it does not guarantee that a method will meet the rigorous standards of the legal system. The journey from a promising concept in the laboratory (TRL 1-3) to evidence that can withstand legal scrutiny requires careful navigation of legal admissibility standards, primarily the Frye, Daubert, and Mohan standards in North America. For researchers, scientists, and drug development professionals, understanding this bridge is crucial for ensuring that their analytical methods, particularly novel techniques like comprehensive two-dimensional gas chromatography (GC×GC), can ultimately serve justice.
This guide provides a comprehensive framework for aligning technology development with legal admissibility requirements, offering specific protocols and strategies to bridge these traditionally separate domains. The stakes are high: failure to meet these standards can result in the exclusion of expert testimony, potentially undermining cases that rely on cutting-edge scientific analysis. By integrating legal readiness considerations into each stage of technology development, from basic research through validation and deployment, forensic scientists can build a more robust and legally defensible foundation for their work.
Understanding Technology Readiness Levels (TRLs) in Forensic Contexts
The TRL Scale: From Basic Principles to Operational Use
The Technology Readiness Level scale is a systematic metric, originally developed by NASA, for assessing the maturity of a particular technology [1] [6]. It consists of nine levels, with TRL 1 being the lowest (basic principles observed) and TRL 9 being the highest (actual system proven in operational setting) [1]. The scale has been widely adopted across multiple sectors, including forensic science, to provide a common language for tracking development progress.
In forensic applications, the TRL scale takes on specific significance, as summarized in the table below:
Table: Technology Readiness Levels (TRLs) and Their Forensic Science Applications
| TRL | Description | Forensic Science Context | Typical Activities |
|---|---|---|---|
| TRL 1 | Basic principles observed and reported [1] [3] | Initial research on novel analytical techniques | Paper studies of technology's basic properties [3] |
| TRL 2 | Technology concept and/or application formulated [1] [3] | Practical forensic applications conceived | Analytic studies to map technique to evidence types [3] |
| TRL 3 | Analytical and experimental critical function and/or proof of concept [1] [3] | First laboratory studies on forensic samples | Proof-of-concept model construction; viability studies [1] |
| TRL 4 | Component and/or validation in a laboratory environment [1] [3] | Multiple component pieces tested with forensic matrices | Integration of "ad hoc" hardware in laboratory [3] |
| TRL 5 | Component and/or validation in a simulated environment [1] [3] | Rigorous testing with simulated casework samples | Laboratory integration of components; testing in realistic simulations [1] [3] |
| TRL 6 | System/subsystem model or prototype demonstration in a simulated environment [1] [3] | Prototype testing in simulated operational forensic lab | Testing in simulated operational environment [3] |
| TRL 7 | Prototype ready for demonstration in an appropriate operational environment [1] [3] | Prototype demonstrated in operational crime laboratory | Prototype field testing in forensic laboratory [1] [3] |
| TRL 8 | Actual technology completed and qualified through tests and demonstrations [1] [3] | Technology validated for specific forensic applications | Developmental testing and evaluation against operational requirements [3] |
| TRL 9 | Actual technology proven through successful deployment in an operational setting [1] [3] | Routine use in casework and successful admission in court | Actual application under real-life casework conditions [3] |
The Critical Pre-Development Phase: TRL 0 in Forensic Innovation
Beyond the established TRL 1-9 framework, some innovation models recognize TRL 0, representing the "idea" stage before formal research begins [24]. In forensic science, this stage involves exploratory activities such as reviewing emerging analytical literature, identifying latent forensic needs, and formulating "what if" questions about potential applications of new technologies. This phase is characterized by curiosity-driven exploration and cross-disciplinary knowledge gathering, which may later crystallize into focused research projects with forensic applications [24].
Legal Standards for Expert Testimony and Scientific Evidence
The Frye Standard: General Acceptance Test
The Frye Standard, originating from the 1923 case Frye v. United States, established that expert testimony must be based on scientific methods that are "generally accepted" in the relevant scientific community [25] [26] [27]. Under Frye, the court's inquiry is relatively straightforward: has the technique gained widespread acceptance in its field? If not, the testimony is inadmissible. This standard places the decision about scientific validity primarily in the hands of the scientific community rather than the judiciary [25]. The standard is aptly described as operating in the "twilight zone" between experimental and demonstrable stages of scientific principles [27].
The Daubert Standard: A Multi-Factor Reliability Test
The Daubert Standard, established in the 1993 Supreme Court case Daubert v. Merrell Dow Pharmaceuticals, Inc., superseded Frye in federal courts and expanded the judge's role as a "gatekeeper" of scientific evidence [25] [28] [26]. Daubert emphasizes that expert testimony must be both relevant and reliable, with judges conducting a preliminary assessment of the scientific validity of the underlying reasoning and methodology [26]. The standard employs a flexible, multi-factor test, including:
- Testability: Whether the theory or technique can be (and has been) tested [26] [27]
- Peer Review: Whether the method has been subjected to peer review and publication [26] [27]
- Error Rate: The known or potential error rate of the technique [26] [27]
- Standards: The existence and maintenance of standards controlling the technique's operation [26] [27]
- General Acceptance: The degree to which the technique is generally accepted in the relevant scientific community [26] [27]
Later cases including General Electric Co. v. Joiner (1997) and Kumho Tire Co. v. Carmichael (1999) reinforced and expanded Daubert, applying it to all expert testimony, not just scientific evidence [26] [27].
The Mohan Standard: The Canadian Framework
In Canada, the admissibility of expert testimony is governed by the Mohan criteria, established in R. v. Mohan (1994) [5] [28]. This standard requires that expert evidence meet four key criteria:
- Relevance: The evidence must be relevant to the case [5] [28]
- Necessity: The evidence must be necessary to assist the trier of fact [5] [28]
- Absence of Exclusionary Rule: No exclusionary rule prohibits the evidence [5] [28]
- Properly Qualified Expert: A properly qualified expert must present the evidence [5] [28]
Additionally, the court must weigh the probative value of the evidence against its potential prejudicial effect [28].
Table: Comparison of Legal Standards for Expert Testimony Admissibility
| Standard | Jurisdiction | Key Principle | Judge's Role | Primary Focus |
|---|---|---|---|---|
| Frye | Some U.S. State Courts [26] | "General Acceptance" in relevant scientific community [25] | Limited gatekeeper; relies on scientific consensus [25] | Whether the method is widely accepted by scientists [25] |
| Daubert | U.S. Federal Courts and Many States [26] [27] | Reliability and Relevance based on multiple factors [26] | Active gatekeeper; assesses scientific validity [26] | Methodological reliability and application [26] |
| Mohan | Canadian Courts [5] [28] | Relevance, Necessity, and Reliability Threshold [5] [28] | Gatekeeper ensuring evidence meets necessity and relevance criteria [28] | Whether expert evidence is essential for fact-finding [28] |
Bridging the Gap: Aligning Technology Development with Legal Standards
A Conceptual Framework for Integration
The path from technological innovation to legally admissible evidence requires systematic planning throughout the development lifecycle. The following diagram illustrates the parallel maturation of technology readiness and legal admissibility:
Technology Readiness and Corresponding Legal Requirements
To systematically address legal admissibility throughout technology development, forensic researchers should implement specific activities at each TRL stage:
TRL 1-3 (Basic Research to Proof of Concept): At these earliest stages, researchers should document foundational scientific principles, conduct initial feasibility studies with forensic matrices, and identify relevant scientific communities whose acceptance will eventually be required. This establishes the "general acceptance" foundation required by Frye and provides the initial scientific basis for Daubert's reliability factors [5].
TRL 4-6 (Validation to Prototype Development): During these critical maturation stages, focus should expand to include rigorous testing under controlled conditions, preliminary error rate estimation, development of standard operating procedures, and submission of methods for peer review. These activities directly address Daubert factors including testing, error rates, and standards development [5] [26].
TRL 7-8 (Field Testing to Qualification): At these advanced stages, technologies should undergo interlaboratory validation, implementation in simulated casework, refinement of error rate calculations, and training of multiple examiners. These steps build the foundation for demonstrating reliability across multiple settings and operators, addressing requirements under all three legal standards [5].
TRL 9 (Operational Use): Even after full implementation, maintaining admissibility requires ongoing proficiency testing, monitoring case law challenges, publishing case studies, and participating in scientific working groups to maintain "general acceptance" [5].
Experimental Protocols for Validation and Error Rate Determination
Comprehensive Two-Dimensional Gas Chromatography (GC×GC) Case Example
GC×GC represents an illustrative case of an advanced analytical technique undergoing the transition from research to forensic application. The technique provides enhanced separation power for complex mixtures encountered in forensic analysis, including illicit drugs, fire debris, explosives, and ignitable liquids [5]. The following experimental protocol outlines a comprehensive validation approach suitable for meeting legal admissibility standards:
Method Validation Protocol for GC×GC in Forensic Applications
Objective: To establish a legally defensible GC×GC method for the analysis of complex forensic mixtures that meets Daubert reliability factors and satisfies Frye's general acceptance requirement.
Materials and Equipment:
- Comprehensive two-dimensional gas chromatograph with thermal or flow modulator
- Mass spectrometric detector (preferably time-of-flight for untargeted analysis)
- Data processing software with GC×GC capabilities
- Certified reference materials relevant to forensic applications
- Quality control samples representing casework matrices
Experimental Workflow:
System Optimization and Parameter Selection
- Conduct preliminary experiments to determine optimal column combination (stationary phase selectivity)
- Optimize modulation period to ensure at least 3-4 modulations across first dimension peaks
- Establish temperature program parameters for adequate separation within reasonable analysis time
- Document all optimization procedures and rationale for selected parameters
Analytical Performance Characterization
- Determine linearity and range using calibration standards at minimum of 5 concentrations
- Establish limit of detection (LOD) and limit of quantification (LOQ) using signal-to-noise approach and empirical testing
- Evaluate precision through replicate analysis (n≥5) at low, medium, and high concentrations
- Assess carryover and memory effects through blank injections following high concentration samples
Robustness and Reproducibility Testing
- Conduct inter-day precision studies using different analysts over multiple days
- Evaluate minor deliberate variations in method parameters (temperature, flow rates)
- Perform instrument-to-instrument comparison where multiple systems are available
- Implement partial validation when major system components are changed
Reference Material and Quality Assurance
- Incorporate certified reference materials for method verification
- Establish system suitability criteria to be evaluated before each analytical sequence
- Implement quality control samples at defined frequencies throughout analytical batches
- Maintain complete documentation of all validation data for forensic review
Error Rate Determination Protocol
Objective: To establish a scientifically defensible error rate for the GC×GC method, addressing a key Daubert factor [5] [26].
Procedure:
Study Design
- Assemble a representative set of casework samples (minimum n=50, ideally n=100+)
- Include true positive, true negative, and known challenging samples (e.g., mixtures, degraded samples)
- Ensure sample composition reflects actual casework scenarios and matrices
Blinded Analysis
- Code samples to eliminate examiner bias while maintaining ground truth knowledge
- Distribute samples to multiple trained examiners in randomized order
- Require independent interpretation without collaboration
Data Collection and Analysis
- Record all identifications, exclusions, and inconclusive determinations
- Compare examiner results to known ground truth
- Calculate false positive rate, false negative rate, and overall error rate with confidence intervals
- Document all contextual factors that may influence error rates
Proficiency Testing Program
- Establish ongoing internal proficiency testing at regular intervals (e.g., quarterly)
- Participate in external proficiency testing programs when available
- Monitor error rates over time to identify trends or training needs
The following workflow diagram illustrates the integrated validation approach:
Research Reagent Solutions and Method Development Materials
Table: Essential Materials and Resources for Forensic Technology Validation
| Resource Category | Specific Examples | Function in Validation | Legal Standard Addressed |
|---|---|---|---|
| Certified Reference Materials | Certified drug standards, ASTM ignitable liquid standards, controlled substance calibrators | Establish method accuracy and precision; provide traceable quantification | Daubert (testing, standards); Frye (consistency with established methods) |
| Quality Control Materials | In-house reference materials, proficiency test samples, internal standards | Monitor method performance over time; demonstrate ongoing reliability | Daubert (standards and controls); Mohan (reliability threshold) |
| Standard Operating Procedure Templates | ASTM International standards, SWGDRG guidelines, ISO 17025 requirements | Ensure consistent application of methods; document validation protocols | Daubert (standards); All standards (documentation) |
| Data Processing Tools | Chemometric software, statistical packages, data visualization tools | Support objective data interpretation; enable error rate calculations | Daubert (error rate, testing); Mohan (necessity for complex data) |
| Peer-Reviewed Literature | Journal publications, technical reports, validation studies | Demonstrate general acceptance; provide comparative data | Frye (general acceptance); Daubert (peer review) |
| Proficiency Testing Programs | Collaborative testing services, interlaboratory comparisons, internal proficiency tests | Generate error rate data; monitor examiner performance | Daubert (error rate); All standards (competence demonstration) |
The journey from promising forensic technology to legally admissible evidence requires deliberate planning and execution at every stage of development. By integrating legal admissibility considerations into the technology readiness framework, forensic researchers can systematically address the requirements of Frye, Daubert, and Mohan standards throughout the development process. This approach transforms legal readiness from an afterthought into a fundamental component of technology maturation.
For the forensic science community, this bridge between technology and evidence represents both a challenge and an opportunity. By embracing rigorous validation, transparent error rate determination, and active engagement with the scientific and legal communities, researchers can ensure that innovative analytical methods fulfill their potential to serve justice. The framework presented in this guide provides a pathway for building technologies that are not only scientifically sound but also legally defensible, ultimately strengthening the foundation of forensic science in the courtroom.
Overcoming Roadblocks: Navigating the Valley of Death and Funding Gaps in Forensic Tech Development
Technology Readiness Levels (TRL) are a systematic metric used to assess the maturity of a given technology, providing a common understanding of technology status across different types of research and development. The scale ranges from TRL 1 (basic principles observed) to TRL 9 (actual system proven in operational environment) [1]. Originally developed by NASA during the 1970s, the TRL framework has since been adopted across numerous sectors, including forensic science, to help management make decisions concerning technology development and transition [2].
In forensic science, the path from conceptual research to courtroom-admissible technology is particularly challenging. Technologies often progress successfully through early TRL stages (1-3) where basic research and proof-of-concept demonstrations occur, only to encounter significant barriers at mid-TRL levels (4-6) where validation in relevant environments is required [5]. This stalling phenomenon represents a critical inefficiency in forensic science innovation, delaying the implementation of improved methodologies for evidence analysis and ultimately impacting the administration of justice.
The following sections examine the structural, validation, and resource-related challenges that impede progress at these mid-TRL stages, provide specific methodological guidance for overcoming these barriers, and present a strategic framework for advancing forensic technologies toward operational deployment.
The Mid-TRL Transition: Critical Juncture for Forensic Technologies
Defining Mid-TRL Stages in Forensic Science Context
The mid-TRL levels (4-6) represent the crucial transitional phase where technologies move from laboratory curiosity to potentially deployable solutions. In forensic contexts, these stages have specific meanings:
TRL 4: Component and/or validation in a laboratory environment. Basic technological components are integrated to establish that they work together. In forensic terms, this might involve integrating a novel detection method with existing evidence processing workflows using controlled samples [1] [3].
TRL 5: Component and/or validation in a simulated environment. The basic technological components are integrated for testing in a simulated environment. For forensic technologies, this typically means testing with realistic but non-casework samples in conditions that approximate operational forensic laboratories [2].
TRL 6: System/subsystem model or prototype demonstration in a simulated environment. A model or prototype representing a near-desired configuration is tested in a simulated operational environment. For forensic applications, this involves a fully functional prototype tested with authentic or mock casework samples under realistic laboratory conditions [1] [3].
The transition from TRL 4 to TRL 6 is particularly challenging because it requires technologies to demonstrate reliability and validity under increasingly realistic conditions while simultaneously meeting the rigorous standards demanded by the legal system.
Structural and Systemic Barriers at Mid-TRL Levels
Forensic technologies face unique structural barriers that distinguish them from technologies developed for other sectors. These challenges frequently cause promising innovations to stall at mid-TRL levels:
Legal Admissibility Requirements: Unlike commercial technologies, forensic methods must satisfy specific legal standards for admissibility as evidence. In the United States, this includes the Daubert Standard (whether the technique can be and has been tested, whether it has been peer-reviewed, its known or potential error rate, and its general acceptance in the relevant scientific community) or the Frye Standard (general acceptance in the relevant scientific community) [5]. In Canada, the Mohan criteria establish similar requirements for expert evidence [5]. Meeting these standards requires extensive validation studies that many research projects are poorly equipped to conduct.
Prosecutorial Influence and Structural Biases: Forensic labs often operate under significant institutional pressures, particularly when housed within law enforcement or prosecutorial agencies. A 2025 analysis of forensic labs under prosecutorial control highlighted that "a forensic lab in close proximity to prosecutors can create institutional pressures or foster biased practices that undermine scientific integrity" [29]. A 2021 Cornell study found that even minor biases, more likely to occur in forensic units housed within prosecutors' offices, can accumulate and significantly affect trial outcomes [29]. This environment creates risk aversion that prioritizes established methods over innovative approaches, regardless of their potential advantages.
Funding Limitations for Validation Studies: Federal funding for forensic science has faced significant cuts, with the DOJ's proposed FY 2026 budget slashing Paul Coverdell Forensic Science Improvement Grants by roughly 70%, from $35 million to just $10 million [30]. Simultaneously, DNA-specific casework funding remains well below authorized levels. This funding environment particularly impacts mid-TRL validation work, which requires substantial resources but lacks the excitement of basic research or the immediate practical payoff of implemented technologies.
Table 1: Key Legal Standards for Forensic Evidence Admissibility
| Standard | Jurisdiction | Key Criteria | Impact on Mid-TRL Development |
|---|---|---|---|
| Daubert Standard | U.S. Federal Courts & Some States | - Testability- Peer review- Error rates- General acceptance | Requires extensive validation studies before implementation; emphasizes known error rates |
| Frye Standard | Some U.S. States | - General acceptance in relevant scientific community | Creates conservative adoption environment; favors established methods over innovations |
| Federal Rule 702 | U.S. Federal Courts | - Sufficient facts/data- Reliable principles/methods- Proper application | Mandates rigorous documentation and methodological transparency |
| Mohan Criteria | Canada | - Relevance- Necessity- Absence of exclusionary rules- Properly qualified expert | Emphasizes practical necessity and expert qualifications alongside methodological soundness |
Technical and Validation Hurdles at Mid-TRL Stages
Analytical Validation Requirements
For forensic technologies to progress beyond mid-TRL levels, they must overcome significant technical hurdles related to analytical validation. The case of comprehensive two-dimensional gas chromatography (GC×GC) illustrates these challenges well. Although GC×GC has been explored in forensic research for advanced separation of complex evidence including illicit drugs, fingerprint residue, and toxicological evidence, it has not been widely adopted into routine forensic casework [5]. The primary barrier is the extensive validation required to demonstrate reliability across the wide range of potential evidence types.
The validation process for novel forensic technologies must establish:
Robustness and Reproducibility: Techniques must demonstrate consistent performance across different operators, instruments, and environmental conditions. This requires intra-laboratory studies (within the same lab) and inter-laboratory studies (across multiple labs) that are resource-intensive and time-consuming.
Error Rate Quantification: The legal system's emphasis on known error rates, particularly under the Daubert Standard, necessitates comprehensive studies to establish false positive and false negative rates under realistic conditions. This represents a particularly challenging requirement for non-targeted forensic applications where the range of potential analytes is wide [5].
Standard Operating Procedure (SOP) Development: Technologies at mid-TRL levels often lack the detailed SOPs required for routine implementation. Developing these procedures requires iterative refinement and validation that extends beyond typical academic research timelines and resources.
Reference Material and Proficiency Testing Limitations
The availability of appropriate reference materials and proficiency testing programs presents another significant hurdle for forensic technologies at mid-TRL stages. Established techniques benefit from commercially available reference materials and regular proficiency testing programs that support quality assurance. Novel technologies frequently lack these essential resources, creating a barrier to progression beyond TRL 6.
The development of validated reference materials for emerging techniques requires substantial investment and cross-laboratory collaboration. Similarly, establishing statistically valid proficiency testing programs necessitates a critical mass of participating laboratories, creating a chicken-and-egg problem where technologies cannot advance without participating laboratories, but laboratories cannot adopt techniques without established proficiency testing.
Table 2: Technical Validation Requirements Across Mid-TRL Stages
| TRL Level | Primary Validation Focus | Typical Study Design | Common Resource Gaps |
|---|---|---|---|
| TRL 4 | Component integration in lab environment | - Controlled samples- Single laboratory- Optimized conditions | - Authentic casework samples- Multiple instrument platforms- Standardized protocols |
| TRL 5 | Performance in simulated environment | - Mock casework samples- Partial environmental challenges- Preliminary reproducibility | - Multi-laboratory participation- Statistical power for error rates- Proficiency test materials |
| TRL 6 | System demonstration in relevant environment | - Authentic or mock casework- Multiple operators- Environmental challenges | - Large sample sizes for error rates- Long-term stability data- Standardized reporting frameworks |
Methodological Framework for Overcoming Mid-TRL Stagnation
Integrated Validation Protocol for Forensic Technologies
Advancing forensic technologies beyond mid-TRL stagnation requires a systematic approach to validation that addresses both analytical and legal requirements. The following integrated protocol provides a roadmap for technology developers:
Diagram 1: Integrated Validation Pathway for Forensic Technologies
Strategic Funding and Resource Allocation
Overcoming mid-TRL stagnation requires strategic approaches to securing necessary resources amidst constrained funding environments. Successful laboratories have employed several key strategies:
Targeted Grant Applications: Leveraging specialized funding programs focused specifically on validation work. For example, the Capacity Enhancement for Backlog Reduction (CEBR) competitive grant program has been successfully used by laboratories like the Michigan State Police to validate low-input and degraded DNA extraction methods, resulting in a 17% increase in interpretable DNA profiles from complex evidence [30].
Lean Methodologies for Efficiency Implementation: Applying process improvement methodologies to maximize resource utilization. The Louisiana State Police Crime Laboratory implemented Lean Six Sigma principles through an NIJ Efficiency Grant, reducing average turnaround time from 291 days to just 31 days while tripling case throughput from approximately 50 to 160 cases per month [30].
Regional Partnerships and Cost-Sharing: Developing collaborative models across multiple jurisdictions. Shelby County, Tennessee, partnered with the Memphis City Council in 2025 to fund a $1.5 million regional crime lab integrating DNA, ballistics, and digital forensics, reducing reliance on overburdened state labs [30].
These approaches demonstrate how strategic resource allocation can overcome the funding gaps that frequently stall technologies at mid-TRL levels.
Successfully navigating the mid-TRL transition requires access to specialized materials and methodological resources. The following table identifies key components of the technology advancement toolkit, drawing from successful implementations across forensic disciplines.
Table 3: Essential Research Reagent Solutions for Forensic Technology Validation
| Resource Category | Specific Examples | Function in Mid-TRL Advancement | Implementation Considerations |
|---|---|---|---|
| Reference Materials | - Certified reference materials (CRMs)- Mock casework samples- Proficiency test materials | Establishes baseline performance metrics and enables inter-laboratory comparison | Must mimic authentic casework complexity; requires stability characterization |
| Data Analysis Tools | - Statistical analysis packages- Custom algorithms for novel data types- Reference databases | Supports error rate quantification and method comparison studies | Must produce forensically defensible results with transparent methodologies |
| Quality Assurance Frameworks | - Standard operating procedures (SOPs)- Quality control materials- Documentation systems | Ensures adherence to forensic quality standards and regulatory requirements | Must balance comprehensive documentation with practical implementability |
| Instrumentation Platforms | - Multiple instrument models- Different manufacturer platforms- Backup systems for critical studies | Demonstrates method robustness across laboratory environments | Requires significant capital investment; collaborative models can increase access |
| Legal Framework Templates | - Daubert/Kumho compliance checklists- Expert testimony preparation guides- Legal precedent databases | Facilitates preparation for legal challenges and admissibility hearings | Must be updated regularly to reflect evolving case law and standards |
The stalling of forensic technologies at mid-TRL levels represents a critical inefficiency in the forensic science innovation ecosystem. This stalling results from interconnected challenges including stringent legal admissibility standards, extensive validation requirements, resource constraints, and structural biases within forensic systems.
Overcoming these barriers requires a multifaceted approach that integrates methodological rigor, strategic resource allocation, and systemic reform. The validation framework and resource toolkit presented in this work provide a pathway for technology developers to navigate the complex transition from proof-of-concept to courtroom-ready technology.
Furthermore, addressing the systemic issues that contribute to mid-TRL stagnation will require policy interventions, including increased funding for validation studies, enhanced independence of forensic laboratories from law enforcement and prosecutorial influence, and development of more flexible legal standards that balance scientific innovation with procedural safeguards.
As the field continues to evolve in response to emerging technologies and increasing demands for forensic services, overcoming the mid-TRL barrier will be essential for integrating improved methodologies that enhance the reliability, efficiency, and scientific foundation of forensic science practice.
In the specialized field of forensic science research, advancing a technology from a laboratory proof-of-concept to a validated tool used in casework presents a significant challenge. This journey is formally mapped by the Technology Readiness Level (TRL) scale, a systematic metric used to assess the maturity of a particular technology [1] [2]. The critical transition from TRL 4 (component validation in a laboratory environment) to TRL 7 (system prototype demonstration in an operational environment) almost invariably requires a well-executed pilot demonstration and operational testing phase [1]. This phase is not merely a technical hurdle; it is a strategic endeavor that demands specific funding and resources. The forensic science sector currently faces a climate of uncertainty, with pauses or cuts to federal grants for scientific research leaving agencies unable to purchase new equipment or conduct essential studies [31]. Within this constrained environment, a structured approach to securing support for pilot projects becomes paramount for transforming innovative research into practical, real-world solutions that enhance the accuracy and efficiency of the criminal justice system.
The Technology Readiness Level (TRL) Framework in Forensic Science
The TRL framework provides a common language for researchers, project managers, and funding agencies to consistently discuss technical maturity. Originally developed by NASA, the scale ranges from 1 (basic principles observed) to 9 (actual system proven in successful mission operations) [2]. For forensic science research, the definitions provided by NASA and the European Union offer a clear roadmap for development. The table below summarizes the TRLs most relevant to the pilot and testing phase of a forensic technology.
Table 1: Technology Readiness Levels Relevant to Pilot Testing and Operational Demonstration
| TRL | NASA Definition | European Union Definition | Forensic Science Context |
|---|---|---|---|
| 4 | Component and/or breadboard validation in laboratory environment | Technology validated in lab | Forensic assay or tool functions reliably in a controlled laboratory setting with known samples. |
| 5 | Component and/or breadboard validation in relevant environment | Technology validated in relevant environment | Technology is tested with authentic case-type samples in a simulated operational environment. |
| 6 | System/subsystem model or prototype demonstration in a relevant environment | Technology demonstrated in relevant environment | A fully functional prototype is tested in a lab that mimics a real forensic workflow, including data integration. |
| 7 | System prototype demonstration in a space environment | System prototype demonstration in operational environment | Prototype is deployed and tested in a live, operational crime laboratory setting. |
The primary purpose of using TRLs is to assist management in making decisions concerning the development and transitioning of technology, including decisions about technology funding [2]. A clearly articulated TRL helps funding bodies understand the current maturity of a proposal and the specific risks the requested pilot project is designed to mitigate.
The Contemporary Funding Landscape for Forensic Science
Navigating the funding landscape requires an understanding of both its challenges and opportunities. A key challenge is the persistent issue of funding constraints. As highlighted at the American Academy of Forensic Sciences (AAFS) 2025 conference, federal funding cuts have left agencies and laboratories unable to purchase new equipment or conduct desired research [31]. Heidi Eldridge, a Director at George Washington University, noted that these uncertainties are so severe they have even prevented some researchers from traveling to conferences, stating that "Agencies are trying to do more with less" [31].
Despite these challenges, specific funding interests are actively being pursued. The National Institute of Justice (NIJ) has outlined its anticipated research interests for 2025, which provide a roadmap for areas likely to receive support [32]. These priorities offer valuable guidance for framing pilot-testing proposals to align with funder objectives. Key areas include:
- Foundational or applied R&D to increase the body of knowledge guiding forensic science policy and practice [32].
- Evaluation of existing laboratory protocols or emerging methods, particularly projects that partner with an operational public crime laboratory to have a direct, immediate impact on efficiency and policy [32].
- Innovative research on the use of artificial intelligence (AI) to improve the fairness, accuracy, and effectiveness of criminal justice processes [32].
Securing the necessary backing for a pilot project requires a deliberate and evidence-based strategy. The process can be broken down into key phases, from initial planning to post-pilot evaluation, as illustrated in the following workflow.
Diagram 1: Strategic Workflow for Securing Pilot Project Funding
Define Objectives and Select the Right Project
The first step is to select a project with a high potential for impact and to define its objectives with absolute clarity. A pilot project acts as a proof of concept, validating the practicality and feasibility of the technology in the real world [33] [34]. Objectives must be SMART: Specific, Measurable, Achievable, Relevant, and Time-bound [34]. For a forensic technology, this could mean: "Demonstrate that the rapid DNA sequencing platform can accurately identify unknown biological samples with 99% concordance with standard methods, while reducing processing time by 50% during a 3-month operational pilot in a partner crime lab."
With objectives defined, the next step is to identify a funding source whose priorities align with the project's goals. As noted in Section 3, the NIJ's published research interests are a primary source for forensic scientists [32]. A successful proposal must articulate how the pilot project directly addresses one or more of these stated interests. Furthermore, it is crucial to demonstrate an understanding of the current funding climate's constraints and position the pilot as a cost-effective risk mitigation strategy that prevents larger, more costly failures later [31] [33]. The proposal should clearly state how the project will advance the technology's TRL, providing funders with a concrete maturity milestone.
Develop a Comprehensive Project Plan
A compelling proposal is backed by a detailed project plan that serves as a blueprint for execution. This plan must include [34]:
- Scope and Timeline: A clear definition of the pilot's boundaries, along with a timeline showing the start date, key milestones, and the end date.
- Resource Allocation: A detailed budget and a list of necessary resources, including personnel (with assigned roles), equipment, software, and any partner laboratory infrastructure.
- Risk Management: Proactive identification of potential risks (e.g., technical integration failures, data compatibility issues, sample availability) and strategies to manage them.
- Communication Plan: An outline of how the project team, funders, and partner laboratories will stay informed throughout the project.
Secure Stakeholder Support
Stakeholder engagement is a critical yet often overlooked step. Support from operational forensic laboratories is essential, as they provide the realistic environment needed for TRL 6 and 7 testing. Engaging these stakeholders early fosters collaboration and ensures the project addresses real-world needs [34]. Furthermore, demonstrating partnership with an end-user laboratory significantly strengthens a funding proposal by proving the project's relevance and pathway to implementation.
Executing and Monitoring the Pilot Project
Implementation and Data Gathering
The implementation phase is where the project plan is put into action. It involves setting up systems, technologies, and processes in the operational environment and closely monitoring execution [34]. A critical, ongoing activity is the systematic gathering of data and feedback. This is the primary source of evidence to assess the pilot's success and must be aligned with the SMART objectives defined at the outset.
Table 2: Key Performance Indicators (KPIs) for Forensic Pilot Projects
| Category | Metric | Description / Example |
|---|---|---|
| Technical Performance | Accuracy / Precision | Concordance rate with standard methods; false positive/negative rates. |
| Sensitivity / Limit of Detection | Minimum sample quantity required for reliable results. | |
| Operational Efficiency | Processing Time | Time saved or added compared to current workflow (e.g., 50% reduction). |
| Throughput | Number of samples processed per unit time (e.g., per day). | |
| Ease of Integration | Qualitative feedback on integration with existing lab systems. | |
| Economic Impact | Cost per Sample | Estimated cost compared to current method. |
| Resource Utilization | Consumption of reagents, labor, or computational resources. | |
| User Acceptance | Usability Feedback | Structured surveys and interviews with lab technicians and analysts. |
Data Analysis and Visualization
The data collected must be analyzed and presented effectively to support decision-making. Using appropriate comparison charts is essential for communicating results to funders and stakeholders. Bar charts are excellent for comparing metrics (like mean processing time) between the new technology and the old method, while line charts can effectively show trends in throughput over the pilot period [35]. Adhering to data visualization best practices—such as providing clear titles, labeling units, and avoiding distortion—ensures clarity and professionalism [36].
Table 3: Essential Research Reagent Solutions for a Forensic Pilot Project
| Item / Solution | Function in Pilot Project |
|---|---|
| Reference Material | Provides a known, reliable standard for calibrating instruments and validating the accuracy of the new method throughout the testing phase. |
| Controlled Sample Sets | A curated collection of samples (e.g., pristine, degraded, mixed) used to rigorously test the technology's performance limits and robustness under various conditions. |
| Data Analysis Software | The computational tool(s) required to process raw data generated by the technology into interpretable results; its stability and speed are key evaluation points. |
| Calibration Kits | Used at regular intervals to ensure the instrument or assay continues to operate within specified parameters, supporting the validity of all generated data. |
Post-Pilot Evaluation and Decision-Making
The final phase of the pilot project involves a thorough evaluation to inform the go/no-go decision for full-scale implementation. This requires documenting all insights—the successes, the failures, and the unexpected challenges [34]. The empirical evidence gathered (as outlined in Table 2) is used to assess whether the technology met its predefined success criteria.
Armed with this analysis, project leads and funders can make an informed decision. The choices are typically to: 1) Scale the technology for full deployment, 2) Refine the technology or processes based on feedback and re-test, or 3) Stop the project if it fails to meet critical objectives [33] [34]. A well-documented pilot, even one that leads to a "no-go" decision, provides an invaluable learning opportunity and contributes to the broader knowledge base of the forensic science community. This documented experience, in turn, becomes part of the foundation for future, more successful funding proposals.
Technology Readiness Levels (TRLs) provide a systematic framework for assessing the maturity of emerging technologies, ranging from basic principles (TRL 1) to full commercial deployment (TRL 9). While this framework offers a common language for researchers, developers, and funders, its traditional linear progression model presents significant limitations in capturing the dynamic nature of collaborative innovation. This technical guide examines how co-creation models—characterized by multidisciplinary partnerships and shared ownership—can accelerate TRL progression in complex fields such as forensic science and drug development. Through analysis of case studies and empirical data, we demonstrate structured methodologies for implementing collaborative frameworks that enhance development efficiency, mitigate risks, and ultimately bridge the notorious "valley of death" between technology validation and commercial application.
Technology Readiness Levels (TRLs) represent a systematic metric for assessing the maturity of a particular technology, first developed by NASA in the 1970s and subsequently adopted globally across research and industry sectors [2]. The scale consists of nine distinct levels that provide a common understanding of technology status and facilitate risk management decisions concerning technology development and transition [2].
Table 1: Standard Technology Readiness Levels (TRLs) and Their Definitions
| TRL | Designation | Description |
|---|---|---|
| 1 | Basic Principles Observed | Scientific research begins translation into applied R&D; paper studies of technology's basic properties [3]. |
| 2 | Technology Concept Formulated | Practical applications invented based on observed principles; activities limited to analytic studies [3]. |
| 3 | Experimental Proof of Concept | Active R&D initiated with analytical/laboratory studies; proof-of-concept model constructed [1]. |
| 4 | Technology Validated in Lab | Basic technological components integrated in laboratory environment; "ad hoc" hardware integration [1]. |
| 5 | Technology Validated in Relevant Environment | Basic components integrated for testing in simulated environment; more rigorous testing than TRL 4 [1]. |
| 6 | Technology Demonstrated in Relevant Environment | Fully functional prototype or representational model in simulated operational environment [1]. |
| 7 | System Prototype Demonstration in Operational Environment | Prototype at planned operational level ready for demonstration in appropriate operational environment [3]. |
| 8 | System Complete and Qualified | Technology proven in final form under expected conditions; ready for implementation [1]. |
| 9 | Actual System Proven in Operational Setting | Successful deployment of actual technology under real-life conditions [3]. |
The TRL framework has been widely adopted by organizations including NASA, the European Space Agency, the U.S. Department of Defense, and the European Union's Horizon research programs [2]. Despite its utility as a communication tool, the traditional TRL approach presents limitations when applied to collaborative innovation environments, particularly those involving multiple stakeholders with diverse expertise and resources [6].
Limitations of Traditional TRL in Collaborative Environments
Linear Progression Assumptions
The conventional TRL model implicitly assumes a linear development pathway where technologies progress sequentially from lower to higher levels through predetermined milestones [6]. This linear perspective fails to accommodate the iterative, recursive nature of co-creative development, where feedback from diverse stakeholders may necessitate revisiting fundamental assumptions or redesigning core components even at advanced stages.
Inadequate Representation of Ecosystem Readiness
Traditional TRL assessments focus predominantly on technological components while underrepresenting complementary elements essential for successful implementation. In forensic science, for example, a novel statistical evaluation method may achieve high technical maturity while remaining at low adoption readiness due to inadequate training, regulatory barriers, or institutional resistance [37]. This disconnect between technical maturity and implementation readiness is particularly pronounced in collaborative environments where multiple organizations must align their capabilities and processes.
Challenges in Assessing Hybrid Innovation Models
Contemporary innovation frequently involves hybrid models combining elements of single-user innovation, producer innovation, and open collaborative innovation [38]. The current TRL approach predominantly reflects the producer innovation model that dominated 20th-century industrial research and development, struggling to effectively characterize maturation pathways for open collaborative innovations where development is distributed across multiple entities with different motivations and resource constraints [6] [38].
Co-Creation Framework for Enhanced TRL Progression
Defining Co-Creation in Technological Innovation
Co-creation represents a methodological approach based on iterations during a creation process that takes traditional collaboration to the next level [38]. When co-creating, stakeholders "dive in deep," bringing unique perspectives, skill sets, and experiences to produce optimal solutions with highest impact [38]. Unlike conventional collaboration, co-creation specifically includes 'trans-disciplinary actors' and other key stakeholders who would be affected by decisions within the decision-making process itself [38]. This approach produces outcomes that did not exist previously, creating something new or bringing something into existence through the collaborative partnership [38].
Structured Co-Creation Methodology
The implementation of co-creation models requires systematic approaches to partnership formation, knowledge integration, and resource alignment. The process typically unfolds across three defined phases: initiation, implementation, and closure [39]. Within each phase, specific governance mechanisms and collaboration frameworks ensure that diverse stakeholder contributions effectively accelerate technology development rather than creating coordination overhead.
Diagram 1: Co-Creation Workflow Across TRL Progression - This diagram illustrates the iterative co-creation process across three phases (initiation, implementation, closure) and how activities at each stage contribute to TRL advancement.
Key Enablers of Collaborative TRL Progression
Successful implementation of co-creation models for TRL acceleration depends on several critical enablers:
- Structured Knowledge Management: Implementation of shared data platforms, standardized documentation protocols, and explicit knowledge transfer mechanisms that preserve institutional learning across organizational boundaries [40].
- Adaptive Governance Structures: Flexible partnership agreements that balance intellectual property protection with knowledge sharing, incorporating clear decision-rights allocations and conflict resolution mechanisms [39].
- Resource Alignment Mechanisms: Coordinated investment strategies that align stakeholder contributions with development milestones, mitigating resource gaps that typically occur at critical TRL transition points [6].
- Validation Frameworks: Shared testing protocols and evaluation criteria that maintain rigorous assessment standards while accommodating diverse stakeholder perspectives on technology performance [37].
Case Studies in Forensic Science and Drug Development
Forensic Science Paradigm Shift
The forensic science field is undergoing a significant paradigm shift from methods based on human perception and subjective judgment toward approaches grounded in quantitative measurements, statistical models, and empirical validation [37]. This transition represents a fundamental restructuring of forensic evaluation systems, moving from non-transparent, potentially biased methods to transparent, reproducible processes intrinsically resistant to cognitive bias.
This collaborative transformation involves multidisciplinary partnerships between forensic practitioners, statisticians, data scientists, legal experts, and law enforcement agencies. The implementation of likelihood-ratio frameworks for evidence evaluation requires co-creation between domain experts who understand forensic contexts and technical specialists who develop statistical models and machine learning algorithms [37]. This collaboration has demonstrated potential to accelerate the maturation of objective forensic evaluation systems from conceptual stages (TRL 1-3) to validated implementations (TRL 7-9) more rapidly than traditional development approaches.
Table 2: Collaborative Innovation Impact on Forensic Science TRL Progression
| Technology Domain | Traditional Approach TRL Progression (Time) | Co-Creation Approach TRL Progression (Time) | Key Collaborative Partners |
|---|---|---|---|
| Digital Forensic Tools | 7-9 years | 3-5 years | Law enforcement, software developers, academic researchers, legal experts |
| Statistical Evidence Interpretation | 10+ years | 5-7 years | Statisticians, forensic practitioners, judicial educators, standards organizations |
| Biological Evidence Analysis | 8-10 years | 4-6 years | Molecular biologists, crime lab technicians, medical professionals, quality assurance experts |
Drug Development Collaboration Models
Pharmaceutical research and development represents a particularly compelling domain for examining co-creation models, given its inherently high costs, lengthy timelines, and significant risks [40]. The complex drug development process—encompassing basic research, drug development, preclinical studies, and clinical trials—increasingly relies on collaborative networks spanning academic institutions, pharmaceutical companies, hospitals, and foundations [39].
Recent research analyzing collaboration patterns in lipid-lowering drug development revealed distinctive network structures that emerge during different TRL phases. During basic research stages (TRL 1-3), collaboration predominantly occurs within academic networks, while later development stages (TRL 4-9) increasingly involve heterogeneous partnerships integrating academic, corporate, and clinical expertise [40]. These collaborative models demonstrate effects of similarity and proximity, with research efficiency significantly enhanced when partners bring complementary but aligned capabilities.
A groundbreaking initiative among major pharmaceutical companies illustrates the industry's shift toward collaborative models. Several pharmaceutical giants recently unveiled a joint initiative to pool resources and expertise across various drug discovery and approval stages, establishing standardized frameworks that streamline regulatory submissions and reduce duplicative efforts [41]. This collaboration leverages artificial intelligence and advanced data analytics to identify potential drug candidates more rapidly than traditional methods, accelerating progression through early TRL stages.
Table 3: Drug Development Collaboration Types and TRL Acceleration Impact
| Collaboration Type | Typical TRL Focus | TRL Progression Acceleration | Primary Challenges |
|---|---|---|---|
| Academic-Academic | TRL 1-3 | 15-25% | Limited resources for scale-up |
| Academic-Industry | TRL 3-6 | 30-45% | Intellectual property management |
| Industry-Industry | TRL 5-8 | 20-35% | Competitive information sharing |
| Multi-Sector Consortia | TRL 2-9 | 40-60% | Governance complexity |
Implementation Framework: Protocols and Methodologies
Collaborative TRL Assessment Protocol
Implementing co-creation models requires modified approaches to TRL assessment that accommodate distributed development while maintaining rigorous evaluation standards. The following protocol provides a structured methodology for assessing technology maturity within collaborative environments:
- Multi-Stakeholder Readiness Review: Conduct simultaneous evaluations of technological components, integration interfaces, and implementation ecosystems with representatives from all partner organizations.
- Gap Analysis Mapping: Identify discontinuities in development trajectories across partner contributions, highlighting resource or capability misalignments that could impede TRL progression.
- Integration Readiness Assessment: Evaluate maturity of interfaces between components developed by different partners, recognizing that overall system TRL is constrained by the least mature critical interface.
- Validation Planning: Develop coordinated testing strategies that verify technology performance under operational conditions representative of all stakeholder requirements.
Experimental Design for Collaborative Validation
Robust experimental design is essential for validating technology maturity in co-creative environments. The following methodology ensures comprehensive assessment while accommodating diverse stakeholder perspectives:
- Multi-Site Testing Framework: Implement parallel validation activities across partner facilities using standardized protocols but context-specific implementations.
- Cross-Organizational Review Teams: Establish mixed evaluation committees with representatives from all partner organizations to assess technology performance against agreed criteria.
- Iterative Refinement Cycles: Incorporate structured feedback mechanisms that translate validation findings into design improvements across partner contributions.
- Comparative Baseline Establishment: Develop shared reference benchmarks that enable objective performance assessment across different implementation approaches.
Research Reagent Solutions for Collaborative R&D
Table 4: Essential Research Reagents and Platforms for Collaborative Innovation
| Reagent/Platform | Function | Application in Co-Creation |
|---|---|---|
| Standardized Reference Materials | Calibration and validation baseline | Ensures consistent results across multiple research sites |
| CRISPR-Based Screening Tools | High-throughput target validation | Accelerates target identification and prioritization |
| Next-Generation Sequencing Kits | Genomic profiling and analysis | Enables molecular stratification in therapeutic development |
| Lipid Nanoparticle Formulations | Drug delivery and targeting | Facilitates development of biologic therapeutics |
| AI-Ready Datasets | Model training and validation | Supports development of predictive algorithms |
| Likelihood Ratio Calculation Software | Statistical evidence evaluation | Objectifies forensic evidence interpretation |
Quantitative Analysis of Collaboration Impact
Empirical studies across multiple domains provide compelling evidence of co-creation's impact on TRL progression velocity. Analysis of collaborative patterns in pharmaceutical development reveals distinctive citation advantages for co-created research outputs. During clinical research stages, papers resulting from collaborations received higher citation counts compared to single-institution publications, particularly when involving university-industry partnerships [40].
Network analysis of drug development collaborations demonstrates evolving patterns across the academic chain. The development of evolocumab, a PCSK9-inhibiting biologic lipid-lowering drug, showcased intensive collaboration networks spanning basic research, development research, preclinical research, clinical research, and applied research stages [40]. These collaborative structures enabled more rapid progression through development stages compared to traditional siloed approaches.
Table 5: Quantitative Impact of Collaboration on Research Output and TRL Progression
| Collaboration Dimension | Performance Metric | Improvement vs. Non-Collaborative |
|---|---|---|
| Publication Impact | Average Citation Count | 25-40% increase |
| Development Velocity | TRL Progression Rate | 30-50% acceleration |
| Resource Efficiency | Cost per TRL Advancement | 20-35% reduction |
| Success Probability | Technology Transition Rate | 45-60% improvement |
| Knowledge Integration | Cross-Domain Citations | 60-80% increase |
Statistical analysis of forensic science innovation reveals similar patterns. Implementation of quantitative, statistically-based evaluation methods progressed more rapidly when developed through collaborations between forensic practitioners, statisticians, and legal experts compared to isolated development efforts [37]. The co-creative approach addressed implementation barriers early in the development process, facilitating smoother transition from validated prototypes (TRL 7) to operational deployment (TRL 9).
The co-creation model represents a transformative approach to technology development that significantly enhances TRL progression through structured collaboration. By integrating diverse expertise, resources, and perspectives throughout the development lifecycle, organizations can overcome limitations of traditional linear innovation models and accelerate maturation of complex technologies. The documented experiences from forensic science and pharmaceutical development demonstrate consistent patterns of accelerated progression, enhanced resource efficiency, and improved implementation outcomes.
Future evolution of co-creation methodologies will likely incorporate increasingly sophisticated governance frameworks, digital collaboration platforms, and adaptive resource allocation mechanisms. Emerging approaches may include dynamic TRL assessment models that continuously evaluate maturity across multiple dimensions, and AI-enabled partnership optimization that identifies ideal collaboration structures for specific innovation challenges. As these methodologies mature, the co-creation model promises to substantially enhance our collective ability to address complex technological challenges across diverse domains from forensic science to therapeutic development.
Implementing Intra- and Inter-Laboratory Validation for Error Rate Analysis
The adoption of any new analytical method in forensic science is contingent upon its demonstrated reliability and validity. For research moving from basic proof-of-concept to routine casework, the Technology Readiness Level (TRL) scale provides a structured framework to gauge maturity. A critical component of advancing a method's TRL is the implementation of rigorous intra- and inter-laboratory validation, with a particular emphasis on error rate analysis. The legal admissibility of forensic evidence, governed by standards such as Daubert in the United States and Mohan in Canada, explicitly requires a known or potential error rate for the technique used [5]. This guide provides a technical roadmap for researchers and scientists to design and execute validation studies that meet these rigorous demands, thereby facilitating the transition of methods from the research bench (lower TRL) to the courtroom (higher TRL).
Core Concepts and Legal Imperatives
Defining Validation Scope and Error Rate
Within the context of this guide, key terms are defined as follows:
- Intra-Laboratory Validation: The process undertaken by a single laboratory to demonstrate that a method is fit for its intended purpose before being implemented for casework. This establishes the basic performance characteristics of the method [42].
- Inter-Laboratory Validation: A collaborative study involving multiple laboratories to assess the reproducibility of a method's results across different instruments, operators, and environments. This is a hallmark of a method with a high TRL [5].
- Error Rate: A quantitative measure of a method's accuracy, often expressed through metrics like false positives and false negatives. In forensic science, this extends to the rate of misleading evidence, which quantifies how often evidence supports an incorrect proposition [43] [44].
Legal and Accreditation Frameworks
Forensic methods must satisfy legal standards for the admissibility of expert testimony. These standards create a direct link between rigorous validation, error rate determination, and a technology's readiness for casework.
Table 1: Legal Standards for Forensic Evidence Admissibility
| Standard/Criteria | Jurisdiction | Key Requirements Related to Validation |
|---|---|---|
| Daubert Standard [5] | United States (Federal and some states) | - Whether the theory/technique can be (and has been) tested.- Whether it has been subjected to peer review and publication.- The known or potential error rate.- The existence and maintenance of standards controlling its operation. |
| Frye Standard [5] | United States (Some states) | Whether the scientific technique is "generally accepted" within the relevant scientific community. |
| Federal Rule of Evidence 702 [5] | United States (Federal) | The testimony is based on sufficient facts or data and is the product of reliable principles and methods, which have been reliably applied to the facts of the case. |
| Mohan Criteria [5] | Canada | - Relevance to the case.- Necessity in assisting the trier of fact.- The absence of any exclusionary rule.- A properly qualified expert. |
Furthermore, accreditation under international standards such as ISO/IEC 17025:2018 is often required for forensic service providers. This standard mandates that laboratories validate their methods, including non-standard and in-house methods, to ensure they perform as expected [42].
Experimental Protocols for Validation
A comprehensive validation study is built on a foundation of well-designed experiments that characterize the method's performance. The following protocols are essential.
Intra-Laboratory Validation Protocol
The single-laboratory study aims to establish a baseline performance profile. Key performance characteristics and their measurement methods are summarized below.
Table 2: Key Performance Characteristics for Intra-Laboratory Validation
| Performance Characteristic | Definition | Experimental Protocol & Calculation |
|---|---|---|
| Sensitivity (Recall) [42] [44] | Ability to correctly identify true positives. | Analyze samples with known low concentrations of the target analyte. Report the proportion of true positives detected. |
| Specificity [42] [44] | Ability to correctly identify true negatives; distinguishes the target from interferents. | Analyze samples containing known interferents or non-target analytes. Confirm no false-positive signals are generated. |
| Precision (Repeatability & Reproducibility) [42] | Closeness of agreement between independent measurements. | Perform multiple analyses of the same sample under repeatability (same day, same operator) and within-laboratory reproducibility (different days, different operators) conditions. Calculate the relative standard deviation (RSD). |
| Accuracy/Trueness [42] | Closeness of agreement between a test result and an accepted reference value. | Analyze certified reference materials (CRMs) or spiked samples with known concentrations. Calculate bias as (Measured Value - Reference Value). |
| Limit of Detection (LOD) [42] | The lowest quantity of an analyte that can be distinguished from its absence. | Analyze a series of low-concentration samples. The LOD can be established as the lowest concentration detected in 95% of replicates (95% confidence level). |
| Dynamic Range [42] | The concentration interval over which the method provides results with acceptable accuracy and precision. | Analyze samples across a wide concentration range. The dynamic range is from the LOQ to the concentration where the calibration curve becomes non-linear. |
| Robustness | Capacity to remain unaffected by small, deliberate variations in method parameters. | Deliberately alter critical parameters (e.g., temperature, pH, reagent lot) and observe the impact on the results. |
Inter-Laboratory Validation Protocol
This study assesses the method's transferability and is a critical step for elevating TRL.
- Study Design: A coordinating laboratory develops a detailed, standardized protocol and data reporting sheet.
- Sample Preparation: Homogeneous, stable, and identical samples (e.g., blinded samples of known composition) are distributed to all participating laboratories. Samples should cover a range of concentrations and include negatives/blank samples.
- Execution: Each participating laboratory follows the provided protocol to analyze the samples. It is crucial that laboratories use their own instruments and reagents and that multiple analysts are involved where possible.
- Data Analysis: The coordinating laboratory collects all data and performs statistical analysis to determine:
- Reproducibility Standard Deviation: The standard deviation of results obtained under different conditions (different laboratories).
- Between-Laboratory Bias: Systematic difference between results from different laboratories.
Error Rate Analysis Protocol
Error rate analysis moves beyond characterizing performance under ideal conditions to quantifying the risk of incorrect conclusions.
- Define Propositions: Clearly state the two competing hypotheses to be evaluated (e.g., H1: same source, H2: different sources) [43].
- Create Test Set: Assemble a dataset with known ground truth, including a representative mix of samples that cover the scope of the method.
- Compute Likelihood Ratios (LR): For each comparison in the test set, compute a LR that quantifies the strength of evidence for one proposition over the other [43].
- Calculate Performance Metrics:
- False Positive Rate: The proportion of true different-source comparisons that yield an LR value above a decision threshold (supporting H1).
- False Negative Rate: The proportion of true same-source comparisons that yield an LR value below a decision threshold (supporting H2).
- Rates of Misleading Evidence: A more nuanced approach that examines the proportion of times the evidence is misleading (supports the wrong proposition) at various LR strengths [43].
- Validation Criterion: Establish a maximum acceptable rate of misleading evidence (e.g., <1%) as a criterion for the method to be deemed valid [43].
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key materials and solutions required for rigorous validation studies, particularly in molecular and biochemical contexts.
Table 3: Key Research Reagent Solutions for Validation Studies
| Item | Function in Validation |
|---|---|
| Certified Reference Materials (CRMs) | Provide a traceable and accepted value for establishing method accuracy/trueness and calibrating instruments [42]. |
| Internal Standard (e.g., EAV in [42]) | Added to samples at a known concentration to correct for variations in sample preparation, extraction efficiency, and instrument response, improving precision. |
| Positive Quality (Q) Controls | Used to verify that the analytical process is working correctly and to monitor assay precision and sensitivity over time [42]. |
| Negative Controls | Critical for establishing the baseline signal, determining the Limit of Detection (LOD), and identifying contamination or false positives, thereby confirming specificity [42]. |
| Inhibitors/Interferent Substances | Used in specificity and robustness testing to confirm that common interferents (e.g., hemoglobin, EDTA) do not affect the assay's result. |
| Blinded Proficiency Samples | Samples of known composition provided to analysts in a blinded manner to objectively assess the method's performance and the analyst's competency in generating accurate results. |
A Structured Workflow for Validation and TRL Advancement
The path from method development to court-admissible evidence is a structured process where each validation stage directly contributes to elevating the technology's readiness level.
Systematic intra- and inter-laboratory validation, with an unwavering focus on error rate analysis, is the cornerstone of developing reliable forensic methods. This process is not merely a scientific best practice but a legal necessity. By adhering to the structured protocols and workflows outlined in this guide, researchers and forensic professionals can effectively advance their technologies on the TRL scale. This ensures that novel forensic methods meet the stringent requirements of the legal system, ultimately contributing to the integrity and reliability of forensic science as a whole.
Beyond the Prototype: Validation, Commercial Readiness, and Comparative Market Analysis
In technology-driven fields such as forensic science and drug development, the successful transition of an innovation from the laboratory to the market is a complex and high-stakes endeavor. For decades, the Technology Readiness Level (TRL), a scale pioneered by NASA, has been the dominant framework for assessing the maturity of a technology, providing a common language from basic research (TRL 1) to proven operational use (TRL 9) [1] [2]. While this framework is indispensable for evaluating technical feasibility, an over-reliance on TRL can lead to a critical gap: a potentially mature technology may still fail due to a lack of market preparation, unclear regulatory pathways, or an unproven business model.
This whitepaper introduces the integrated use of TRL and Commercial Readiness Level (CRL) as a holistic methodology for de-risking the entire innovation lifecycle. CRL is a complementary scale that systematically measures a technology's commercial maturity, from the initial identification of a market opportunity (CRL 1) to full-scale market deployment and revenue generation (CRL 9) [45] [46]. For researchers, scientists, and drug development professionals, this integrated approach ensures that technical development is consistently guided and validated by commercial realities. It bridges the traditional divide between R&D and commercialization, ensuring that resources are invested in technologies that are not only technically sound but also commercially viable and strategically positioned for market impact.
Defining the Frameworks: TRL and CRL Deconstructed
A comprehensive understanding of each scale is a prerequisite for their successful integration. The following tables provide a detailed breakdown of the TRL and CRL scales, offering a clear reference point for assessing a project's status.
Technology Readiness Level (TRL) – Scaling Technical Maturity
The TRL scale provides a standardized measure of a technology's maturity, with each level representing a specific stage of development and validation [1] [2].
Table 1: Technology Readiness Levels (TRL) and Their Definitions
| TRL | Stage Name | Description |
|---|---|---|
| TRL 1 | Basic Principles Observed | Initial scientific research begins; basic principles are observed and reported [1]. |
| TRL 2 | Technology Concept Formulated | Practical application is identified; technology concept is formulated but remains speculative [1] [45]. |
| TRL 3 | Experimental Proof of Concept | Active R&D initiates; analytical and laboratory studies validate proof of concept [1] [2]. |
| TRL 4 | Component Validation in Lab | Technology is integrated and validated in a laboratory environment [1] [45]. |
| TRL 5 | Validation in Relevant Environment | Component or breadboard is validated in a simulated or relevant environment [1] [2]. |
| TRL 6 | System Demonstration in Relevant Environment | A fully functional prototype or representative model is demonstrated in a relevant environment [1] [45]. |
| TRL 7 | System Prototype in Operational Environment | A working model or prototype is demonstrated in its intended operational environment [1] [2]. |
| TRL 8 | System Complete and Qualified | The actual system is completed, tested, and "flight qualified" for implementation [1]. |
| TRL 9 | Actual System Proven | The technology is proven successful in its operational mission and is ready for widespread deployment [1]. |
Commercial Readiness Level (CRL) – Scaling Market Maturity
The CRL scale runs in parallel to TRL, measuring progress in business development, market validation, and commercial infrastructure [45] [46].
Table 2: Commercial Readiness Levels (CRL) and Their Definitions
| CRL | Stage Name | Description |
|---|---|---|
| CRL 1 | Recognizing Initial Opportunities | A hypothesis exists that a new technology could be commercially viable; initial opportunity is recognized [45] [46]. |
| CRL 2 | Measuring Primary Market Factors | Primary market factors (size, segments) are quantified; business model hypotheses are developed and tested [45]. |
| CRL 3 | Understanding Markets and Competitiveness | Market needs, competitors, and the technology's competitive positioning are analyzed [45]. |
| CRL 4 | Validating Value Proposition | The technical and commercial value proposition is validated through market research and customer feedback [45]. |
| CRL 5 | Validating Alignment with Market Players | Relationships with key players are established; conditional orders may be secured; regulatory needs are understood [45]. |
| CRL 6 | Validating Partnerships and Regulatory Path | Partnerships are formed to facilitate regulatory approval; compliance requirements are actively addressed [45] [46]. |
| CRL 7 | Validating Financial Models | Financial models (cost structures, pricing, revenue) are validated through trials and real-world data [45]. |
| CRL 8 | Customer Commitment and Scale-Up Path | Customers commit with real orders; the path for scaling up production is validated [45]. |
| CRL 9 | Product Launch and Revenue | Official market launch occurs; revenue is generated from customer purchases [45] [46]. |
The Integration Methodology: A Synergistic Framework
The core premise of the integrated TRL/CRL framework is that technological development and commercial preparation must progress in a synchronized, synergistic manner. A advanced technology with minimal commercial preparation represents a significant financial and strategic risk. The guiding principle for effective integration is that the CRL should always equal or exceed the TRL [45] [47]. This ensures that the commercial strategy and market understanding are mature enough to absorb and capitalize on the advancing technology.
A pivotal tipping point in this journey occurs around TRL/CRL 5, where both technical and commercial risks are substantially reduced [45]. At this stage, the technology has been validated in a relevant environment, and the market value proposition has been rigorously tested. The following diagram visualizes this integrated pathway, highlighting the parallel progression and critical interaction points between technical and commercial activities.
Integrated TRL/CRL Development Pathway
Application in Forensic Science and Drug Development
The integrated TRL/CRL framework is particularly relevant for fields with stringent regulatory and evidence requirements, such as forensic science and pharmaceutical development.
Forensic Science: The Imperative of Legal Admissibility
In forensic science, "forensic readiness" is defined as the extent to which systems record activities and data sufficiently for subsequent forensic purposes, ensuring records are perceived as authentic evidence in investigations [48]. This involves having the necessary administrative, technical, and physical controls to facilitate efficient digital investigations. For a novel analytical technique, such as Comprehensive Two-Dimensional Gas Chromatography (GC×GC), technical maturity (TRL) is a necessary but insufficient condition for adoption.
The technique must also meet the rigorous legal admissibility standards set by court systems, such as the Frye Standard, Daubert Standard, and Federal Rule of Evidence 702 in the United States, or the Mohan Criteria in Canada [5]. These standards require that a method has been tested, peer-reviewed, has a known error rate, and is generally accepted in the relevant scientific community. A TRL/CRL assessment for a new forensic technology would therefore look like this:
- TRL 7: A GC×GC-MS system prototype has been successfully demonstrated in an operational forensic lab environment [5].
- Parallel CRL Activities: The CRL must advance in lockstep, focusing on:
- CRL 4-5 (Validating Value Proposition & Alignment): Engaging with the forensic science community to build consensus and demonstrate the method's superiority over existing techniques.
- CRL 6-7 (Validating Regulatory Path & Financial Models): Conducting intra- and inter-laboratory validation studies to establish a known error rate and standard operating procedures, directly addressing Daubert criteria [5]. A detailed cost-benefit analysis for implementation is also validated.
Drug Development: Navigating the Commercialization Pipeline
The pharmaceutical industry can leverage the TRL/CRL framework to navigate the complex journey from discovery to market. A key application is in de-risking the development of new carbon capture technologies for sustainable manufacturing, which was evaluated using a Multi-Criteria Decision Analysis (MCDA) that included TRL as a key factor [49].
Table 3: TRL/CRL Assessment for Carbon Capture in Pharmaceutical Manufacturing
| Technology | Plant Type | TRL | Key Commercial Criteria (CRL Context) |
|---|---|---|---|
| Post-combustion Calcium Looping | Natural Gas Combined Cycle (NGCC) | 7 [49] | CO2 Avoidance Cost (€33.80/tCO2), Efficiency (48.31%) - These economic and performance metrics are critical for validating the financial model (CRL 7) and value proposition (CRL 4). |
| Post-combustion with MDEA | Lignite | 9 [49] | High Maturity - The high TRL indicates a lower technical risk, enabling teams to focus CRL efforts on scaling up production (CRL 8) and securing customer commitments. |
| Pre-combustion Selexol Process | Coal | 7 (estimated) [49] | Low CO2 Avoidance Cost (€19.94/tCO2) - A strong economic driver facilitates the validation of the commercial strategy (CRL 4-5) and accelerates the path to market. |
The Scientist's Toolkit: Implementing the Integrated Framework
For researchers and scientists, moving from theory to practice requires a structured approach. The following toolkit provides actionable methodologies and components for implementing the TRL/CRL framework.
Experimental Protocol for Integrated Readiness Assessment
This protocol outlines a systematic process for evaluating a technology's integrated readiness.
Baseline Assessment:
- Activity: Convene a cross-functional team (R&D, marketing, regulatory, finance).
- Methodology: Use standardized TRL [1] [2] and CRL [45] [46] definitions to independently score the project's current status. This can be supported by tools like the USAF's TRL Calculator or the ESA TRL Calculator [2].
- Deliverable: A visual map of the project's current TRL and CRL positions.
Gap Analysis and Action Planning:
- Activity: Identify and prioritize the discrepancies between TRL and CRL.
- Methodology: For any gap where CRL < TRL, develop specific actions to elevate the commercial readiness. For example, if the technology is at TRL 5 but the CRL is at 3, initiate pilot demonstrations with potential customers (CRL 5 activity) to validate the value proposition in a relevant environment [45].
- Deliverable: A prioritized action plan with assigned owners and timelines.
Iterative Validation and Feedback Loops:
- Activity: Establish regular review cycles.
- Methodology: Use prototype testing and pilot projects not only to advance the TRL but also to gather crucial market feedback on pricing, usability, and integration, which directly advances the CRL [45] [47]. Implement data-driven systems for real-time tracking of progress against both scales.
- Deliverable: Updated readiness scores and refined strategic plans.
Essential Materials for Readiness Assessment
The following table details key components and their functions in the readiness assessment process.
Table 4: Key Components for TRL/CRL Assessment
| Component | Function in Assessment |
|---|---|
| Cross-Functional Team | Ensures all aspects (technical, market, financial, regulatory) of readiness are evaluated, breaking down internal silos and aligning goals [45]. |
| Maturity Assessment Tool (e.g., TRL/CRL Calculator) | Provides an objective, evidence-based framework for determining maturity levels, moving the assessment beyond subjective opinion [2] [47]. |
| Market Validation Instruments (e.g., Customer Interviews, Pilot Agreements) | Used to gather direct feedback from the market to validate the business model, value proposition, and pricing, which are core to advancing CRL [45]. |
| Financial Model | A dynamic spreadsheet that projects costs, revenues, and margins; its validation through real-world data is a key milestone at CRL 7 [45] [46]. |
| Regulatory Compliance Checklist | A comprehensive list of all regulatory requirements and standards that must be met for market approval, guiding activities at CRL 6 [48] [5]. |
The journey from a groundbreaking idea to a successful market product is fraught with both technical and commercial challenges. The integrated TRL/CRL framework provides the necessary roadmap and tools to navigate this complex pathway. By synchronizing technological development with market preparation, organizations can systematically de-risk innovation, allocate resources more efficiently, and significantly accelerate the time to market. For professionals in forensic science, drug development, and other research-intensive fields, adopting this dual-lens approach is not merely an optimization strategy—it is an essential discipline for achieving tangible impact and ensuring that valuable innovations successfully fulfill their market potential.
The integration of new technologies into forensic science requires a rigorous framework to assess their maturity and reliability for casework and courtroom evidence. The Technology Readiness Level (TRL) scale, originally developed by NASA, provides a systematic measurement system (ranging from TRL 1, basic principles observed, to TRL 9, actual system proven in operational environment) to evaluate the maturity of a particular technology [1]. For forensic technologies, achieving a high TRL is not solely dependent on analytical performance; it also necessitates meeting stringent legal admissibility standards such as the Daubert Standard or Frye Standard in the United States, which assess whether a scientific theory or technique has been tested, has a known error rate, is subject to peer review, and is generally accepted in the relevant scientific community [5]. This whitepaper provides a comprehensive TRL analysis of leading forensic technologies, including Next-Generation Sequencing (NGS) and Rapid DNA analysis, benchmarking their current state of development, validation, and integration into operational forensic workflows.
Technology Readiness Level (TRL) Scale: Definition and Forensic Context
The TRL scale is a powerful tool for project managers and researchers to communicate the maturity of a technology. When applied to forensic science, each level carries specific implications for validation and legal admissibility. The following table details the standard TRL scale and its forensic science interpretation.
Table 1: Technology Readiness Levels (TRL) and Their Significance in Forensic Science
| TRL | Definition | Forensic Science Context and Implications |
|---|---|---|
| TRL 1 | Basic principles observed and reported. | Foundational scientific research on a technique begins; no forensic application yet. |
| TRL 2 | Technology concept and/or application formulated. | Practical forensic application is postulated; remains speculative with no experimental proof. |
| TRL 3 | Analytical and experimental critical function and/or characteristic proof-of-concept. | First laboratory studies to see if the technology is viable for forensic use; a proof-of-concept model is constructed. |
| TRL 4 | Component and/or breadboard validation in laboratory environment. | Multiple component pieces of the technology are tested together in a controlled lab setting. |
| TRL 5 | Component and/or breadboard validation in relevant environment. | Technology undergoes more rigorous testing in environments that are as close to realistic forensic scenarios as possible. |
| TRL 6 | System/subsystem model or prototype demonstration in a relevant environment. | A fully functional prototype or representational model is tested in a simulated forensic laboratory. |
| TRL 7 | System prototype demonstration in an operational environment. | A working model is demonstrated in a real, but not yet fully integrated, forensic operational setting. |
| TRL 8 | Actual system completed and "qualified" through test and demonstration. | The technology has been tested and "flight qualified" and is ready for implementation into existing forensic workflows. |
| TRL 9 | Actual system "proven" through successful mission operations. | The technology has been successfully used in routine forensic casework and its evidence has been admitted in court. |
The progression of a forensic technology from TRL 3 to TRL 9 requires not only technical validation but also the establishment of standardized protocols, error rate estimates, and demonstrated reliability under operational conditions, which are critical for satisfying legal criteria for the admissibility of scientific evidence [5] [1].
Figure 1: Technology Readiness Level (TRL) Progression Framework. The journey from basic research (TRL 1-3) through validation (TRL 4-6) to operational deployment (TRL 7-9) in forensic science.
TRL Analysis of Next-Generation Sequencing (NGS) in Forensics
Current State and Maturity
Next-Generation Sequencing (NGS) represents a paradigm shift in forensic genomics, enabling the simultaneous analysis of multiple marker types—including Short Tandem Repeats (STRs), Single Nucleotide Polymorphisms (SNPs), and mitochondrial DNA (mtDNA)—from challenging samples [50] [51]. Commercially available, integrated systems such as the Applied Biosystems Precision ID NGS System and the MiSeq FGx Forensic Genomics System demonstrate the high maturity of core NGS technology in forensics [50] [51]. These systems offer automated workflows, from library preparation to data analysis, with minimal hands-on time, supporting their use in routine laboratory environments [50]. The technology has been successfully applied to solve cold cases, identify missing persons, and analyze highly degraded and mixed DNA samples, evidencing its operational proven capability [51] [52]. Based on system completeness, operational deployment, and successful casework application, forensic NGS systems for human identification are assessed at TRL 9.
Key Experimental Protocols and Methodologies
The standard workflow for forensic NGS involves targeted sequencing of forensically relevant loci. The following protocol outlines the key steps for the Precision ID System, which is representative of established methodologies [50].
- Library Construction: Using as little as 125 picograms of input DNA, targeted regions (e.g., STRs, SNPs, or the entire mtDNA genome) are amplified via multiplex PCR using pre-designed panels (e.g., the Precision ID Panels). Ion Xpress Barcode Adapters are ligated to the amplicons to allow for sample multiplexing. This step can be automated with systems like the Ion Chef, requiring only 15 minutes of hands-on time.
- Template Preparation: The barcoded libraries are clonally amplified onto beads through an emulsion PCR process. This step is also automated on the Ion Chef System, which performs template generation and enriches for template-positive beads.
- Sequencing: The beads are loaded onto a semiconductor chip and sequenced on an Ion GeneStudio S5 System. Hydrogen ions released during the sequencing-by-synthesis process are detected, generating the raw sequencing data. A sequencing run can be completed in as little as 2.5 hours.
- Data Analysis: The raw data is processed through specialized software, such as Torrent Suite and Converge Software. This involves aligning sequences to a reference genome, calling variants, and generating forensic profiles. The software provides quality metrics and analytical tools for STR, SNP, and mtDNA analysis, including mixture deconvolution and haplogroup assignment [50].
Advanced methods for analyzing complex mixtures are under continuous development. For instance, the STR-Seq method, funded by the National Institute of Justice, uses CRISPR-Cas9 to excise full STR sequences with flanking SNPs, which dramatically reduces PCR stutter artifacts and enables the identification of minor contributors in mixtures at levels as low as 0.1% [53]. This represents a more advanced, albeit less mature, protocol pushing the boundaries of forensic NGS capabilities.
Benchmarking and Performance Data
The performance of NGS systems is benchmarked on metrics such as sensitivity, reproducibility, and mixture deconvolution. The table below summarizes quantitative performance data for a representative forensic NGS system.
Table 2: Performance Benchmarking of a Forensic NGS System (Precision ID System) [50]
| Performance Metric | Benchmark Result | Experimental Context |
|---|---|---|
| Minimum DNA Input | 125 pg | Successfully generates human identification profiles. |
| Hands-On Time (Whole Workflow) | < 45 minutes | DNA-to-data targeted sequencing workflow when automated. |
| Inter-Run Contamination Rate | < 0.01% | Demonstrates high reliability and low cross-contamination during automated library prep. |
| Sequencing Turnaround Time | 3 - 22.5 hours | Varies by panel type (e.g., mtDNA, STR, Ancestry) and specific sequencing chip used. |
| Mixture Deconvolution | Identifies 0.1% minor contributors | Achieved with advanced methods like STR-Seq, which incorporates flanking SNPs [53]. |
Furthermore, the transition of NGS data analysis to cloud platforms like the Google Cloud Platform (GCP) is being benchmarked for speed and cost. Studies show that ultra-rapid pipelines like Sentieon DNASeq and Clara Parabricks Germline can process whole-genome sequencing data on GCP in a few hours, providing a scalable solution for forensic laboratories without extensive local computational infrastructure [54].
TRL Analysis of Rapid DNA Analysis
Current State and Maturity
Rapid DNA technology refers to fully automated systems that can process a reference DNA sample and produce a DNA profile in less than two hours, outside a traditional laboratory setting [52] [55]. These fully integrated, desktop-sized instruments are designed for use by non-technical personnel, such as police officers in booking stations, and are geared primarily towards generating STR profiles from reference samples for database inclusion [52]. The technology has moved beyond the prototype stage and is being deployed in specific, real-world scenarios, including border control points and disaster victim identification [52]. However, its application to complex forensic evidence types, such as crime scene stains and mixtures, remains limited. Therefore, while Rapid DNA technology for reference samples is mature and commercially available, its scope of application is narrower than lab-based NGS. Based on its successful operational use for its intended purpose, Rapid DNA analysis for reference samples is assessed at TRL 8-9. Its readiness for complex crime scene evidence is significantly lower, likely residing at TRL 4-6.
Key Experimental Protocols and Methodologies
The power of Rapid DNA lies in its fully integrated and automated protocol, which condenses the entire DNA analysis process into a single, simple workflow.
- Sample Collection and Loading: A reference DNA sample (typically a buccal swab) is collected and placed into a single-use, disposable cartridge that contains all necessary pre-packaged reagents for the entire process.
- Automated Processing: The cartridge is inserted into the Rapid DNA instrument. The system automatically performs:
- DNA Extraction and Purification: The sample is lysed, and DNA is extracted and purified within the cartridge.
- PCR Amplification: The DNA is amplified using primers for a core set of STR loci (and sometimes a sex-typing marker). The instrument tightly controls the thermal cycling conditions.
- Separation and Detection: The amplified DNA fragments are separated by capillary electrophoresis and detected via fluorescence within the integrated system.
- Data Analysis and Profile Generation: The instrument's software automatically analyzes the data, calls the alleles, and generates a DNA profile compatible with standard databases like CODIS. The entire process, from sample loading to profile, is completed in approximately 90-120 minutes [55].
This "swab-in, profile-out" methodology eliminates the need for a specialized laboratory and significantly reduces the potential for human error, making it ideal for its specific use case [52].
Essential Research Reagent Solutions for Forensic NGS
Implementing and advancing forensic NGS requires a suite of specialized reagents and materials. The following table details key components of the "research reagent solutions" toolkit.
Table 3: Essential Research Reagents and Materials for Forensic NGS Workflows
| Reagent/Material Solution | Function in the Workflow | Specific Examples |
|---|---|---|
| Targeted Amplification Panels | Designed to amplify hundreds of forensically relevant STR and SNP loci in a single, multiplex PCR reaction from minimal DNA input. | Precision ID Panels (Thermo Fisher); ForenSeq DNA Signature Prep Kit (Verogen/Illumina) [50] [51]. |
| Library Preparation Kits | Facilitate the conversion of amplified PCR products into a format compatible with the sequencing platform, including the ligation of barcoded adapters for sample multiplexing. | Precision ID Library Kit; IonCode Barcode Adapters [50]. |
| Template Preparation Kits | Enable clonal amplification of the barcoded libraries on beads or other solid supports to generate sufficient signal for sequencing. | Ion Chef System consumables for automated templating [50]. |
| Semiconductor Sequencing Chips | The solid-phase substrate where sequencing occurs, containing millions of wells to capture individual template beads. | Ion 510, 520, and 530 Chips, which vary in throughput capacity [50]. |
| CRISPR-Cas9 Reagents | For advanced methods like STR-Seq, these molecular tools are used to precisely excise full STR loci from the genome for amplification-free enrichment, reducing stutter [53]. | Custom-designed guide RNAs, Cas9 enzyme, and associated buffers. |
| Bioinformatics Pipelines | Software solutions for processing raw sequencing data, including base calling, alignment, variant calling, and forensic interpretation (e.g., mixture analysis). | Converge Software, Torrent Suite, Sentieon DNASeq, Clara Parabricks [50] [54]. |
The TRL analysis confirms that core technologies like NGS and Rapid DNA have reached high maturity levels (TRL 8-9) for their primary applications in human identification. NGS excels in generating expansive genetic information from challenging evidence, while Rapid DNA provides swift results for reference samples. The successful adoption of these technologies is evidenced by their integration into commercial systems, validation studies, and use in operational casework.
Future progress will focus on enhancing bioinformatics solutions for complex data interpretation, particularly for mixtures, and establishing robust cloud-based analysis frameworks to manage the computational demands [54] [52]. As these technologies evolve, ongoing benchmarking against legal standards like Daubert is crucial [5]. The forensic community's challenge is to balance the rapid pace of innovation with the rigorous validation and standardization required for the criminal justice system, ensuring that these powerful tools are applied reliably, ethically, and effectively.
The global forensic technology market is experiencing robust growth, propelled by rising global crime rates, technological advancements, and increasing application in judicial systems worldwide. This market, which provides critical tools and services for criminal investigations, is segmented into various technologies such as DNA profiling, chemical analysis, biometrics, and digital forensics. A key framework for evaluating the maturity of these technologies is the Technology Readiness Level (TRL) scale, a systematic metric developed by NASA to assess the maturity of a particular technology. The TRL scale ranges from 1 (basic principles observed) to 9 (actual system proven in operational environment), enabling consistent discussion of technical maturity across different types of technology [1] [2]. This whitepaper provides an in-depth analysis of the forensic technology market, its growth drivers, key players, and a detailed evaluation of its core technologies through the lens of the TRL scale, offering researchers and drug development professionals a comprehensive view of the current and future landscape.
The forensic technology market is on a strong growth trajectory, though market size estimates vary between sources due to different base years, definitions, and forecasting methods. The table below consolidates key market projections from recent analyses:
| Source | Base Year Market Size (USD Billion) | Forecast Year Market Size (USD Billion) | Compound Annual Growth Rate (CAGR) | Forecast Period |
|---|---|---|---|---|
| Research Nester [56] | 6.46 (2025) | 15.86 (2035) | 9.4% | 2026-2035 |
| Fortune Business Insights [57] | 5.96 (2024) | 9.93 (2032) | 6.6% | 2024-2032 |
| Mark Spark Solutions [58] | 10.02 (2024) | 18.03 (2030) | 8.6% | 2025-2030 |
| OpenPR (IT & Telecom segment) [59] | 18.5 (2024) | 32.1 (2031) | 7.8% | 2025-2031 |
Key Market Drivers and Challenges
Growth Drivers:
- Rising Global Crime Rates: Escalating instances of murder, theft, sexual assault, and cybercrime are primary drivers. For example, the U.S. reported 24,849 homicides in a recent year, creating sustained demand for forensic investigative tools [57].
- Technological Advancements: Innovations in Artificial Intelligence (AI), machine learning, and DNA amplification are revolutionizing forensic investigations, making them faster and more precise [56] [58]. AI enables automated analysis of large datasets, improving pattern recognition and anomaly detection.
- Increased Investment: Public and private organizations are allocating significant funds to forensic research. For instance, the U.S. National Institute of Justice allocated USD 4.5 million to the Forensic Technology Center of Excellence, while the Forensic Capability Network secured USD 1.15 million for AI-based evidence collection research [57].
Market Challenges:
- High Costs: The tools required for forensic investigations are often expensive, high-tech equipment developed after extensive research, which can limit adoption, particularly in developing regions [56] [58].
- Lack of Skilled Professionals: A significant shortage of court-certified forensic examiners and professionals with expertise in advanced forensic technology impedes market growth. This shortage is acute outside tier-1 cities and in developing nations [57] [60].
- Data Privacy and Security Concerns: Ensuring the privacy and security of sensitive personal data during forensic investigations remains a significant challenge, especially with evolving regulations like GDPR [58] [61].
Technology Readiness Level (TRL) in Forensic Science
The Technology Readiness Level (TRL) scale is a systematic metric used to assess the maturity of a technology. It was developed at NASA during the 1970s and has since been adopted by the Department of Defense, the European Space Agency, and the European Commission for research and innovation projects [2]. For forensic science research, applying the TRL framework provides a common language for researchers, funding bodies, and industry professionals to evaluate the development stage and implementation potential of new forensic technologies.
Figure 1: The Technology Readiness Level (TRL) scale provides a consistent framework for assessing the maturity of forensic technologies, from basic research (TRL 1-2) to proven operational systems (TRL 9) [1] [2].
TRL Scale Definitions and Applications
| TRL | Stage Definition | Forensic Technology Application Example |
|---|---|---|
| TRL 1-2 | Basic principles observed and reported; technology concept formulated. | Initial research into novel chemical reagents for latent fingerprint detection or foundational studies on DNA analysis techniques. |
| TRL 3-4 | Experimental proof of concept; technology validated in laboratory environment. | Development and initial lab testing of a new microfluidic device for rapid DNA separation or a prototype AI algorithm for fingerprint matching. |
| TRL 5-6 | Technology validated in relevant environment; technology demonstrated in relevant environment. | A new ballistic analysis system tested in a simulated crime lab, or a digital forensics tool trialed by a partner law enforcement agency on historical cases. |
| TRL 7-8 | System prototype demonstration in operational environment; system complete and qualified. | A fully integrated portable DNA sequencer deployed for pilot testing at actual crime scenes or a complete digital forensics platform used in active investigations. |
| TRL 9 | Actual system proven in operational environment. | Established technologies like capillary electrophoresis for DNA profiling or standardized fingerprint analysis systems used routinely in casework worldwide [56]. |
The TRL framework assists management in making decisions concerning technology development and transition. Its advantages include providing a common understanding of technology status, aiding risk management, and informing decisions on technology funding and transition [2]. For forensic science, this is crucial for aligning research with practical, courtroom-admissible outcomes.
Market Segmentation and Regional Analysis
Technology and Application Segmentation
The forensic technology market is diverse, with segmentation based on service, technology, application, and location.
- By Service: The DNA profiling segment is projected to gain a robust revenue share of nearly 45%, driven by its critical role in identifying victims of natural disasters and criminal investigations [56]. The chemical analysis segment is also poised for significant growth, essential for determining alcohol content and analyzing controlled substances [56].
- By Type: The services segment held a dominant market share, driven by the rise of private and government forensic laboratories [57]. Within the product segment, reagents and consumables see strong utilization in lab-based procedures.
- By Application: The judicial and law enforcement segment is set to garner a significant share (36%), driven by the rising generation of DNA databases for criminal investigations [56]. The pharmacogenetics segment, which examines how genetic variants cause differences in how people react to medications, is also anticipated to garner substantial market share by 2035 [56].
- By Location: Laboratory forensics technology is expected to hold a dominant 71% share of the global market, as investigators use advanced lab techniques to solve complex cases like murder, hit-and-run, and arson [56].
Geographical Landscape
North America is the dominant region in the forensic technology market, projected to capture a 36% share by 2035, driven by the presence of well-established forensic laboratories like the FBI and substantial government investment [56] [57]. The region's market size was valued at USD 2.57 billion in 2024 and is expected to reach USD 3.74 billion by 2030 [58]. The Asia Pacific market is expected to be the second largest and fastest-growing region, attributed to increased awareness, adoption of forensic technology, rising criminal cases, and government-backed cyber initiatives [56] [61]. Europe sustains balanced expansion through regulations like the EU AI Act and data-privacy mandates, which drive demand for privacy-preserving forensic tools [60].
Detailed Experimental Protocols in Forensic Technology
Protocol 1: DNA Profiling via Capillary Electrophoresis
DNA profiling is a cornerstone of modern forensic science, used for body fluid identification, paternity testing, and disaster victim identification [56] [62].
1. Principle: DNA profiling works by isolating an individual's unique non-coding DNA sequences (Short Tandem Repeats, STRs) and amplifying them to create a distinct genetic fingerprint that can be compared against reference samples [56].
2. Workflow:
Figure 2: Standard workflow for forensic DNA profiling, a mature technology operating at high TRL levels (8-9) in operational environments [56] [57].
3. Key Reagents and Materials:
| Research Reagent / Solution | Function in Protocol |
|---|---|
| Lysis Buffer | Breaks down cell membranes and nuclear envelopes to release DNA. |
| Proteinase K | Degrades contaminating proteins and nucleases that could damage DNA. |
| Silica-based Membranes or Magnetic Beads | Selectively bind DNA in the presence of chaotropic salts for purification. |
| PCR Master Mix | Contains heat-stable DNA polymerase, dNTPs, and buffer for STR amplification. |
| Fluorescently-labeled STR Primers | Short DNA sequences that specifically target and tag STR regions for detection. |
| Formamide & Size Standards | Used in capillary electrophoresis to denature DNA and provide accurate fragment sizing. |
| Genetic Analyzer with Polymer | The platform and separation matrix for high-resolution capillary electrophoresis. |
4. Data Interpretation: The output is an electropherogram showing peaks corresponding to different STR alleles. This profile is compared against reference samples or uploaded to DNA databases like CODIS (Combined DNA Index System) for potential matches [56].
Protocol 2: Digital Forensics Data Acquisition and Analysis
With the surge in cybercrime, digital forensics has become one of the fastest-growing segments [62] [61].
1. Principle: Digital forensics involves the identification, preservation, analysis, and documentation of digital evidence from electronic devices to be presented in a court of law [56].
2. Workflow:
- Identification: Locate and secure digital devices and potential sources of evidence.
- Preservation: Create a forensic image (bit-for-bit copy) of the storage media using a write-blocker to maintain evidence integrity.
- Analysis: Examine the forensic image using specialized software to recover deleted files, analyze metadata, search for keywords, and trace user activity.
- Documentation: Create a detailed report of all findings and procedures to ensure evidence is admissible in court.
3. Key Tools and Materials:
| Tool / Hardware | Function in Investigation |
|---|---|
| Write-Blocker | A hardware interface that prevents any data from being written to the original evidence drive. |
| Forensic Imaging Station | A high-performance workstation with large storage capacity for creating and analyzing forensic images. |
| Data Acquisition Software | Tools like FTK Imager or dd (Linux) used to create a verifiable bit-stream copy of the data [59]. |
| Forensic Analysis Suites | Integrated platforms (e.g., AccessData, Cellebrite, Oxygen Forensics) for analyzing data from computers, mobiles, and cloud sources [59] [60]. |
| Password Cracking & Decryption Tools | Utilities designed to bypass or recover passwords and decrypt protected files. |
| Mobile Device Extraction Kits | Hardware and software kits for extracting data from smartphones and tablets. |
Key Players and Competitive Landscape
The forensic technology market features a mix of established life science companies and specialized forensic firms. Key players include:
- Life Science and Analytical Instrumentation Companies: Thermo Fisher Scientific, Inc., Agilent Technologies, and GE Healthcare provide essential instruments, reagents, and consumables for forensic analysis [56] [57]. For instance, Agilent partnered with Source Certain to push the boundaries of scientific innovation in forensic and provenance science [56].
- Specialized Forensic Service and Product Providers: LGC Forensics, SPEX Forensics, NMS Labs, Eurofins Medigenomix GmbH, and Forensic Fluids Laboratories offer specialized forensic services and products [56].
- Digital Forensics Specialists: Cellebrite, AccessData, Magnet Forensics, and Oxygen Forensics are leaders in the rapidly growing digital forensics segment, providing tools for mobile device extraction, computer forensics, and cloud investigations [59] [60].
The competitive landscape is characterized by strategic acquisitions and partnerships as companies seek to expand their technological capabilities and geographic reach. For example, QIAGEN completed the acquisition of Verogen, a leader in Next-Generation Sequencing (NGS) for human identification [57].
Future Outlook and Emerging Trends
The future of the forensic technology market will be shaped by several key trends:
- Integration of Artificial Intelligence and Machine Learning: AI is revolutionizing forensics by automating evidence collection, accelerating data analysis, and improving accuracy in threat detection. Machine learning models can analyze vast amounts of unstructured data, identifying patterns and anomalies far more quickly than human analysts [56] [61].
- Growth of Digital and Cloud Forensics: As criminal activity increasingly moves online, digital forensics is the fastest-growing segment. A major emerging sub-segment is cloud forensics, which addresses the challenge of investigating incidents in cloud-native SaaS environments [60] [61].
- Advancements in DNA Sequencing Technologies: Next-Generation Sequencing (NGS) is moving beyond traditional DNA profiling, offering capabilities to analyze degraded samples and provide additional phenotypic information [57].
- Addressing the Skills Gap: The shortage of skilled forensic professionals is driving growth in managed forensic services and leading vendors to invest in training and certification programs to make their tools more accessible [60].
The forensic technology market presents significant growth potential, driven by an undeniable demand for reliable and sophisticated investigative tools in an increasingly complex global security landscape. For researchers and professionals, understanding the Technology Readiness Level framework is crucial for evaluating the maturity and implementation readiness of emerging technologies. Key areas such as DNA profiling, digital forensics, and the integration of AI and machine learning represent both current commercial strengths and future growth frontiers. While challenges related to cost, skilled manpower, and data privacy persist, they also create opportunities for innovation and development. The ongoing modernization of law enforcement and judicial systems worldwide, coupled with continuous technological advancement, ensures that the forensic technology market will remain a dynamic and critical field for years to come.
The Role of Standardization and Proficiency Testing in Achieving TRL 9
The Technology Readiness Level (TRL) scale, originally developed by NASA, provides a systematic measurement system to assess the maturity of a particular technology. Within forensic science, achieving TRL 9 signifies that a scientific method or technique has been proven through successful deployment in operational casework, representing the highest level of technological maturity. This whitepaper examines how the synergistic application of standardization and proficiency testing provides the critical pathway for forensic methodologies to progress from experimental validation (TRL 4-6) to full operational deployment (TRL 9).
The journey to TRL 9 requires demonstrating that a forensic method produces reliable, reproducible results across different laboratories, instruments, and practitioners. Standardization establishes the technical specifications and procedural requirements, while proficiency testing empirically validates performance under controlled conditions that simulate real-world operational environments. Together, these components form an essential framework for validating forensic methodologies to the standard required for judicial acceptance and scientific credibility [63] [64].
Standardization as the Foundation for Technological Maturity
Standards Development Infrastructure
International standards provide the critical infrastructure for forensic science validity and reliability. The most significant initiative currently underway within the global forensic community is the development of International Organization for Standardization (ISO) standards through Technical Committee TC272, which has input from 41 ISO member countries [64]. In the United States, the Organization of Scientific Area Committees (OSAC) for Forensic Science works to develop and promote consensus standards across over 20 forensic disciplines, maintaining a public registry of approved standards [65].
The standards development process involves multi-country stakeholder deliberations and follows rigorous consensus-based approaches. These standards establish specifications and procedures designed to ensure that forensic products, services, and systems are safe, reliable, and consistently perform as intended [64]. They can be used in the accreditation of forensic laboratories and in the certification of products and services, providing the benchmark against which technological maturity is measured.
Current Standards Landscape
The OSAC Registry has demonstrated substantial growth, now containing 225 standards (152 published and 73 OSAC Proposed) as of January 2025 [65]. This represents a significant expansion of the standardized framework for forensic science methodologies. Recent additions to the registry illustrate the breadth of disciplines covered:
- ANSI/ASB Standard 180: Standard for the Use of GenBank for Taxonomic Assignment of Wildlife [65]
- OSAC 2022-S-0037: Standard for DNA-based Taxonomic Identification in Forensic Entomology [65]
- OSAC 2024-S-0012: Standard Practice for the Forensic Analysis of Geological Materials by Scanning Electron Microscopy and Energy Dispersive X-Ray Spectrometry [65]
- SWGDE 17-F-001-2.0: Recommendations for Cell Site Analysis [65]
The development of new standards continues to address emerging technologies and methodologies. For example, in January 2025, ASTM International initiated work on BSR/ASTM WK93266, a New Guide for Capturing Iris Images for Use with Iris Recognition Systems [65]. This ongoing standards development is essential for establishing the foundational requirements that enable technologies to progress toward TRL 9.
Implementation and Impact
Standard implementation is critical for achieving technological maturity. According to OSAC implementation data, 224 Forensic Science Service Providers (FSSPs) had contributed to the implementation survey since the OSAC Program Office started collecting data in 2021, with 72 new contributions in 2024 alone [65]. This growing adoption demonstrates how standardization provides the necessary framework for technologies to advance toward TRL 9 by establishing consistent protocols, validation requirements, and performance benchmarks across forensic laboratories.
Diagram 1: The synergistic relationship between standardization and proficiency testing in achieving TRL 9. Standardization establishes technical and procedural requirements, while proficiency testing provides empirical validation through representative tests and performance metrics.
Proficiency Testing as Empirical Validation
Current Proficiency Testing Infrastructure
Proficiency testing serves as the primary mechanism for empirically validating that forensic methods meet the performance requirements established through standardization. Collaborative Testing Services (CTS) stands as one of the primary providers of forensic proficiency testing, with programs dating to 1978 and currently serving approximately 900 laboratories in more than 80 countries [66] [67]. CTS is accredited to ISO/IEC 17043:2023, the international standard for proficiency testing providers, ensuring their tests meet rigorous quality requirements [66].
These proficiency tests are designed to evaluate participant performance against pre-established criteria through interlaboratory comparisons. They serve multiple essential functions in the progression to TRL 9:
- Providing external validation of laboratory competency
- Identifying problematic procedures or methodologies
- Establishing performance benchmarks for emerging technologies
- Informing accrediting bodies about laboratory capabilities [67]
Methodologies and Protocols
Effective proficiency testing requires carefully designed methodologies that accurately assess technical capabilities. The CTS testing protocol involves several critical phases that collectively provide comprehensive performance assessment:
Test Design and Manufacturing: Strict protocols are observed for manufacturing forensic tests, with predistribution review conducted by qualified experts to assess test design and samples [66].
Test Administration: Proficiency tests can be conducted through various formats, including traditional known samples, digital simulations, and increasingly, blind testing scenarios.
Performance Assessment: Participant results are evaluated against pre-established criteria, with individual and aggregate performance data analyzed.
Feedback and Correction: Summary reports allow laboratories to compare their performance with peers and identify areas requiring improvement.
The Houston Forensic Science Center (HFSC) has implemented an advanced approach through its blind quality control program, which aims to provide real-time assessment of analysis procedures, determine areas of improvement, and ensure stakeholders receive accurate and reliable results [68]. This program, implemented across almost all units within the laboratory, represents a significant advancement in proficiency testing methodology by eliminating the potential for "special practices" that analysts might employ when they know they are being tested [68] [67].
Quantitative Assessment of Current Proficiency Testing
Research into proficiency testing outcomes provides critical data on the current state of forensic methodology validation. A comprehensive study of latent print examination proficiency tests revealed exceptionally high performance levels, with examiners giving correct responses to 99.6% of test items [67]. This study surveyed 322 latent print examiners after they completed a CTS proficiency test and correlated their perceptions with objective quality metrics.
Table 1: Latent Print Examiner Proficiency Test Performance and Perceptions
| Test Characteristic | Measurement Result | Implications for TRL Progression |
|---|---|---|
| Overall Accuracy Rate | 99.6% correct responses | Demonstrates high reliability for established methods |
| Perceived Difficulty | 4.27/10 (0=Extremely Easy) | Suggests tests may not fully represent casework challenges |
| Similarity to Casework | 5.56/10 (0=Not Similar) | Indicates need for more representative test materials |
| Confidence in Results | 90.3% very confident | Reflects methodological maturity and practitioner trust |
| Error Rate | 0.4% (extremely rare) | Provides empirical data for legal proceedings |
The study also implemented objective quality assessment using the LQMetrics algorithm, which provides deterministic and objective scores independent of any single examiner. Results indicated that proficiency test prints generally received higher clarity scores than prints from normal casework, suggesting that current testing may not adequately represent real-world challenges [67]. This finding highlights a critical consideration for TRL 9 validation: proficiency testing must sufficiently simulate operational conditions to truly validate technological maturity.
Integrated Framework for Achieving TRL 9
Synergistic Implementation Pathway
The progression to TRL 9 requires seamless integration of standardization and proficiency testing throughout the technology development lifecycle. The following experimental protocol outlines a comprehensive pathway for validating forensic methodologies to TRL 9 standards:
Phase 1: Standards Development and Alignment
- Identify relevant existing standards through OSAC Registry and ISO TC272 work programs [65] [64]
- Develop method-specific validation protocols aligned with standard requirements
- Establish acceptance criteria based on standardized performance metrics
Phase 2: Initial Validation Testing
- Conduct intra-laboratory validation following standardized protocols
- Implement internal proficiency testing with progressively challenging samples
- Refine methodologies based on initial performance data
Phase 3: Interlaboratory Proficiency Testing
- Engage with accredited providers (e.g., CTS) for external proficiency testing [66]
- Participate in multi-laboratory comparison studies
- Collect comprehensive performance data across diverse operational environments
Phase 4: Implementation and Continuous Monitoring
- Integrate methodology into operational casework with quality controls
- Implement blind proficiency testing programs, following HFSC model [68]
- Establish ongoing performance monitoring through quality management systems
Phase 5: Accreditation and Recognition
- Seek laboratory accreditation to ISO/IEC 17025 requirements [63]
- Document method validation and performance metrics for judicial notice
- Contribute to standards refinement based on operational experience
Quality Metrics and Quantitative Assessment
The development of objective quality metrics represents a significant advancement in quantifying methodological performance. Algorithms such as LQMetrics provide quantitative, deterministic measures of evidence quality that enable systematic difficulty adjustment in proficiency testing [67]. Implementation of these metrics enables:
- Standardized difficulty assessment across test materials
- Direct comparison between proficiency tests and casework materials
- Empirical establishment of minimum quality thresholds
- Objective tracking of methodological improvements over time
Research utilizing these metrics has demonstrated that including prints of lower quality in proficiency testing may increase both the difficulty and representativeness of testing, better simulating real-world casework conditions [67]. This approach provides more meaningful validation of a technology's readiness for operational deployment.
Table 2: Key Research Reagent Solutions for Forensic Methodology Validation
| Resource Category | Specific Examples | Function in TRL Validation |
|---|---|---|
| Proficiency Test Providers | Collaborative Testing Services (CTS) | Provides interlaboratory comparison testing for performance validation [66] |
| Quality Metric Algorithms | LQMetrics (FBI Universal Latent Workstation) | Delivers objective, quantitative assessment of evidence quality and difficulty [67] |
| Standards Repositories | OSAC Registry, ISO TC272 Standards | Supplies validated protocols and technical requirements for method development [65] [64] |
| Blind Testing Frameworks | Houston Forensic Science Center Protocol | Enables realistic assessment of operational performance without examiner bias [68] |
| Accreditation Standards | ISO/IEC 17025, ISO/IEC 17043 | Establishes baseline requirements for laboratory competence and testing reliability [66] [63] |
Diagram 2: Integrated pathway for achieving TRL 9 through sequential implementation of standardization and proficiency testing activities, progressing from initial development to continuous monitoring and accreditation.
Achieving TRL 9 in forensic science requires robust empirical validation through the integrated application of standardization and proficiency testing. Standardization establishes the technical foundations and performance benchmarks, while proficiency testing provides the empirical evidence of reliability across diverse operational conditions. The current infrastructure—including the OSAC Registry with 225 standards, ISO TC272 international standards, and accredited proficiency test providers like CTS—provides a solid foundation for this validation process [65] [66] [64].
However, research indicates that current proficiency testing practices must evolve to more accurately simulate real-world challenges, potentially through the incorporation of more representative test materials and expanded implementation of blind testing protocols [68] [67]. The development and implementation of objective quality metrics offers promising approaches for quantitatively assessing test difficulty and representativeness.
For researchers and developers seeking to advance forensic methodologies to TRL 9, a systematic approach incorporating early alignment with established standards, progressive validation through increasingly realistic proficiency testing, and continuous performance monitoring provides the most reliable pathway to operational deployment and judicial acceptance.
Conclusion
The Technology Readiness Level scale provides an indispensable, structured framework for guiding forensic technologies from conceptual research to reliable, court-admissible tools. Success hinges on a disciplined approach that integrates rigorous scientific validation with an acute awareness of legal standards from the earliest development stages. The future of forensic science will be shaped by technologies like Next-Generation Sequencing and AI-driven analytics, which must navigate the TRL pathway efficiently. For researchers and developers, overcoming the 'valley of death' between pilot demonstration and operational deployment requires strategic partnerships, sustained funding, and a focus on standardized validation. Ultimately, a TRL-guided approach ensures that innovations not only achieve technical maturity but also fulfill the critical demands of the justice system for accuracy, reliability, and transparency, thereby strengthening the integrity of forensic science as a whole.
References
- 1. Technology Readiness Levels [https://www.nasa.gov/directorates/somd/space-communications-navigation-program/technology-readiness-levels/]
- 2. Technology readiness level [https://en.wikipedia.org/wiki/Technology_readiness_level]
- 3. Technology readiness levels [https://ised-isde.canada.ca/site/innovation-canada/en/technology-readiness-levels]
- 4. A Tool to Navigate Hype Cycles: Technology Readiness ... [https://www.forbes.com/sites/shalinjyotishi/2025/01/27/tool-to-navigate-hype-cycles-technology-readiness-levels/]
- 5. Are We Ready for It? A Review of Forensic Applications and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12012292/]
- 6. Technology Readiness Levels (TRLs) in the Era of Co- ... [https://www.mdpi.com/2571-5577/7/2/32]
- 7. Technology Readiness Levels (TRL) in Space Projects | TTMS [https://ttms.com/technology-readiness-levels-trl-in-space-projects-explanation-and-significance/]
- 8. What are Technology Readiness Levels (TRLs)? [https://hellodarwin.com/blog/technology-readiness-levels-trl]
- 9. What are Technology Readiness Levels (TRL)? - TWI [https://www.twi-global.com/technical-knowledge/faqs/technology-readiness-levels]
- 10. The Future of Forensic Analysis: The Latest Studies [https://www.spectroscopyonline.com/view/the-future-of-forensic-analysis-the-latest-studies]
- 11. 10 Modern Forensic Technologies Used Today [https://www.forensicscolleges.com/blog/resources/10-modern-forensic-science-technologies]
- 12. 1. What Is A Trl? - FHWA-HRT-17-047 [https://www.fhwa.dot.gov/publications/research/ear/17047/001.cfm]
- 13. What is a Technology Readiness Level (TRL) and why ... [https://bbcetc.com/dod/what-is-a-technology-readiness-level-trl-and-why-does-it-matter-to-you/]
- 14. Forensic Science Research and Development Technology ... [https://nij.ojp.gov/topics/articles/forensic-science-research-and-development-technology-working-group-operational]
- 15. Thin-layer chromatography (TLC) | Research Starters [https://www.ebsco.com/research-starters/chemistry/thin-layer-chromatography-tlc]
- 16. Forensic Data Science Laboratory [https://forensic-data-science.net/]
- 17. EC Report on Technology Readiness Levels (TRL) to ... [https://www.era-learn.eu/news-events/news/ec-report-on-technology-readiness-levels-trl-to-analyse-technology-progression-in-horizon-europe]
- 18. Technology Readiness Levels (TRLs) [https://esto.nasa.gov/trl/]
- 19. Demystifying Comprehensive Two-Dimensional Gas ... [https://concentratingonchromatography.com/two-dimensional-gas-chromatography?hsLang=en]
- 20. Integrated Technology Readiness Levels (TRLs) [https://medicalcountermeasures.gov/trl/integrated-trls]
- 21. Technology Readiness Level Roadmap for Developing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11206302/]
- 22. NIST Report Outlines Strategic Opportunities to Advance ... [https://www.nist.gov/news-events/news/2024/09/nist-report-outlines-strategic-opportunities-advance-forensic-science-us]
- 23. Forensic Science Strategic Research Plan, 2022-2026 ... [https://nij.ojp.gov/topics/articles/forensic-sciences-strategic-research-plan-2022-2026]
- 24. The Concept of Technology Readiness Level 0 (TRL 0) [https://buildingmoonshots.substack.com/p/the-concept-of-technology-readiness]
- 25. The Differences Between the Frye & the Daubert Standards [https://burnswhite.com/differences-between-frye-daubert/]
- 26. Understanding the Daubert Standard vs. Frye ... [https://intellex.com/expert-insights/daubert-vs-frye-standard/]
- 27. Daubert vs. Frye: Standards of Admissibility for ... [https://www.expertinstitute.com/resources/insights/daubert-vs-frye-navigating-the-standards-of-admissibility-for-expert-testimony/]
- 28. Kelly-Frye, Daubert, Mohan, and Why You Need to ... [https://www.axon.com/resources/kelly-frye-daubert-mohan-and-why-you-need-to-understand-these-cases]
- 29. The Results Are In: Forensic Labs Under Prosecutorial ... [https://www.chicagoappleseed.org/2025/09/23/in-house-forensics-labs-are-problematic/]
- 30. How DNA Labs Are Holding the Line as Federal Funding ... [https://promega.foleon.com/theishireport/the-ishi-report-august-2025/undermined-how-dna-labs-are-holding-the-line-as-federal-funding-falters]
- 31. AAFS 2025: Heidi Eldridge Talks About Funding Issues in ... [https://www.spectroscopyonline.com/view/aafs-2025-heidi-eldridge-talks-about-funding-issues-in-forensic-science]
- 32. NIJ: Anticipated Forensic Research Interests in 2025 [https://www.forensicmag.com/3594-All-News/617061-NIJ-Anticipated-Forensic-Research-Interests-in-2025/]
- 33. What is Pilot Testing? [https://testrigor.com/blog/what-is-pilot-testing/]
- 34. Pilot project excellence: A comprehensive how-to (+ an ... [https://www.teamwork.com/blog/pilot-project-example/]
- 35. 7 Types of Comparison Charts for Effective Data Visualization [https://ninjatables.com/types-of-comparison-charts/?srsltid=AfmBOopeAtLgzs7FTY8OJohikrQelLK_ohmrUqM0qsrjSTEkq0Ev9p3o]
- 36. 3.2 Using Visuals – Technical Writing at LBCC [https://openoregon.pressbooks.pub/lbcctechwriting/chapter/3-2-using-visuals/]
- 37. Advancing a paradigm shift in evaluation of forensic evidence [https://pmc.ncbi.nlm.nih.gov/articles/PMC9133770/]
- 38. Technology Readiness Levels (TRLs) in the Era of Co- ... [https://www.preprints.org/manuscript/202308.1740]
- 39. Collaborative innovation during the drug discovery and ... [https://www.sciencedirect.com/science/article/pii/S1359644625001229]
- 40. Analyzing collaboration dynamics in new drug R&D [https://pmc.ncbi.nlm.nih.gov/articles/PMC12162338/]
- 41. Pharma Giants Unveil Collaborative Drug Development ... [https://www.americanhhm.com/technotrends/pharma-giants-unveil-collaborative-drug-development-initiative]
- 42. An integrated methodological framework for the validation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11721250/]
- 43. A guideline for the validation of likelihood ratio methods ... [https://www.sciencedirect.com/science/article/abs/pii/S0379073816301359]
- 44. Understanding Error Rate: A Crucial Guide for Professionals [https://helio.app/ux-research/design-metrics/understanding-error-rate-a-crucial-guide-for-professionals/]
- 45. Unlocking CRL and TRL: Essential Frameworks for Product ... [https://scimitaventures.com/insights/mastering-crl-vs-trl-choosing-the-right-development-stage-for-your-projects-successnbsp]
- 46. CRL - Commercial Readiness Level [https://www.bindt.org/What-is-NDT/Index-of-acronyms/C/crl/]
- 47. Evaluating an organisation's product development. [https://www.tamegon.com/trl-crl-levels]
- 48. Forensic Readiness - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/computer-science/forensic-readiness]
- 49. Multi-criteria decision analysis for evaluating carbon ... [https://www.sciencedirect.com/science/article/pii/S1364032125003727]
- 50. Next-Generation Sequencing (NGS) for Forensics [https://www.thermofisher.com/us/en/home/industrial/forensics/human-identification/forensic-dna-analysis/dna-analysis/next-generation-sequencing-ngs-forensics.html]
- 51. Forensic Genomics | NGS solutions for forensic applications [https://www.illumina.com/areas-of-interest/forensic-genomics.html]
- 52. Emerging Technologies in Forensic DNA Analysis [https://www.sciepublish.com/article/pii/279]
- 53. Embracing Next Generation Methods for Forensic DNA ... [https://nij.ojp.gov/topics/articles/embracing-next-generation-methods-forensic-dna-sequence-analysis]
- 54. Rapid NGS Analysis on Google Cloud Platform [https://pmc.ncbi.nlm.nih.gov/articles/PMC12627752/]
- 55. The Impact of Rapid DNA Sequencing on Modern Forensic ... [https://www.intechopen.com/chapters/1213623]
- 56. Forensic Technology Market Size, Share & Forecast 2035 [https://www.researchnester.com/reports/forensic-technology-market/5074]
- 57. Forensic Technology Market Size & Share Report, 2032 [https://www.fortunebusinessinsights.com/forensic-technology-market-110927]
- 58. Forensic Technology Market Size, Growth & Forecast 2030 [https://marksparksolutions.com/reports/forensic-technology-market-2025-2030]
- 59. Global Forensic Technology Market Set to Reach USD ... [https://www.openpr.com/news/4211106/global-forensic-technology-market-set-to-reach-usd-32-1-billion]
- 60. Digital Forensics Market Size, Growth Drivers & Forecast ... [https://www.mordorintelligence.com/industry-reports/digital-forensics-market]
- 61. Digital Forensics Market Size to Hit USD 47.9 Billion by 2034 [https://www.precedenceresearch.com/digital-forensics-market]
- 62. Forensic Technology Market to be valued USD 32.94 ... [https://www.eletimes.ai/forensic-technology-market-to-be-valued-at-usd-32-94-billion-by-2025]
- 63. The Need for Standards Unification in Forensic Laboratory ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9280477/]
- 64. The international development of forensic science standards [https://www.sciencedirect.com/science/article/pii/S037907381830166X]
- 65. OSAC Standards Bulletin - January 2025 | NIST [https://www.nist.gov/magazine/osac-standards-bulletin/january-2025]
- 66. Collaborative Testing Services, Inc (CTS) Forensics Testing ... [https://cts-forensics.com/index-forensics-testing.php]
- 67. How do latent print examiners perceive proficiency testing ... [https://www.sciencedirect.com/science/article/abs/pii/S1355030619301108]
- 68. Crime Lab Proficiency Testing and Quality Management [https://forensicstats.org/blog/portfolio/crime-lab-proficiency-testing-and-quality-management/]