Foundational Research in Novel Psychoactive Substances (NPS) Identification: Trends, Methods, and Future Directions
This article provides a comprehensive overview of the foundational research landscape for identifying Novel Psychoactive Substances (NPS).
Foundational Research in Novel Psychoactive Substances (NPS) Identification: Trends, Methods, and Future Directions
Abstract
This article provides a comprehensive overview of the foundational research landscape for identifying Novel Psychoactive Substances (NPS). It synthesizes the most current data on emerging NPS trends, including the proliferation of synthetic opioids, benzodiazepines, and non-pharmaceutical adulterants like xylazine and medetomidine. Aimed at researchers, scientists, and drug development professionals, the content explores advanced methodological frameworks for NPS detection, discusses challenges in laboratory analysis and public health response, and evaluates validation strategies for testing protocols. The article concludes by highlighting critical research gaps and future directions necessary to combat the dynamic and expanding threat of NPS to public health and safety.
The Evolving NPS Landscape: Key Substances and 2025 Prevalence Trends
The landscape of illicit drug use is being rapidly reshaped by the emergence of novel psychoactive substances (NPS), posing unprecedented challenges for public health, forensic science, and clinical medicine. Among these, three substances have recently garnered significant attention: xylazine, medetomidine, and tianeptine. These compounds exemplify the evolving complexity of the illicit drug market, where substances are increasingly adulterated with potent synthetic chemicals not intended for human consumption. According to the United Nations Office on Drugs and Crime, NPS are defined as substances of abuse that are not controlled by international drug conventions but which may pose a public health threat [1]. The rapid proliferation of these substances creates a critical knowledge gap regarding their toxicology, detection methodologies, and clinical management, necessitating foundational research to inform evidence-based responses.
This technical guide provides an in-depth analysis of the pharmacological properties, analytical detection methods, and public health implications of these three emerging threats. Within the broader context of NPS identification and research, understanding these specific substances is paramount for developing effective surveillance systems, treatment protocols, and harm reduction strategies in an increasingly volatile drug supply landscape.
Substance Profiles and Epidemiological Trends
Comparative Pharmacology
Table 1: Pharmacological Profiles of Emerging NPS Threats
| Parameter | Xylazine | Medetomidine | Tianeptine |
|---|---|---|---|
| Primary Pharmacological Class | Alpha-2 adrenergic receptor agonist [2] | Alpha-2 adrenergic receptor agonist [3] | Atypical tricyclic compound with mu-opioid receptor agonist activity [4] |
| Primary Approved Use | Veterinary sedative and analgesic [2] | Veterinary sedative [3] [5] | Antidepressant (in some countries; not approved in the U.S.) [4] |
| Relative Potency | Reference agonist | 100-200 times more potent than xylazine [3] [5] | N/A |
| Key Clinical Effects | Deep sedation, bradycardia, hypotension, severe skin wounds [2] | Profound sedation, bradycardia, severe autonomic withdrawal [3] [5] | Opioid-like euphoria, respiratory depression (at high doses) [4] |
| Response to Naloxone | Sedation not reversed [2] | Sedation not reversed [5] [6] | Reversible (due to opioid activity) |
Emerging Prevalence and Distribution
The infiltration of these substances into the illicit drug supply, particularly the opioid supply, represents a significant shift in the NPS landscape. Medetomidine has emerged as a particularly rapid and potent threat. Initially detected in Philadelphia's drug supply in May 2024, it quickly supplanted xylazine as the most common adulterant in the illegal opioid supply in that region. Within six months, its prevalence in Philadelphia drug samples skyrocketed from 29% to 87%, while xylazine presence fell from 97% to 42% in the same period [5]. This trend is not isolated; similar emergences have been documented in Chicago, Illinois [6], North Carolina, Ohio, and other states [5].
National data from toxicology laboratories corroborate this trend. In the first half of 2025, xylazine and its metabolite 4-hydroxyxylazine constituted the largest proportion of compounds detected in the "NPS-Other" class, though their prevalence showed a decreasing trend from Q1 to Q2 [4]. Conversely, detections of medetomidine and its metabolite 3-hydroxy medetomidine increased by 34% and 29%, respectively, during the same period, indicating rapid proliferation [4]. Tianeptine also showed a marked increase, with detections of its metabolite MC5 rising by 44% from Q1 to Q2 2025 [4]. These data underscore the dynamic and interconnected nature of the illicit drug market, where one potent adulterant can rapidly displace another.
Analytical Methodologies for Identification and Quantification
The accurate identification and quantification of xylazine, medetomidine, tianeptine, and their metabolites in biological specimens are foundational to clinical toxicology, forensic analysis, and public health surveillance. The constant emergence of new substances demands robust, sensitive, and comprehensive analytical workflows.
Sample Preparation and Extraction
Proper sample preparation is critical for reliable results. Common biological matrices include blood (whole blood, plasma, serum), urine, and oral fluid [2]. Protein precipitation is a typical first step for blood-based matrices, often using solvents like acetonitrile or methanol. This is frequently followed by a solid-phase extraction (SPE) or liquid-liquid extraction (LLE) to isolate the analytes of interest from the complex biological matrix and reduce ion suppression/enhancement effects in mass spectrometry. The choice of extraction method and solvents is optimized based on the chemical properties (e.g., polarity, pKa) of the target analytes and their metabolites.
Instrumental Analysis
Liquid Chromatography coupled with Tandem Mass Spectrometry (LC-MS/MS) is the gold-standard technique for the simultaneous identification and quantification of these NPS and their metabolites in biological samples due to its high sensitivity and specificity [2].
- Liquid Chromatography (LC): Reversed-phase chromatography (e.g., C18 column) is standard. Mobile phases typically consist of water and an organic modifier (e.g., methanol or acetonitrile), often with additives like formic acid or ammonium formate to improve chromatographic separation and ionization efficiency. The LC system effectively separates the analytes from each other and from matrix interferences.
- Mass Spectrometry (MS): Electrospray Ionization (ESI) in positive mode is commonly used. The analysis is performed in Multiple Reaction Monitoring (MRM) mode, where the precursor ion for each analyte is selected in the first quadrupole, fragmented in the collision cell, and one or more characteristic product ions are monitored in the third quadrupole. This MRM transition provides a highly specific "fingerprint" for each compound.
Table 2: Key Analytical Targets and Methodological Considerations
| Analyte | Key Metabolites | Primary Biospecimens | Major Analytical Challenge |
|---|---|---|---|
| Xylazine | 4-Hydroxyxylazine [2] [4] | Blood, Urine, Oral Fluid [2] | Co-extraction and separation from other basic drugs and adulterants; wide range of concentrations in postmortem vs. clinical samples. |
| Medetomidine | 3-Hydroxy medetomidine [4] | Blood, Urine, Oral Fluid [2] | Extremely low concentrations (ng/mL range) requiring high analytical sensitivity [2]. |
| Tianeptine | MC5 [4] | Blood, Urine | Lack of commercially available reference standards for all potential metabolites. |
The following diagram illustrates the core workflow for the LC-MS/MS analysis of these substances in biological specimens:
Data Interpretation and Confirmation
The identification of a substance is confirmed by matching both the retention time and the MRM transition(s) of the analyte in the sample with those of a certified reference standard. Quantification is achieved by constructing a calibration curve using analyte-spiked matrix samples. Quality control samples at multiple concentrations are essential to ensure accuracy and precision. Given the complexity of polydrug exposures, methods must be designed to detect a wide panel of substances, including fentanyl analogs, benzodiazepines, and other stimulants, which are commonly co-ingested [4] [6].
Mechanisms of Action and Clinical Toxicodynamics
Understanding the distinct mechanisms of action of these substances is critical for predicting their toxicological profiles and developing targeted therapeutic interventions.
Xylazine and Medetomidine: Alpha-2 Adrenergic Agonism
Both xylazine and medetomidine are centrally-acting alpha-2 adrenergic receptor agonists. Their primary mechanism involves agonism at presynaptic α2-receptors in the central nervous system, particularly in the locus coeruleus. This action inhibits norepinephrine release, leading to a decrease in sympathetic outflow and resulting in sedative, analgesic, and muscle relaxant effects [3] [2]. The key differentiator is potency; medetomidine is 100-200 times more potent than xylazine at the alpha-2 receptor, explaining its more profound sedative effects and the severity of its autonomic withdrawal syndrome [3] [5]. The following diagram outlines the core signaling pathway and physiological effects:
Abrupt cessation after chronic use can precipitate a severe autonomic withdrawal syndrome, characterized by a hyperadrenergic state. This is particularly severe with medetomidine, manifesting as hypertension, tachycardia, tremors, and uncontrollable vomiting, often requiring intensive care management [3] [5]. It is crucial to note that because their effects are mediated through adrenergic, not opioid, receptors, the profound sedation they cause is not reversible with naloxone [5] [2] [6].
Tianeptine: Atypical Opioid Activity
Tianeptine presents a complex pharmacological profile. While structurally an atypical tricyclic compound and originally marketed as an antidepressant, its abuse potential stems from its activity as a mu-opioid receptor agonist [4]. At high doses, it produces opioid-like effects, including euphoria and respiratory depression, and can lead to the development of tolerance and physical dependence. Withdrawal from tianeptine resembles classic opioid withdrawal. This dual nature complicates its clinical presentation and management.
The Scientist's Toolkit: Essential Research Reagents and Materials
Advancing research on these emerging threats requires a specific set of reagents, reference materials, and analytical tools.
Table 3: Key Research Reagents and Materials for NPS Investigation
| Reagent / Material | Function and Application in Research |
|---|---|
| Certified Reference Standards | Pure, quantified analytical standards of xylazine, medetomidine, tianeptine, and their known metabolites (e.g., 4-hydroxyxylazine, 3-hydroxy medetomidine, MC5). Essential for method development, calibration, and confirmation in LC-MS/MS analysis [2] [4]. |
| Stable Isotope-Labeled Internal Standards | (e.g., deuterated analogs like xylazine-d4). Added to samples prior to processing to correct for matrix effects and losses during sample preparation, ensuring quantitative accuracy in mass spectrometry. |
| Solid-Phase Extraction (SPE) Cartridges | Used for clean-up and concentration of analytes from complex biological matrices (blood, urine) to reduce ion suppression and improve sensitivity in LC-MS/MS [2]. |
| Chromatography Columns | High-quality UPLC/HPLC columns (e.g., C18) for optimal separation of target analytes, their metabolites, and potential isobaric interferences. |
| Mass Spectrometry Instrumentation | LC-MS/MS systems capable of high-sensitivity analysis in Multiple Reaction Monitoring (MRM) mode. The core instrument for definitive identification and quantification [2]. |
| Drug Checking Test Strips | Immunoassay-based strips (e.g., medetomidine test strips) for rapid, point-of-care screening of substances in a community or harm reduction setting [5]. |
The emergence of xylazine, medetomidine, and tianeptine within the illicit drug supply represents a critical and evolving public health crisis. Their presence, particularly as adulterants in opioids like fentanyl, complicates clinical management of overdose and withdrawal, stretches healthcare system resources, and increases risks for people who use drugs [3] [5] [6].
The rapid displacement of xylazine by the more potent medetomidine in some regions demonstrates the volatile nature of the NPS market and the need for agile surveillance systems [3] [4]. Key public health responses include the expansion of harm reduction services (such as distribution of naloxone and medetomidine test strips), development of clinical guidelines for managing severe withdrawal syndromes, and enhancing forensic and toxicological capacity for rapid identification [5] [2] [6].
Foundational research into the pharmacology, toxicokinetics, and health impacts of these substances is paramount. Future efforts must focus on elucidating the long-term health consequences, developing targeted antagonist or treatment therapies, and implementing robust, real-time drug early warning systems to mitigate the harms of the next wave of novel psychoactive substances.
The illicit drug supply is characterized by its dynamic and evolving nature, driven by the continuous emergence of novel psychoactive substances (NPS). These substances are designed to mimic the effects of controlled drugs while circumventing legal restrictions, posing significant challenges to public health, forensic science, and clinical response [1]. Among the most concerning classes of NPS are synthetic opioids, which include fentanyl analogs, nitazenes, and emerging compounds like methylfentanyl isomers. These substances are of particular concern due to their high potencies and association with overdose deaths [7] [8]. This whitepaper provides an in-depth technical analysis of the current landscape of these three specific synthetic opioid groups, focusing on their identification, prevalence, and the advanced analytical methodologies required for their detection in a research context.
Chemical and Pharmacological Profiles
Fluoro Fentanyl and its Isomers
Fluoro fentanyl is a synthetic opioid and a fentanyl analog that has been a dominant force in the NPS opioid market. Its name refers to the addition of a fluorine atom to the fentanyl core structure, which exists in different positional isomers (ortho-, meta-, and para-) that standard immunoassays may not distinguish [4]. The para-fluorofentanyl isomer has been specifically and frequently associated with overdose deaths [4]. For multiple years, fluoro fentanyl was the most frequently detected NPS in some datasets. Although it was overtaken by the non-opioid sedative xylazine in 2023, it remains the second most prevalent NPS detected overall in certain large-scale testing programs [4]. Its potency is considered to be similar to or slightly less than that of fentanyl itself [7].
Nitazenes (Benzimidazole-Opioids)
Nitazenes are a class of synthetic opioids that are structurally distinct from fentanyl, characterized by a benzimidazole core. Historically, they were developed in the 1950s as potential analgesics but were never approved for medical use in humans due to their high risk of overdose [7]. These compounds are potent mu-opioid receptor agonists, with some analogs exhibiting a potency several times greater than fentanyl [8]. Their emergence in the illicit drug supply represents a significant escalation of the opioid crisis. The group includes substances such as metonitazene, N-desethyl metonitazene, and N-pyrrolidino protonitazene [4] [9]. The high potency of nitazenes presents a grave public health threat; for instance, seizures of tablets containing an average of 29 mg of metonitazene have been documented, an amount equivalent to approximately 145 times the estimated fatal dose of fentanyl [8].
Methylfentanyl Isomers
Methylfentanyl represents the latest wave of synthetic opioids to emerge in the recreational drug supply. This compound appears in three positional isomeric forms: ortho-, meta-, and para-methylfentanyl [4] [8]. The ortho-methylfentanyl isomer has been the subject of recent public health alerts, having first emerged in early 2023 in British Columbia, Canada, before spreading to the United States [8]. It is an active mu-opioid receptor agonist with a potency considered similar to that of fentanyl [8]. Its identification in over 200 forensic specimens from North America in a short timeframe underscores its rapid proliferation [8]. In 2023, it was reported as the most commonly identified NPS in samples analyzed by Health Canada's Drug Analysis Service [9].
Table 1: Comparative Profile of Key Synthetic Opioids
| Opioid / Analog | Structural Class | Relative Potency | Key Metabolites/Related Compounds | Primary Public Health Concern |
|---|---|---|---|---|
| Fluoro Fentanyl | Fentanyl analog (phenylpiperidine) | Similar or slightly less than fentanyl [7] | Despropionyl fluorofentanyl, Fluoro norfentanyl [4] | High prevalence; specific isomers (e.g., para-) linked to fatalities [4] |
| Nitazenes | Benzimidazole-opioid | Several times more potent than fentanyl (varies by analog) [8] | N-desethyl metonitazene, 4-hydroxy nitazene [4] | Extreme potency; high lethality even in small quantities [8] |
| Methylfentanyl (ortho-) | Fentanyl analog (phenylpiperidine) | Similar to fentanyl [8] | Not specified in sources | Rapid emergence and proliferation across North America [8] |
Analytical Methodologies for Identification and Quantification
The identification of synthetic opioids in complex street-drug mixtures requires sophisticated analytical techniques capable of high sensitivity, specificity, and the ability to perform non-targeted discovery.
High-Resolution Mass Spectrometry (HRMS)
Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry (LC-QTOF-MS) is a powerful tool for the non-targeted screening of emerging substances. This technique combines chromatographic separation with high-resolution accurate mass (HRAM) measurement. As applied in one study, analytical separation was achieved using an ACQUITY UPLC BEH C18 column (2.1 mm x 150 mm, 1.8 µm particle size) at 50°C with a mobile phase flow rate of 0.4 mL/min [8]. The QTOF instrument operates in positive electrospray ionization mode, scanning a mass range of 50-1000 m/z. Identification criteria for a compound include: a chromatographic peak within ±0.25 minutes of the expected retention time, an observed precursor mass within ±5 ppm of the theoretical mass, and the presence of fragment ions within ±2 mDa of theoretical fragments [8]. This method is particularly valuable for identifying new substances without available reference standards by enabling the determination of molecular formulae from accurate mass and the prediction of structures using in silico fragmentation tools [10].
Paper-Spray Mass Spectrometry (PS-MS)
Paper-spray mass spectrometry is an ambient ionization technique that allows for rapid analysis of complex street-drug samples with minimal preparation. A developed PS-HRMS method using a data-dependent acquisition (DDA) mode has been shown effective for detecting newly emerging compounds [10]. In this workflow, a small mass of street-drug sample (0.5–2.1 mg) is dissolved in methanol to create a 1 mg/mL solution. A small volume (10 µL) of this solution is then deposited onto a specialized paper substrate mounted on a sample plate [10]. A spray solvent (e.g., acetonitrile/water/formic acid, 90/9.9/0.1 v/v%) is applied, and a high voltage is used to generate ions directly from the paper substrate for mass spectrometric analysis. This technique can be coupled to high-resolution mass spectrometers, combining rapid analysis with the ability to perform untargeted screening [10].
Targeted Quantification Methods
For precise quantification of known synthetic opioids, liquid chromatography-tandem mass spectrometry (LC-MS/MS) is the gold standard. This technique operates in multiple reaction monitoring (MRM) mode, which offers high sensitivity and selectivity. A typical methodology involves a triple quadrupole mass spectrometer with an electrospray ionization source. Samples are prepared by dilution in an internal standard solution containing deuterated analogs of target analytes to correct for matrix effects and instrument variability [10]. This approach was used to quantify a panel of over 100 targeted compounds, including opioids, adulterants, and cutting agents, providing precise concentration data crucial for understanding drug potency and overdose risk [10].
Diagram 1: Synthetic Opioid Analysis Workflow
Current Prevalence and Emerging Trends
Recent data from the first half of 2025 reveals a dynamic and shifting landscape of synthetic opioids. The following table summarizes quantitative data on the prevalence of key substances.
Table 2: Synthetic Opioid Prevalence and Trends in 2025 (Q1-Q2)
| Substance Category | Specific Compound | Prevalence in H1 2025 (Positives) | Q1 to Q2 2025 Trend (% Change in Proportion) | Common Co-occurring Substances |
|---|---|---|---|---|
| Designer Opioids | Fluoro Fentanyl & related compounds | ~59% of designer opioid detections in Q1 [4] | ~13% decrease (metabolite) [4] | Fentanyl, Xylazine [4] [8] |
| ortho-Methylfentanyl | Emerging, >200+ forensic specimens (North America) [8] | Significant increase [4] | Fentanyl, para-fluorofentanyl [8] | |
| Despropionyl m/p-Methylfentanyl | Similar proportion to N-desethyl metonitazene in Q1 [4] | 109% increase [4] | Not specified | |
| N-desethyl metonitazene (Nitazene) | Significant proportion in Q1 [4] | 77% decrease [4] | Fentanyl, NPS Benzodiazepines [4] [8] | |
| Norcarfentanil | Low detection count [4] | 121% increase [4] | Not specified | |
| Acryl Fentanyl | Low detection count [4] | 164% increase [4] | Not specified | |
| Emerging Adulterants | Xylazine | Highest prevalence in NPS-Other class (~70% in Q1) [4] | -40% (metabolite) [4] | Fentanyl, synthetic opioids [4] |
| Medetomidine | Second most prevalent in NPS-Other class [4] | 34% increase [4] | Not specified |
The data indicates that while fluoro fentanyl remains highly prevalent, its dominance may be waning slightly [4]. Meanwhile, methylfentanyl isomers are showing a marked increase, with despropionyl m/p-methylfentanyl detections more than doubling from Q1 to Q2 2025 [4]. In contrast, some nitazene analogs like N-desethyl metonitazene have seen substantial decreases in detection rates over the same period [4]. The street drug supply is further complicated by the presence of non-opioid adulterants like xylazine and medetomidine, which are alpha-2 adrenergic receptor agonists approved only for veterinary use. These substances compound the risk of opioid use by introducing additional central nervous system depression and causing severe side effects like necrotic skin ulcers [4].
The Researcher's Toolkit: Essential Reagents and Materials
Successful identification and analysis of synthetic opioids require a suite of specialized reagents, reference materials, and instrumentation.
Table 3: Essential Research Reagents and Materials for Synthetic Opioid Analysis
| Reagent / Material | Function / Application | Example Specification / Note |
|---|---|---|
| Deuterated Internal Standards | Mass spectrometry quantification; corrects for matrix effects and ionization variability | e.g., 17 deuterated reference compounds (including fentanyl-d5) at 100 ng/mL in methanol [10] |
| HPLC-grade Solvents | Mobile phase preparation; sample reconstitution; minimizes background interference | Acetonitrile, Methanol, Water (with 0.1% Formic Acid) [10] |
| LC Columns | Chromatographic separation of complex mixtures | e.g., ACQUITY UPLC BEH C18, 2.1 x 150 mm, 1.8 µm particle size, 50°C [8] |
| Reference Standards | Compound identification and method calibration | Pure analytical standards for targeted analytes (e.g., fentanyl, nitazenes, novel opioids) |
| Paper-Spray Substrates | Sample substrate for paper-spray ionization | VeriSpray sample plates [10] |
| High-Resolution Mass Spectrometer | Accurate mass measurement; non-targeted screening | LC-QTOF systems (e.g., Waters Xevo G2-S) capable of ±5 ppm mass accuracy [8] |
| Tandem Mass Spectrometer | Sensitive and specific targeted quantification | LC-MS/MS systems (e.g., TSQ Fortis) operating in MRM mode [10] |
Diagram 2: Opioid Receptor Signaling Pathway
The synthetic opioid landscape is defined by continuous chemical evolution, presenting a persistent and shifting threat to global public health. The ongoing transition from established fentanyl analogs like fluoro fentanyl to newer entities such as methylfentanyl isomers and highly potent nitazenes necessitates equally adaptive and sophisticated research responses. Foundational research in this field must leverage advanced mass spectrometry techniques, including both targeted quantification and non-targeted screening approaches, to keep pace with the emergence of new substances. A comprehensive understanding of the chemical, analytical, and pharmacological dynamics of these compounds is paramount for developing effective public health interventions, clinical treatments, and regulatory strategies to mitigate the harms caused by these potent substances.
Overcoming Critical Challenges in NPS Identification and Response
Addressing the Legislative Lag and Chemical Structure Modification
The rapid emergence of novel psychoactive substances (NPS) presents a dual challenge: a persistent legislative lag in control mechanisms and the continuous chemical structure modification by manufacturers to circumvent existing laws. This whitepaper provides an in-depth technical analysis for researchers and drug development professionals, framing these issues within the broader context of foundational NPS identification research. It details the current legislative landscape, advanced analytical methodologies for structural elucidation, and proposes a unified framework to accelerate the identification-to-regulation pipeline, thereby mitigating public health risks associated with these evolving compounds.
Novel psychoactive substances (NPS), often termed "designer drugs," are a class of compounds designed to mimic the effects of controlled substances while avoiding legal restrictions [11]. Their proliferation is characterized by unprecedented speed and chemical diversity, directly exploiting the slow pace of traditional legislative scheduling processes. This legislative lag creates a dangerous window during which new, potentially harmful substances can circulate unregulated. The core of the problem lies in the ability of manufacturers to perform systematic chemical structure modification—creating analogs and derivatives—of existing controlled compounds. These modifications are often slight (e.g., altering a halogen substituent, adding or removing a methyl group, or modifying the core scaffold) but are sufficient to render the new substance legally distinct from its controlled predecessor. For researchers, this translates into a continuous race to identify, characterize, and assess the risk of new molecules faster than they can be introduced into the market.
The Legislative and Regulatory Landscape
Current Federal Framework and Its Gaps
The primary U.S. regulatory framework for controlling dangerous substances, the Controlled Substances Act (CSA), operates on a reactive model. A substance must be specifically identified and scheduled through a formal rulemaking process, which can take months or years. While the CSA includes "analog" provisions to cover structurally similar compounds with similar effects, their application can be complex and require legal as well as scientific proof.
Concurrently, the Toxic Substances Control Act (TSCA) is being used to address the industrial chemical aspect of NPS pre-cursors. A significant recent development is a final rule from December 2024, which makes PFAS (per- and polyfluoroalkyl substances) categorically ineligible for low volume exemptions (LVE) in TSCA's new chemical review process [12]. This demonstrates a regulatory shift towards using categorical approaches to manage structurally related families of chemicals, a strategy that could be instructive for NPS control. However, a resolution (H.J. Res. 76) has been introduced in Congress to disapprove this final rule, highlighting the ongoing political and regulatory tension surrounding chemical management [12].
Quantitative Analysis of Legislative Activity
The following table summarizes key federal and state legislative and regulatory actions from the first quarter of 2025, illustrating the multifaceted governmental response.
Table 1: Key PFAS/NPS-Related Legislative and Regulatory Actions (Q1 2025)
| Bill/Regulation | Type | Key Provision | Status (as of Q1 2025) |
|---|---|---|---|
| U.S. House Bill 705 [12] | Federal Legislation | Establishes a compensation fund for military firefighters exposed to PFAS. | Referred to Committee on Armed Services. |
| U.S. House Bill 1267 [12] | Federal Legislation | Exempts water utilities from CERCLA liability for PFOA/PFOS releases. | Referred to Energy & Commerce and Transportation & Infrastructure Committees. |
| U.S. H.J. Res. 76 [12] | Federal Disapproval Resolution | Seeks to nullify the EPA's Dec 2024 TSCA rule that makes PFAS ineligible for LVEs. | Referred to House Committee on Energy and Commerce. |
| CA Senate Bill 682 [12] | State Legislation | Phased ban on sale/distribution of covered products with intentionally added PFAS. | Hearing scheduled for April 2, 2025. |
| IL House Bill 1295 [12] | State Legislation | Bans specific consumer products with intentionally added PFAS, starting 2026; comprehensive ban by 2033. | Re-referred to Rules Committee (March 2025). |
| EPA TSCA SNURs [13] | Federal Regulation | Final Significant New Use Rules for certain chemical substances, requiring 90-day pre-manufacture notification. | Effective September 15 & 29, 2025. |
Analytical Methodologies for NPS Identification
Overcoming the NPS challenge requires robust, rapid, and precise analytical techniques for the identification of unknown compounds and the detection of known analogs in complex biological matrices.
Core Analytical Workflow
The general workflow for NPS identification and characterization involves sample preparation, screening, confirmation, and data analysis. The following diagram illustrates the logical relationships and pathways in this multi-step process.
Detailed Experimental Protocols
Protocol for Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) Analysis of Synthetic Cathinones
This protocol is adapted from a published method for determining 73 synthetic cathinones and related metabolites in urine [11].
1. Sample Preparation (Solid-Phase Extraction - SPE):
- Reagents: β-glucuronidase enzyme, phosphate buffer (pH 6.0), SPE cartridges (e.g., Oasis MCX mixed-mode cation-exchange), methanol, HPLC-grade water, 2% formic acid in water, 5% ammonium hydroxide in methanol.
- Procedure: A 1 mL aliquot of urine is incubated with β-glucuronidase in phosphate buffer at 37°C for 1 hour to hydrolyze metabolites. The sample is then centrifuged. The supernatant is loaded onto a pre-conditioned SPE cartridge. After washing with 2% formic acid and methanol, the analytes are eluted with 5% ammonium hydroxide in methanol. The eluent is evaporated to dryness under a gentle nitrogen stream and reconstituted in 100 µL of initial mobile phase for LC-MS/MS analysis.
2. Instrumental Analysis (LC-MS/MS):
- Chromatography: A C18 reverse-phase column (e.g., 2.1 x 100 mm, 1.8 µm) is used. The mobile phase consists of (A) 0.1% formic acid in water and (B) 0.1% formic acid in acetonitrile. A gradient elution is performed from 5% B to 95% B over 10-15 minutes. The flow rate is 0.3 mL/min, and the column temperature is maintained at 40°C.
- Mass Spectrometry: The mass spectrometer is operated in positive electrospray ionization (ESI+) mode with multiple reaction monitoring (MRM). The ion source parameters (gas flow, temperature, voltages) are optimized for the specific instrument. For each cathinone, two precursor ion → product ion transitions are monitored for unambiguous identification and quantification.
Protocol for High-Resolution Mass Spectrometry (HRMS) and Non-Targeted Screening
This protocol is critical for identifying entirely unknown or unexpected NPS.
1. Sample Preparation:
- Similar to the targeted approach, but broader-spectrum SPE or dilute-and-shoot methods can be employed to capture a wider range of chemical functionalities.
2. Instrumental Analysis (LC-HRMS):
- Chromatography: Uses a similar UHPLC system as above with a generic, wide-scope gradient to separate a diverse chemical space.
- Mass Spectrometry: A high-resolution mass spectrometer (e.g., Q-TOF or Orbitrap) is used. Data-Dependent Acquisition (DDA) is the standard mode: a full-scan MS1 spectrum (e.g., m/z 50-1000) is acquired at high resolution (>25,000), and the most intense ions from this scan are automatically selected for fragmentation to generate MS2 spectra. Data-Independent Acquisition (DIA) is an alternative where all ions within a predefined m/z window are fragmented, reducing the chance of missing low-abundance NPS.
3. Data Processing:
- Raw HRMS data is processed using software to deconvolute spectra, detect chromatographic peaks, and align features across samples.
- Tentative Identification: Achieved by querying the accurate mass of the precursor ion (from MS1) against chemical databases (e.g., UNODC NPS database, Cayman Chemical) and confirming with the experimental MS2 fragmentation pattern. In-silico fragmentation tools can aid in matching when no reference standard is available.
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key reagents, materials, and software solutions essential for conducting foundational NPS research.
Table 2: Key Research Reagent Solutions for NPS Identification
| Item/Category | Function & Application | Specific Example(s) |
|---|---|---|
| Certified Reference Standards | Essential for method development, calibration, and definitive identification via retention time and fragmentation pattern matching. | Synthetic cathinones (e.g., MDPV, Mephedrone), synthetic cannabinoids, phenethylamines. |
| Stable Isotope-Labeled Internal Standards (e.g., Deuterated, ^13^C) | Correct for matrix effects and analyte loss during sample preparation; crucial for accurate quantification. | Cannabinol-D3, Cocaine-D3, Amphetamine-D11. |
| Solid-Phase Extraction (SPE) Cartridges | Clean-up and pre-concentration of analytes from complex biological matrices, improving sensitivity and instrument longevity. | Mixed-mode (e.g., Oasis MCX for bases), C18 reverse-phase. |
| Liquid Chromatography Columns | Separation of complex mixtures of NPS and their metabolites prior to mass spectrometric detection. | C18 reverse-phase, HILIC (for polar compounds), fused-core particles for fast analysis. |
| HRMS Forensic Libraries & Databases | Software tools containing spectral data for known NPS; enable rapid tentative identification from HRMS data. | UNODC NPS Database, Cayman Chemical Mass Spectrometry Library, Thermo Fisher mzCloud. |
| Text Mining & NLP Tools (Large Language Model-Assisted) | Analyze vast volumes of scientific literature, online forum data, and legal documents to track emerging NPS trends and terminology [14]. | Custom scripts for "Large Language Model-assisted Text Mining (LATeM)" [14]. |
A Unified Framework for Mitigating Legislative Lag
To bridge the gap between NPS emergence and regulatory control, a proactive, integrated framework is required. This framework leverages advanced analytics and data sharing to inform the legislative process.
This framework emphasizes a continuous cycle of monitoring, identification, sharing, and action. The adoption of class-wide or analog-based scheduling, similar to the categorical approach seen with PFAS under TSCA, could significantly reduce the administrative burden of scheduling each individual NPS analog as it appears [13] [12]. Furthermore, the integration of large language model-assisted text mining (LATeM) can uncover emerging research trends and predict new NPS variants by analyzing scientific literature and open-source data, providing an early warning system for regulators and researchers alike [14].
The rapid proliferation of novel psychoactive substances (NPS) represents a significant challenge for forensic science, clinical toxicology, and public health. Within this evolving landscape, synthetic opioids, particularly fentanyl analogs, have emerged as a particularly dangerous class of compounds due to their high potencies and associated overdose risks [15] [16]. Among the most analytically challenging aspects of this crisis is the emergence of positional isomers—compounds sharing identical molecular formulas and mass-to-charge ratios but differing in the arrangement of atoms, which can lead to dramatic differences in pharmacological activity and toxicity [17].
Methylfentanyl analogs, which feature a methyl group at different positions on the core fentanyl structure, exemplify this analytical problem. These isobaric compounds produce nearly identical mass spectra under standard conditions, complicating their identification in forensic casework and toxicological analyses [17]. The accurate differentiation of these isomers is not merely an academic exercise; it is foundational research critical for understanding structure-activity relationships, tracking illicit manufacturing trends, and informing public health responses to the opioid epidemic [18] [19]. This technical guide examines the core challenges in distinguishing methylfentanyl positional isomers and details advanced analytical strategies to address this growing problem.
The Methylfentanyl Isomer Landscape
The term "methylfentanyl" encompasses multiple structural analogs of fentanyl where a methyl group is incorporated at different positions of the parent molecule. These positional changes create compounds with identical molecular weights but potentially different biological activities and legal statuses [17] [20].
Structural Classification and Nomenclature
The general fentanyl structure (N-phenyl-N-(1-phenethylpiperidin-4-yl)propionamide) provides five primary regions for methyl substitution, designated as R1 through R5 [17]:
- R1 (Aniline ring substitution): Includes ortho-, meta-, and para-methylfentanyl
- R2 (Amide moiety substitution): Includes butyryl and isobutyryl fentanyl
- R3 (Piperidine ring substitution): Includes trans-3-methyl fentanyl
- R4 (N-alkyl chain substitution)
- R5 (Monocyclic substituent)
Among these, the aromatic ring positional isomers (particularly ortho-methylfentanyl) present exceptional analytical challenges due to their nearly identical physicochemical properties [17] [20]. The recent identification of ortho-methylfentanyl in toxicological casework in late 2023 highlights the continuous emergence of these challenging analogs [20].
Pharmacological Significance of Isomeric Differences
The position of methyl substitution significantly influences the pharmacological profile of fentanyl analogs. In vitro studies indicate that ortho-methylfentanyl is an active mu opioid agonist with potency similar to or slightly less than fentanyl itself [20]. However, different substitution patterns can dramatically alter receptor binding affinity and functional activity.
For 3-methylfentanyl, the spatial orientation of the methyl group creates distinct cis and trans stereoisomers with different potencies, creating an additional layer of analytical complexity [21]. The accurate identification of these stereoisomers is crucial for toxicological interpretation, as they exhibit different biological activities despite identical elemental composition.
Table 1: Methyl-Substituted Fentanyl Analogs and Their Key Characteristics
| Analog Name | Substitution Position | Molecular Formula | Molecular Weight (g/mol) | Protonated Molecule [M+H]+ (m/z) |
|---|---|---|---|---|
| ortho-Methyl fentanyl | Aniline ring (ortho) | C23H30N2O | 350.50 | 351.2431 |
| 4-Methyl fentanyl | Piperidine ring | C23H30N2O | 350.50 | 351.2431 |
| 4′-Methyl fentanyl | Aniline ring (para) | C23H30N2O | 350.50 | 351.2431 |
| α-Methyl fentanyl | N-alkyl chain | C23H30N2O | 350.50 | 351.2431 |
| β-Methyl fentanyl | N-alkyl chain | C23H30N2O | 350.50 | 351.2431 |
| trans-3-Methyl fentanyl | Piperidine ring | C23H30N2O | 350.50 | 351.2431 |
| Butyryl fentanyl | Amide moiety | C23H30N2O | 350.50 | 351.2431 |
| Isobutyryl fentanyl | Amide moiety | C23H30N2O | 350.50 | 351.2431 |
Analytical Challenges in Isomer Differentiation
Limitations of Traditional Seized Drug Analysis
Traditional seized drug analysis methodologies face significant limitations when applied to fentanyl isomer differentiation. Colorimetric tests, while rapid and inexpensive, provide subjective information with limited selectivity and are not ideal for distinguishing fentanyl analogs [17]. Gas chromatography-electron ionization-mass spectrometry (GC-EI-MS), considered the gold standard for seized drug analysis, struggles with positional isomers due to nearly identical electron ionization mass spectra with only subtle differences in fragmentation patterns [17].
The "hard" ionization nature of EI often complicates molecular weight determination through the frequent absence of molecular ions, particularly problematic for isobaric compounds where the molecular ion provides critical information [17]. While chromatographic separation can potentially resolve some isomers, the similarity in physicochemical properties often results in co-elution or minimal retention time differences, insufficient for definitive identification alone.
Mass Spectrometric Limitations
Without collisional activation, all eight isobaric methyl-substituted fentanyl analogs included in a recent study were indistinguishable, producing only the protonated molecule at nominal m/z 351 [17]. While collision-induced dissociation (CID) can generate distinguishable product ion spectra for most analogs, the interpretation remains challenging and requires careful analysis of subtle differences in fragmentation patterns influenced by the methyl substitution location [17].
The situation is further complicated for 3-methylfentanyl, which exists as cis and trans stereoisomers with different potencies [21]. Their differentiation requires specialized analytical approaches capable of distinguishing stereochemistry, adding another dimension to an already complex identification challenge.
Advanced Analytical Strategies
Mass Spectrometry with Alternative Fragmentation Approaches
Direct Analysis in Real Time-Mass Spectrometry (DART-MS) has emerged as a powerful technique for rapid seized drug screening with minimal sample preparation [17]. Unlike EI, DART is a soft ionization technique that produces predominantly protonated molecules, enabling molecular weight determinations critical for isobaric compound analysis [17].
To overcome the limitation of insufficient fragmentation with soft ionization, several complementary approaches have been developed:
- In-source collision-induced dissociation (IS-CID): Applies collision energy within the source region to induce fragmentation without precursor ion selection
- All ions fragmentation (AIF): Fragments all ions simultaneously without precursor selection
- Tandem mass spectrometry (MS/MS): Provides targeted fragmentation of specific precursor ions
The combination of fragmentation data collected at multiple collision energies (low, medium, high) creates distinctive fragmentation signatures that can facilitate isomer differentiation [17].
The NIST/NIJ Data Interpretation Tool and ILSA Algorithm
The National Institute of Standards and Technology (NIST), in collaboration with the National Institute of Justice (NIJ), has developed specialized resources to address the challenges of interpreting complex DART-MS data for forensic applications [17]. The core component is the NIST/NIJ Data Interpretation Tool (DIT), which incorporates the inverted library search algorithm (ILSA) for mixture interpretation based on IS-CID mass spectra collected at multiple collision energies [17].
Recent research has demonstrated that optimized weighting of scoring metrics within ILSA significantly improves isomer differentiation capabilities. A 50:50 weighting of medium and high fragmentation mass spectra, combined with an optimized 0.80 reverse match factor threshold, produced the highest correct presumptive identifications and minimized false positives for methyl-substituted fentanyl analogs [17].
Orthogonal Analytical Techniques
While mass spectrometry provides powerful capabilities for isomer differentiation, orthogonal techniques can deliver additional confirmatory data:
- Liquid chromatography-high resolution mass spectrometry (LC-HRMS): Provides exceptional mass accuracy and resolution for distinguishing isobaric compounds, with the capability to separate cis and trans stereoisomers of 3-methylfentanyl [21]
- Gas chromatography-infrared spectroscopy (GC-IRD): Offers distinctive infrared spectra that can differentiate positional isomers based on subtle differences in vibrational modes [17]
- Gas chromatography-vacuum ultraviolet spectroscopy (GC-VUV): Provides characteristic absorption spectra that are highly specific to molecular structure [17]
Table 2: Performance Comparison of Analytical Techniques for Methylfentanyl Isomer Differentiation
| Analytical Technique | Key Strengths | Limitations | Suitable for Isomer Differentiation? |
|---|---|---|---|
| Colorimetric Tests | Rapid, inexpensive, simple | Low specificity, subjective | No |
| GC-EI-MS | Gold standard, good sensitivity | Similar fragmentation patterns, hard ionization | Limited |
| LC-MS/MS | High sensitivity, targeted | Requires precursor ion selection | Good (with HRMS) |
| DART-MS with AIF/IS-CID | Rapid, minimal prep, soft ionization | Complex spectra without separation | Good to Excellent (with optimized algorithms) |
| GC-IRD | Excellent structural specificity | Lower sensitivity, requires separation | Excellent |
| GC-VUV | Characteristic spectra | Emerging technique, requires separation | Good |
Experimental Protocols for Isomer Differentiation
DART-MS Analysis with All Ions Fragmentation
Instrumentation: DART ionization source coupled to a high-resolution mass spectrometer (e.g., JEOL AccuTOF) [17]
Sample Preparation:
- Prepare certified reference materials at appropriate concentrations in suitable solvents
- Utilize 12-Dip-it tips for direct sample introduction
- Implement minimal sample preparation to maintain rapid analysis times
Data Acquisition:
- Collect mass spectra at three different collision energy conditions: low (10 eV), medium (20 eV), and high (40 eV)
- Maintain consistent gas temperature and pressure conditions across analyses
- Acquire data in positive ion mode with mass resolution >6000
Data Interpretation:
- Process raw data using the NIST/NIJ Data Interpretation Tool
- Apply inverted library search algorithm with optimized 50:50 weighting for medium and high fragmentation spectra
- Use reverse match factor threshold of 0.80 for presumptive identification
LC-HRMS Method for 3-Methylfentanyl Stereoisomers
Chromatographic Conditions:
- Column: C18 reverse phase column (100 × 2.1 mm, 1.7 μm)
- Mobile Phase: Gradient of 0.1% formic acid in water and 0.1% formic acid in acetonitrile
- Flow Rate: 0.4 mL/min
- Column Temperature: 40°C
Mass Spectrometric Conditions:
- Ionization: Electrospray ionization (ESI) in positive mode
- Mass Analyzer: Time-of-flight (TOF) or Q-TOF with resolution >25,000
- Mass Range: 100-500 m/z
- Source Temperature: 150°C
Sample Preparation:
- Extract blood samples using solid phase extraction
- Reconstitute in mobile phase
- Utilize a linear dynamic range of 0.1-10 ng/mL for quantification [21]
Essential Research Reagents and Materials
The accurate identification and differentiation of methylfentanyl isomers requires access to specialized reference materials and analytical tools. The following table details essential research reagents for this field of study.
Table 3: Essential Research Reagent Solutions for Methylfentanyl Isomer Analysis
| Reagent/Material | Supplier Examples | Application/Function | Critical Specifications |
|---|---|---|---|
| Certified Reference Materials | Cayman Chemical, Cerilliant | Method development/validation, quantitative analysis | Purity >98%, concentration verification |
| ortho-Methylfentanyl Standard | Cayman Chemical | Identification of emerging isomer | Structural confirmation, spectral library building |
| ortho-Methyl 4-ANPP Standard | Cayman Chemical | Precursor/metabolite identification | Purity, stability |
| Solid Phase Extraction Cartridges | Various | Sample clean-up and concentration | Recovery efficiency >85% for fentanyl analogs |
| LC-MS Grade Solvents | Various | Mobile phase preparation | Low UV absorbance, high purity |
| DART Dip-It Tips | IonSense | Sample introduction for DART-MS | Consistency, minimal background |
| NIST DART-MS Forensics Database | NIST | Spectral matching and identification | Comprehensive coverage, regular updates |
| NIST/NIJ Data Interpretation Tool | NIST | Data processing and algorithm application | ILSA optimization, customizable thresholds |
The differentiation of methylfentanyl positional isomers represents a significant analytical challenge with direct implications for public health and forensic science. The continuous emergence of new analogs, such as the recent identification of ortho-methylfentanyl, underscores the dynamic nature of the NPS landscape and the need for advanced analytical strategies [20]. While traditional techniques like GC-EI-MS face limitations in distinguishing these isobaric compounds, the integration of DART-MS with all ions fragmentation and sophisticated data interpretation tools like the NIST/NIJ DIT with optimized ILSA algorithms provides a powerful approach to this problem [17]. The critical importance of this foundational research extends beyond analytical chemistry to toxicological interpretation, structure-activity relationship studies, and the development of evidence-based drug control policies. As the synthetic opioid market continues to evolve, ongoing refinement of these methodologies will be essential for maintaining effective responses to this persistent public health crisis.
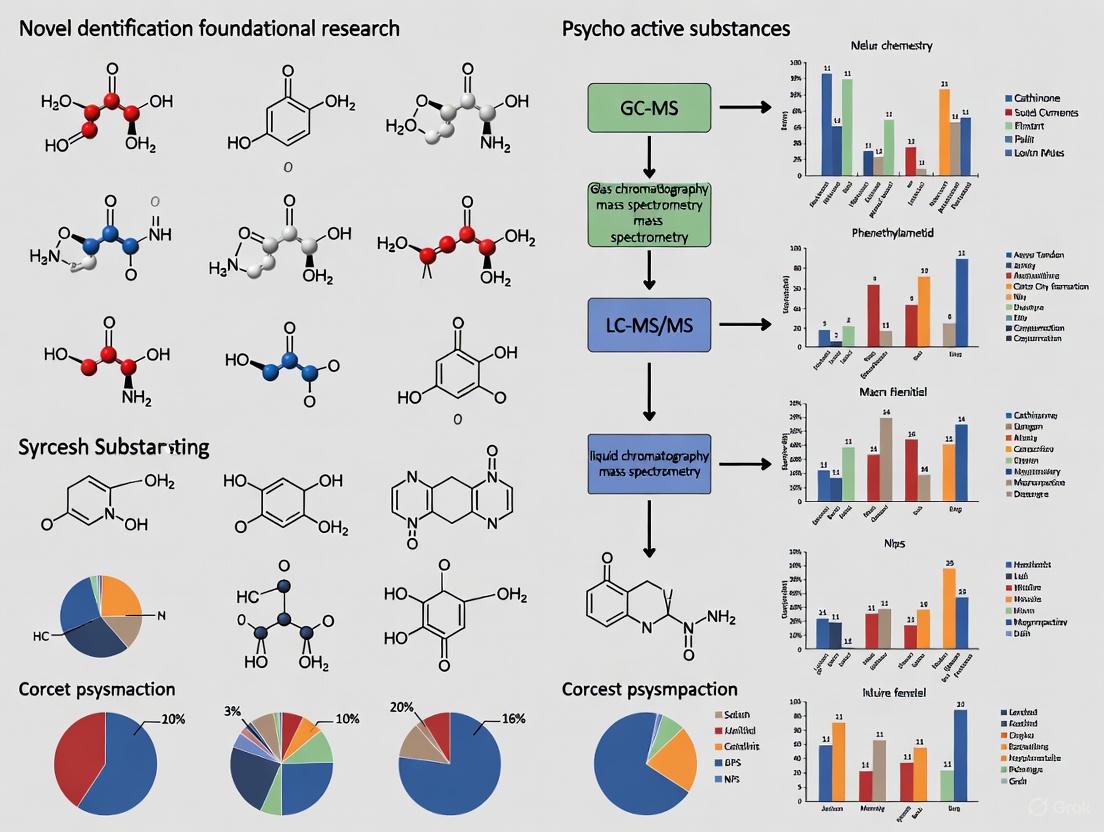
Within foundational research on Novel Psychoactive Substances (NPS), the interpretation of detection data presents a significant methodological challenge. A critical, often underappreciated, confounding variable is the impact of test order rates—the frequency with which specific NPS classes are requested by clinicians—on the observed prevalence of these substances. This whitepaper delineates the systematic biases introduced by heterogeneous ordering practices and provides researchers with explicit protocols to distinguish true epidemiological trends from analytical artifacts. Failure to account for these factors can severely distort the perceived threat level of emerging drugs, misdirect public health resources, and compromise the validity of scientific conclusions.
The Foundational Challenge in NPS Research
The global NPS market is characterized by its dynamism and diversity, with continuous emergence of new substances designed to circumvent controlled substance legislation [22]. This fluidity complicates all phases of NPS research, from analytical detection to prevalence estimation. A primary obstacle in obtaining accurate prevalence data is the analytical identification of NPS in biological samples, which is particularly challenging when samples are limited or when a drug is rapidly and extensively metabolized [22]. This inherent analytical difficulty contributes to a pervasive lack of information on the real diffusion of NPS across different populations and geographic regions.
In this complex landscape, test order rates—the frequency with which specific NPS classes are requested by healthcare providers—introduce a substantial non-random bias into surveillance data. These rates are not uniform; they vary significantly based on clinician awareness, regional concerns, testing panel design, and perceived patient risk profiles. Consequently, raw detection counts (positivity) are not equivalent to true prevalence. A substance may appear "prevalent" simply because it is tested for more frequently, while a truly emerging threat might be missed due to low testing rates. Recognizing this, leading laboratories like Aegis Sciences Corporation explicitly caution that "order rates may impact NPS detection and observed prevalence both within a class and between classes" [4] [23]. The challenge, therefore, is to disentangle the signal of true drug use from the noise of testing bias.
Quantitative Evidence of Order Rate Variance
Empirical data from large-scale testing laboratories provides unequivocal evidence of significant disparities in NPS class ordering patterns. This variance directly impacts the observed prevalence of different NPS categories, making quantitative adjustment a necessity for accurate interpretation.
The table below summarizes NPS class order rates as a percentage of samples for which any NPS class was ordered, based on 2024 and Q1-Q2 2025 data [4] [23]. This reveals a clear hierarchy in testing focus.
Table 1: NPS Class Order Rates (as % of samples with any NPS class ordered)
| NPS Class | 2024 Order Rate (%) | 2025 Order Rate (Q1 & Q2 Avg, %) |
|---|---|---|
| Designer Opioids | ~95% | ~95% |
| Designer Benzodiazepines | ~95% of Designer Opioids | ~90% |
| NPS-Other | ~80% of Designer Opioids | ~76% |
| Synthetic Stimulants | ~68% | ~62% |
| Synthetic Cannabinoids | ~67% | ~62% |
| Hallucinogens/Dissociatives | ~42% of Designer Opioids | ~40% |
This data demonstrates a consistent and pronounced bias. Designer opioids and benzodiazepines are the primary focus of surveillance efforts, ordered in the vast majority of cases. In contrast, the hallucinogens/dissociatives class is consistently the least-ordered, with rates less than half that of the top classes. This discrepancy means that a detection of a hallucinogen, while statistically rarer in the dataset, may be more significant than it initially appears. The stability of these rates between 2024 and 2025 indicates a persistent, systemic bias in testing priorities rather than a temporary fluctuation.
Impact on Positivity and Prevalence Observations
The direct consequence of variable order rates is that raw positivity counts (the number of times a substance is detected) do not reflect true prevalence. A class with high order volume will inevitably accumulate more total detections, potentially making it appear more "common" than a class with lower order volume but potentially higher rates of use among those tested.
To mitigate this confounder, data must be normalized. The recommended metric is positivity rate (also called percent positivity), calculated as [4] [23]: Positivity Rate = (Number of Positives for a Substance / Number of Tests Ordered for that Substance's Class) * 100
This adjustment is critical for valid intra-class and inter-class comparisons. For instance, Aegis Sciences Corporation notes that the hallucinogens/dissociatives class has the lowest positivity rates but also has the lowest order rates, implying that the raw data likely underrepresents its true presence in the user population [23]. Presenting data as a proportion of detections within a class, rather than as absolute counts, helps correct for the volume of testing and provides a clearer picture of trends [4].
Experimental Protocols for Bias-Minimized NPS Detection Analysis
To ensure the validity of NPS research findings, the following experimental protocols provide a structured approach to mitigate the confounding effects of order rate bias.
Protocol A: Longitudinal Analysis of Pooled Urban Urine Samples
This protocol, adapted from a long-term study in central London, offers a population-level perspective that is independent of clinical ordering patterns [24].
- Objective: To determine long-term trends in the use of NPS within a defined geographic population without the bias of individual test orders.
- Sample Collection: Collect pooled urine samples on a monthly basis from portable, stand-alone public urinals deployed in a city center over a multi-year period (e.g., 12 urinals over 12-hour periods from 1800–0600) [24].
- Sample Processing: Pool samples from all urinals for each calendar month to create a composite monthly sample representing community-wide drug use.
- Instrumental Analysis: Analyze composite samples using high-performance liquid chromatography interfaced with a high-resolution accurate mass spectrometer (HPLC-HRAM). Process data against a comprehensive database containing thousands of drugs and metabolites, including NPS [24].
- Data Analysis & Normalization: Identify all detected NPS. Report detection frequency as the number of months a substance was detected out of the total months in the study period. Trends are analyzed by tracking the appearance and disappearance of substances over time, independent of clinical test volume.
Protocol B: Normalization of Clinical Toxicology Data
This protocol is designed for the analysis of data generated from clinical patient testing, where order rate bias is inherent.
- Objective: To accurately assess the prevalence and trends of NPS within a clinical population by normalizing detection data for variable test order rates.
- Data Sourcing: Utilize large-scale, anonymized datasets from clinical toxicology laboratories that include both test orders and resultant findings. Data should be segmented by NPS class (e.g., Designer Opioids, NPS-Other) and over defined time periods (e.g., quarterly) [4] [23].
- Data Processing:
- For each time period, calculate the total number of tests ordered for each NPS class.
- For each specific NPS analyte (e.g., xylazine, fluorofentanyl), count the number of confirmed detections within its respective class.
- Calculation of Normalized Metrics:
- Class-Level Positivity Rate: For each NPS class and time period, calculate
(Total Detections in Class / Total Tests for Class) * 100. - Analyte-Level Proportion: For each specific analyte, calculate its proportion within its class as
(Number of Analyte Detections / Total Detections in its Class) * 100[4].
- Class-Level Positivity Rate: For each NPS class and time period, calculate
- Trend Analysis: Evaluate temporal trends by tracking the percent change in the normalized
Analyte-Level Proportionfrom one period to the next (e.g., Q1 to Q2). This reveals whether a substance is becoming more or less common relative to other substances in its class, controlling for changes in class order volume [4].
The following workflow diagram illustrates the core decision-making process for selecting and applying the appropriate methodological approach.
The Scientist's Toolkit: Research Reagent Solutions
Successfully implementing the aforementioned protocols requires a suite of specialized reagents and analytical standards.
Table 2: Essential Research Reagents for NPS Detection and Analysis
| Item | Function & Application |
|---|---|
| Certified Reference Materials (CRMs) | High-purity chemical standards for definitive identification and quantification of NPS and their metabolites via mass spectrometry; essential for method development and validation [22]. |
| Stable Isotope-Labeled Internal Standards | Isotopically labeled analogs (e.g., ¹³C, ²H) of target NPS; added to samples to correct for matrix effects, ionization efficiency, and instrument variability, ensuring quantitative accuracy in HRAM analysis [24]. |
| Liquid Chromatography (LC) Solvents & Buffers | Ultra-purity mobile phases and volatile buffers (e.g., ammonium formate) for optimal separation of NPS and metabolites in complex biological matrices prior to mass spectrometric detection. |
| Sample Preparation Consumables | Solid-phase extraction (SPE) cartridges, filtration units, and enzymatic hydrolysis kits (e.g., β-glucuronidase) for efficient cleanup, pre-concentration, and deconjugation of metabolites from urine and other biological samples. |
| Comprehensive NPS Metabolite Database | A curated, expandable digital library containing high-resolution mass spectra and fragmentation patterns for thousands of known NPS and predicted metabolites; the core for automated data processing and unknown identification [24]. |
Case Study: Applying Normalization to 'NPS-Other' Class Data
The "NPS-Other" category, which includes adulterants like xylazine and medetomidine, provides a clear illustration of the power of normalized analysis. While xylazine remained the most detected substance in this class in Q1 and Q2 2025, its proportion within the class decreased in Q2, primarily due to a -40% change in its metabolite's proportion [4]. This trend would be masked by looking at raw detection counts alone. Conversely, during the same period, the proportion of medetomidine and its metabolite increased by 34% and 29%, respectively, signaling a real and rapid emergence that might otherwise be underestimated if only overall class positivity was considered [4]. This demonstrates how proportional analysis reveals the dynamic competition between substances within a class.
In foundational NPS research, a failure to account for the confounding effect of test order rates renders any conclusion regarding prevalence and trends fundamentally unreliable. Raw detection counts are not a proxy for true prevalence. Researchers must systematically implement methodological safeguards, primarily through the normalization of data into positivity rates and analyte-specific proportions within classes. The experimental protocols and analytical frameworks detailed in this whitepaper provide a path toward more rigorous, unbiased, and actionable intelligence on the evolving threat of novel psychoactive substances, ensuring that public health and safety resources are allocated based on an accurate assessment of risk.
Evaluating NPS Detection Systems and Comparative Threat Assessment
The rapid global proliferation of Novel Psychoactive Substances (NPS) represents a critical challenge to public health systems, forensic science, and clinical practice. These substances, designed to mimic the effects of traditional illicit drugs while circumventing legal controls, contribute significantly to the burden of overdose deaths and adverse health events [11]. The dynamic nature of the NPS market, characterized by continuous chemical modifications, creates a moving target for detection, surveillance, and clinical intervention. Understanding the precise impact of NPS requires a multidisciplinary approach integrating forensic toxicology, epidemiology, and analytical chemistry to develop effective countermeasures against this evolving threat.
This technical assessment examines the current landscape of NPS within the broader context of foundational research for their identification and characterization. It synthesizes the latest surveillance data on overdose mortality, details the analytical methodologies enabling NPS detection, and explores the pharmacological mechanisms driving their toxicity. The objective is to provide researchers and public health professionals with a comprehensive resource for understanding and mitigating the public health impact of these substances through evidence-based approaches.
Current Landscape of Overdose Mortality
National Overdose Surveillance Data
Tracking overdose deaths provides the most definitive metric of the public health impact of NPS and other substances. According to the Centers for Disease Control and Prevention (CDC), provisional drug overdose death counts are a critical surveillance tool, though subject to reporting delays that can cause underestimates in recent data [25]. These data are vital for identifying emerging trends and evaluating public health interventions.
Table 1: CDC Provisional Drug Overdose Death Data Overview
| Data Aspect | Specification |
|---|---|
| Reporting Timeline | Estimates provided within 4 months of death date (improved from 6 months in 2022) |
| Data Coverage | 50 states and District of Columbia; Puerto Rico listed separately |
| Key Metric | 12-month ending provisional counts to account for seasonality |
| Data Limitations | Incomplete reporting; causes of death may be pending investigation |
| Adjustment Methods | Predicted counts generated to address reporting delays |
The CDC's surveillance system notes that some states are experiencing longer-than-usual delays in reporting, which should be considered when comparing recent data to previous periods [25]. This is particularly relevant for NPS-related deaths, which often require lengthy investigation including comprehensive toxicology testing. The provisional nature of this data necessitates careful interpretation, as initial counts often represent underestimates relative to final figures.
Emerging Threats in the Illicit Drug Supply
Recent data from public health and forensic toxicology sources reveal several concerning trends in the illicit drug supply that contribute to overdose risk:
Proliferation of Synthetic Opioids: Beyond fentanyl, newer synthetic opioids including nitazene analogs and fentanyl analogs (fentalogs) continue to emerge. According to Aegis Laboratories' 2025 data, fluoro fentanyl and its metabolites represented approximately 59% of designer opioids detected in Q1 2025 [4]. These compounds often exhibit potencies equal to or greater than fentanyl, significantly increasing overdose risk.
Emergence of Non-Opioid Adulterants: Xylazine, an alpha-2 adrenergic receptor agonist approved only for veterinary use, has become the most prevalent NPS detected in some testing populations, surpassing even fentanyl analogs in prevalence [4]. Its presence complicates overdose management as it does not respond to naloxone and is associated with severe necrotic skin ulcers.
Rapidly Evolving Drug Market: The NPS market continues to evolve with new substances emerging rapidly. Recent data shows significant increases in detections of medetomidine (34% increase from Q1 to Q2 2025) and tianeptine (36-44% increase), indicating their expanding presence in the illicit drug supply [4].
Table 2: Select Emerging NPS and Their Public Health Impact (Q1-Q2 2025)
| NPS Category | Example Compounds | Q1-Q2 2025 Trend | Primary Health Concerns |
|---|---|---|---|
| Designer Opioids | Fluoro fentanyl, Ortho-methylfentanyl, Nitazene analogs | Mixed trends by specific compound | Extreme potency, respiratory depression |
| Veterinary Sedatives | Xylazine, Medetomidine | Decreasing for xylazine; Increasing for medetomidine | Naloxone-resistant sedation, tissue damage |
| Atypical Substances | Tianeptine, Phenibut, BTMPS | Increasing for tianeptine and phenibut | Opioid-like effects, GABAergic activity, unknown toxicity |
The combination of multiple substance classes in the illicit drug supply creates a synergistic toxicity that complicates clinical management of overdoses. Particularly concerning is the frequent detection of NPS benzodiazepines in combination with opioids, which markedly increases the risk of fatal respiratory depression [19].
Analytical Methodologies for NPS Identification
Advanced Screening Techniques
The identification of NPS in biological specimens and seized materials requires sophisticated analytical approaches capable of detecting novel chemical structures at low concentrations. Several complementary techniques form the foundation of modern NPS screening programs.
Liquid Chromatography-High Resolution Mass Spectrometry (LC-HRMS) has emerged as a cornerstone technology for comprehensive NPS screening. Its application in a Swedish healthcare region enabled the retrospective identification of 58 different NPS in oral fluid samples from 201 patients by re-analyzing stored data as new compounds were identified [26]. This approach demonstrates the value of data banking for retrospective surveillance as new threats emerge.
Benchtop Nuclear Magnetic Resonance (NMR) spectroscopy represents an advancing front in NPS identification. Recent research has demonstrated the development of an automated database containing 1H NMR spectra for 360 NPS reference compounds, including 107 synthetic cannabinoids, 89 synthetic cathinones, 46 phenethylamines, and 38 fentanyl analogues [27]. The methodology employs multiple matching algorithms (Purity Peak Search, Cosine Similarity, and Pearson Similarity) to identify unknown compounds through database comparison, with the Purity Peak Search algorithm demonstrating superior performance with a 92% success rate for correct identification as the top hit [27].
Experimental Protocol: Benchtop NMR Screening for NPS
The following detailed protocol outlines the automated benchtop NMR approach for NPS screening as described by Liu et al. (2025) [27]:
Sample Preparation:
- Prepare standard solutions of NPS reference compounds at 10 mg/mL in DMSO-d6 containing 0.05% TMS as internal standard.
- Transfer 600 μL of each standard solution to 5 mm NMR tubes for analysis.
- For unknown samples, use the same preparation method with appropriate safety precautions.
Instrumental Parameters:
- Utilize an 80 MHz benchtop NMR spectrometer equipped with a permanent magnet.
- Set acquisition parameters: spectral width of 16 ppm, 32 scans, relaxation delay of 10 seconds, acquisition time of 4 seconds, and temperature maintenance at 30°C.
- Employ the standard single-pulse sequence with pre-saturation during the relaxation delay for solvent signal suppression.
Spectral Processing and Database Construction:
- Process all spectra with baseline correction, phase correction, and chemical shift Referencing to TMS at 0 ppm.
- Implement automated peak picking with sensitivity set to 1.0% of the largest peak and minimum peak area of 0.2%.
- Construct the database using MestReNova software, incorporating chemical structures, molecular weights, and chemical formulas for all 360 reference compounds.
- Apply chemical shift correction and remove solvent peaks to optimize search accuracy.
Database Search and Compound Identification:
- Process unknown samples using the same spectral processing parameters.
- Execute simultaneous searches using three algorithms: Purity Peak Search, Cosine Similarity, and Pearson Similarity.
- Set match threshold to 70% for positive identification.
- For ambiguous results, employ secondary confirmation with LC-MS/MS when available.
This protocol enables rapid screening of unknown substances with minimal sample preparation, making it particularly valuable for early warning systems and forensic laboratories.
Multidisciplinary Workflow for NPS Surveillance and Response
Effective monitoring of the public health impact of NPS requires coordination across multiple disciplines and sectors, from initial detection to public health response.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for NPS Identification and Characterization
| Reagent/Resource | Function and Application | Specific Examples |
|---|---|---|
| NPS Reference Standards | Essential for method development, calibration, and compound identification; purity must be certified | 360 compounds including synthetic cannabinoids, cathinones, fentanyl analogs [27] |
| Deuterated Solvents | Required for NMR spectroscopy; enables lock signal and minimizes solvent interference | DMSO-d6 with 0.05% TMS for chemical shift referencing [27] |
| LC-HRMS Systems | High-sensitivity detection and identification of NPS and metabolites in complex matrices | Systems capable of retrospective data re-interrogation [26] |
| Benchtop NMR Spectrometers | Structural elucidation of unknown compounds; increasingly accessible to non-specialist labs | 80 MHz permanent magnet systems with automated database matching [27] |
| Certified Quality Controls | Quality assurance for analytical methods; monitors method performance over time | Positive and negative controls for each analytical batch |
Discussion and Public Health Implications
The evolving landscape of NPS continues to present significant challenges for public health surveillance, clinical management, and forensic analysis. The interdisciplinary approach outlined in this assessment—combining advanced analytical techniques with robust epidemiological monitoring—provides the foundation for an effective response to this dynamic threat.
The data presented reveals several critical trends. First, the increasing complexity of the illicit drug supply, with multiple NPS classes frequently detected in combination, creates a toxicological landscape where polysubstance use is the norm rather than the exception. This complexity necessitates comprehensive testing approaches that can detect a wide spectrum of substances simultaneously. Second, the rapid turnover of NPS requires equally agile surveillance systems capable of identifying new threats as they emerge. Programs like NPS Discovery demonstrate the value of early warning systems that integrate data from public health and public safety sources [28].
From a clinical perspective, the rising prevalence of non-opioid adulterants like xylazine and medetomidine presents particular challenges. These substances do not respond to standard opioid reversal agents and can produce complex toxidromes that complicate diagnosis and treatment. Furthermore, the frequent co-occurrence of NPS benzodiazepines with opioids creates a heightened risk of respiratory depression that may be refractory to naloxone administration alone [19] [4].
Future directions in NPS research should focus on several key areas: (1) enhancing the speed and comprehensiveness of analytical methods to keep pace with emerging compounds; (2) developing point-of-care testing technologies that can provide rapid information in clinical settings; and (3) strengthening data sharing networks between public health, forensic, and clinical entities to facilitate early detection of new threats. Additionally, more research is needed to elucidate the pharmacology and toxicology of emerging NPS to inform the development of targeted antidotes and treatment protocols.
The public health impact of NPS, as measured through overdose deaths and adverse events, remains substantial despite ongoing efforts to curb their proliferation. The dynamic nature of these substances demands equally dynamic approaches to their identification, surveillance, and clinical management. Foundational research into NPS identification, particularly through advanced analytical techniques like LC-HRMS and benchtop NMR, provides the critical infrastructure necessary for monitoring this evolving threat.
As the NPS landscape continues to shift, with emerging threats including nitazene-type opioids, non-opioid adulterants, and novel benzodiazepines, the importance of robust, multidisciplinary surveillance systems cannot be overstated. Continued investment in analytical capabilities, reference materials, and data sharing networks is essential for mitigating the public health impact of these substances. Furthermore, the translation of analytical findings into actionable clinical guidance remains a critical pathway for reducing the mortality and morbidity associated with NPS use. Through coordinated efforts across public health, forensic, and clinical domains, it is possible to develop effective countermeasures against this persistent and evolving challenge.
Comparative Analysis of NPS Positivity in Forensic Casework
The rapid global emergence of Novel Psychoactive Substances (NPS) presents a critical challenge for forensic toxicology, public health, and drug policy. Defined by the United Nations Office on Drugs and Crime as "new narcotic or psychotropic drugs, in pure form or in preparation, not controlled by international conventions but which may pose a public health threat comparable to that presented by scheduled substances," NPS are synthetically designed to mimic effects of traditional illicit drugs while circumventing regulatory controls [29] [30]. The dynamic NPS market, characterized by constantly evolving chemical structures, creates substantial analytical challenges for forensic casework, requiring continuous methodological advancements to accurately monitor prevalence and positivity rates.
This technical guide provides a comprehensive analysis of NPS positivity trends in forensic samples, detailing current analytical frameworks and emerging technologies essential for foundational NPS research. For researchers and drug development professionals, understanding the evolving landscape of NPS detection is crucial for developing effective public health responses, legislative controls, and harm reduction strategies. We present quantitative data on NPS class prevalence, detailed experimental protocols for identification and confirmation, and visualization of complex analytical workflows to support forensic toxicology practice and policy development.
Current Landscape of NPS Positivity
Quantitative Analysis of NPS Class Prevalence
Forensic toxicology data from 2025 reveals distinct patterns in NPS class distribution across casework samples. Analysis of order rates and detection frequencies provides crucial insights into the relative prevalence of different NPS categories and informs resource allocation for laboratory testing protocols.
Table 1: 2025 Mid-Year NPS Order Rates by Class (as % of samples with any NPS class ordered) [31]
| NPS Class | Q1 2025 Order Rate | Q2 2025 Order Rate | Trend |
|---|---|---|---|
| Designer Opioids | ~95% | ~95% | → Stable |
| Designer Benzodiazepines | ~90% | ~90% | → Stable |
| NPS-Other | ~76% | ~76% | → Stable |
| Synthetic Cannabinoids | ~63% | ~61% | → Slight decrease |
| Synthetic Stimulants | ~63% | ~62% | → Slight decrease |
| Hallucinogens/Dissociatives | ~40% | ~40% | → Stable |
Order rates provide only partial insight into the NPS landscape; detection frequencies reveal actual substance prevalence. The "NPS-Other" category requires particular attention as it contains emerging adulterants that don't fit traditional classifications, including veterinary medications and industrial chemicals repurposed for human consumption.
Table 2: NPS-Other Compound Detection Trends in Forensic Casework (2025) [31]
| Compound | Category | Q1 2025 Proportion | Q2 2025 Proportion | % Change Q1-Q2 | Total Detections (H1 2025) |
|---|---|---|---|---|---|
| Xylazine & Metabolites | Veterinary sedative | ~70% | Decreased | -40% (metabolite) | Highest |
| Medetomidine & Metabolites | Veterinary sedative | Second highest | Increased | +29-34% | High |
| Tianeptine (MC5 metabolite) | Atypical antidepressant | ~Equal prevalence | Increased | +44% | 465 |
| Phenibut | Synthetic GABA analog | ~Equal prevalence | Increased | +88% | 505 |
| BTMPS | Industrial chemical | ~Equal prevalence | Decreased | -6% | Emerging |
| Phenylbutazone | Anti-inflammatory (withdrawn) | Minimal | Increased | Substantial (but low n) | Low |
| Etomidate | Human sedative (new addition) | Not tested | Detected | New to testing | 3 (June only) |
Emerging Trends in Specific NPS Classes
Designer Opioids
The designer opioid market continues to evolve rapidly, with fluoro-fentanyl and related compounds (despropionyl fluorofentanyl and fluoro norfentanyl) representing approximately 59% of designer opioids detected in Q1 2025 [31]. While fluoro-fentanyl was previously the most frequently detected NPS at Aegis laboratories, it was overtaken by xylazine in 2023 and has remained the second most prevalent NPS detected across all classes since. The ortho-methylfentanyl isomer has emerged as a significant concern, with the Center for Forensic Science Research and Education (CFSRE) issuing a public alert about its proliferation across North America in late 2024 [31].
Notable trends within the designer opioid class include substantial increases in despropionyl m/p-Methylfentanyl (+109%), norcarfentanil (+121%), and acryl fentanyl (+164%) from Q1 to Q2 2025. Conversely, significant decreases were observed in valeryl fentanyl carboxy metabolite and various nitazene analogs including N-desethyl metonitazene (-77%) [31]. This volatility in specific compound prevalence underscores the rapidly adapting illicit market as manufacturers substitute controlled substances with structurally similar but non-regulated analogs.
NPS Discovery Program Data
Broader surveillance efforts through the NPS Discovery program at the Center for Forensic Science Research and Education (CFSRE) provide additional context for forensic casework trends. Since 2018, the program has reported 174 newly discovered NPS in the United States, with NPS opioids remaining the largest subclass [32]. In 2024 alone, the program reported discovery of 20 new NPS and detected 103 different NPS in forensic samples, with more than 5,200 total NPS detections within examined sample populations [32]. This represents a portion of nearly 20,000 total NPS detections since the program launched in 2018, highlighting the substantial and growing challenge of NPS monitoring in forensic casework.
Analytical Methodologies for NPS Detection
Sample Selection and Preparation Protocols
The choice of biological matrix significantly influences detection capabilities in NPS analysis. Each matrix offers distinct advantages and limitations for forensic toxicology applications.
Table 3: Biological Matrices for NPS Analysis in Forensic Casework [29] [33]
| Matrix | Detection Window | Advantages | Limitations | Primary Applications |
|---|---|---|---|---|
| Blood | Hours | Direct correlation with impairment; difficult to adulterate | Invasive collection; lower analyte concentrations | Impairment assessment; acute intoxication |
| Urine | Days to weeks | Non-invasive; higher analyte concentrations | Possible manipulation; mainly metabolites present | Consumption monitoring; workplace testing |
| Oral Fluid | Hours to 2 days | Non-invasive; correlates with blood concentrations | Small volume; potential contamination | Roadside testing; recent use assessment |
| Hair | Months to years | Longest detection window; historical profile | External contamination concerns; complex preparation | Historical use patterns; abstinence monitoring |
Sample preparation protocols vary by matrix but generally aim to extract analytes while removing interfering compounds. For blood samples, protein precipitation, liquid-liquid extraction (LLE), salting-out assisted LLE, solid phase extraction (SPE), or microwave assisted extraction may be employed [29]. Urine samples typically undergo SPE or LLE, though dilute-and-shoot methods have also been reported [29]. Hair analysis requires washing steps to remove external contamination, followed by pulverization and extraction using organic solvents such as methanol, often with incubation at elevated temperatures (e.g., 55°C for 15 hours) [33].
Analytical Detection Techniques
The analytical approach for NPS detection generally consists of two stages: preliminary screening followed by confirmatory testing [29]. This sequential methodology efficiently allocates limited confirmatory resources to presumptively positive samples.
Preliminary Screening Methods
Immunoassays, while rapid and cost-effective, face significant limitations in NPS detection [29]. These tests require antibodies specifically binding to target structures, development of which is complicated by the rapid turnover of NPS on the drug market. Some activity-based assays that detect receptor activation rather than specific structures have been explored, but these may produce false positives from classical recreational drugs activating the same receptors [29].
Confirmatory Methods
Liquid chromatography and gas chromatography coupled with mass spectrometry (LC-MS/MS, GC-MS) represent the gold standard for NPS confirmation in forensic casework [29] [34]. These techniques provide the sensitivity, specificity, and flexibility required to identify both known and emerging NPS in complex biological matrices.
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) has become the preferred method for NPS analysis, with QTRAP instruments enabling high-quality MRM quantitation with simultaneous identification through enhanced product ion (EPI) MS/MS confirmation scans in a single injection [34]. The full scan MS/MS spectra obtained can be matched against spectral libraries to improve identification confidence, significantly reducing false negative and false positive rates [34].
High-resolution mass spectrometry (HRMS) platforms, including QTOF and Orbitrap systems, provide additional advantages for NPS analysis through accurate mass measurements [30] [34]. This capability is particularly valuable for creating digital records of samples that can be retrospectively re-interrogated as new compounds emerge [34]. The acquisition of accurate mass data and analyte-specific MS/MS fragment spectra enables both targeted quantification and untargeted screening approaches essential for comprehensive NPS monitoring.
Advanced Technical Workflows
Non-Targeted Screening Using HRMS
The rapid introduction of new NPS to the illicit drug market has necessitated a shift from traditional targeted methods to non-targeted screening approaches using high-resolution mass spectrometry (HRMS). This methodology is particularly valuable for detecting previously uncharacterized NPS and their metabolites, which may be absent from conventional mass spectral libraries [30].
Non-targeted HRMS workflows typically involve data-independent acquisition (DIA) techniques such as SWATH, which fragment all ions within predetermined mass isolation windows across the full mass range [34]. The resulting comprehensive dataset contains fragment ion information for all detectable compounds, creating a permanent digital record that can be retrospectively mined as new NPS are identified. Advanced data processing techniques, including diagnostic fragment ion analysis and neutral loss screening, enable the identification of structural analogs based on characteristic fragmentation patterns even without reference standards [30].
Artificial Intelligence in NPS Analysis
Artificial intelligence (AI) and machine learning (ML) are increasingly transforming NPS analytical toxicology by enhancing data processing capabilities and facilitating the identification of emerging substances [35]. These technologies are particularly valuable for handling the complex, high-volume datasets generated by non-targeted HRMS approaches.
The most frequent applications of AI in NPS analysis include compound identification, molecular structure prediction, and retention time prediction [35]. Machine learning models can recognize patterns in mass spectral data that correlate with specific structural features, enabling more accurate identification of novel analogs without reference standards. AI-powered tools can also predict pharmacological activity and toxicity based on chemical structure, providing valuable insights for risk assessment of newly identified NPS [35].
Research Reagents and Essential Materials
Successful NPS analysis requires specialized reagents, reference materials, and instrumentation tailored to the unique challenges of novel substance identification.
Table 4: Essential Research Reagents and Instrumentation for NPS Analysis [29] [30] [33]
| Category | Specific Examples | Function/Application | Technical Considerations |
|---|---|---|---|
| Chromatography Columns | C18 columns (e.g., 2.1 × 150 mm, 3.5 μm; 100 mm × 2.1 mm, 1.8 μm) | Compound separation prior to MS detection | Column chemistry, particle size, and dimensions affect resolution |
| Mobile Phase Additives | Ammonium formate, formic acid, ammonium acetate | Improve ionization efficiency and chromatographic separation | Concentration and pH impact signal intensity and separation |
| Extraction Materials | SPE cartridges (C18, mixed-mode), LLE solvents (diethyl ether, ethyl acetate, chloroform) | Sample clean-up and analyte concentration | Selection depends on analyte physicochemical properties |
| Mass Spectrometry Systems | QTRAP, QTOF, Orbitrap systems (e.g., ZenoTOF 7600, X500R QTOF) | Compound identification and quantification | Balance of sensitivity, resolution, and scan speed requirements |
| Reference Standards | Certified NPS and metabolite standards | Method development/validation and compound identification | Limited availability for newest NPS; substantial cost |
| Data Analysis Software | SCIEX OS, vendor-neutral platforms | Data acquisition, processing, and library matching | Compatibility with various instrument platforms and data formats |
The comparative analysis of NPS positivity in forensic casework reveals a rapidly evolving landscape characterized by the continuous introduction of novel substances and shifting prevalence of established compounds. The dominance of designer opioids and emerging adulterants like xylazine and medetomidine highlights critical public health concerns requiring ongoing surveillance. Effective monitoring of this dynamic market necessitates sophisticated analytical approaches, particularly non-targeted HRMS workflows complemented by emerging AI-powered identification tools.
For researchers and forensic professionals, maintaining current NPS detection capabilities requires continuous method evaluation, expansion of spectral libraries, and implementation of retrospective data mining strategies. The structural and pharmacological diversity of NPS demands flexible, comprehensive analytical frameworks that can adapt to new substances while providing reliable data for public health intervention and policy development. As the NPS market continues to evolve, foundational research focusing on metabolite identification, structure-activity relationships, and high-throughput screening methodologies will be essential for effective forensic response to this persistent challenge.
Validating Comprehensive Testing Panels Against Emerging Substance Threats
The rapid emergence of Novel Psychoactive Substances (NPS) represents a critical challenge for forensic toxicology laboratories and public health authorities worldwide. As of February 2025, the United Nations Office on Drugs and Crime (UNODC) has documented 1,265 distinct NPS classified into 15 chemical structural groups [27]. These compounds are specifically designed to mimic the effects of controlled substances such as cannabis, heroin, cocaine, amphetamine, or LSD while circumventing legal regulations [27]. The structural diversity and rapid emergence of these substances in the global market necessitate advanced analytical methodologies, enhanced laboratory capabilities, and comprehensive routine screening protocols to ensure effective detection and monitoring [27]. This technical guide examines validation frameworks for testing panels against emerging NPS threats, focusing on robust experimental protocols, instrumentation advances, and data interpretation strategies essential for foundational research in NPS identification.
The critical challenge lies in the accelerated development timeline of new substances compared to traditional drug validation workflows. Forensic laboratories face substantial obstacles due to the lack of knowledge about NPS composition, purity, and potency, combined with limited availability of reference analytical standards for confirmatory testing [36]. Without timely screening approaches for accurate detection and identification, public health responses to emerging threats become significantly hampered. This guide provides a comprehensive technical framework for validating testing panels that can adapt to this rapidly evolving landscape, emphasizing high-throughput methodologies, orthogonal verification techniques, and computational approaches that enhance screening capabilities against emerging substance threats.
High-Throughput Screening Technologies for NPS Identification
Benchtop NMR Spectroscopy
Nuclear Magnetic Resonance (NMR) spectroscopy has emerged as a powerful tool for structural identification and elucidation of unknown compounds. Traditional high-field NMR systems require extensive infrastructure and cryogenic cooling, limiting their deployment to specialized research facilities [27]. Modern low-field or benchtop NMR systems utilize compact, permanent magnet designs that eliminate the need for cryogens, enabling deployment in standard laboratory environments including fume hoods while enhancing accessibility and cost-effectiveness [27]. This accessibility makes benchtop NMR particularly valuable for rapid screening of emerging NPS when reference standards may not be available.
Recent research has demonstrated the application of benchtop NMR for constructing comprehensive spectral databases. One study established a benchtop 1H NMR spectral database comprising 360 NPS reference compounds, including 107 synthetic cannabinoids, 89 synthetic cathinones, 46 phenethylamines, 38 fentanyl analogues, 15 phencyclidine-type substances, 13 tryptamines, and 52 "other substances" [27]. This approach enables the development of automated, rapid, accurate, and efficient methods for NPS screening based on benchtop NMR combined with database search algorithms. The validation of this methodology using 50 seized NPS samples demonstrated its effectiveness for reliable identification of emerging threats, with chemical shift correction and solvent peak removal identified as critical factors influencing database search accuracy [27].
Liquid Chromatography-Mass Spectrometry (LC-MS/MS)
High-resolution liquid chromatography-mass spectrometry systems provide another fundamental technology for NPS screening in biological matrices. The SCIEX vMethod application for forensic toxicology screening on the X500R QTOF system provides a comprehensive workflow for sample preparation and LC-MS/MS detection of 664 forensic compounds, including many NPS, in biological matrices in a single injection method [36]. This approach has been expanded to include full characterization of 130 new and prevalent NPS that have recently emerged on the recreational drug market, with parameters including retention times, linear correlation, inter-and intra-day precision and accuracy determined in human urine and whole blood matrices [36].
The LC-MS/MS workflow provides several advantages for NPS identification, including the ability to acquire accurate mass information on all precursor ions, followed by acquisition of multiple, dependent and analyte-specific MS/MS spectra that enable confident identification, confirmation and/or library matching of known or newly emerged substances [36]. Additionally, this approach supports retrospective analysis of previously acquired MS and MS/MS datasets to screen for the presence of new substances without having to re-inject samples, a crucial capability for adapting to emerging threats [36].
Experimental Protocol: Benchtop NMR Database Construction and Validation
Objective: Establish a validated benchtop NMR spectral database for automated identification of novel psychoactive substances.
Materials and Equipment:
- 80 MHz benchtop NMR spectrometer
- Dimethyl sulfoxide-d6 (DMSO-d6) with 0.05% TMS
- 360 NPS reference standards across major structural classes
- MestReNova software with Gears/DB Search toolkit
Methodology:
Sample Preparation:
- Prepare standard solutions of each reference compound in DMSO-d6 with 0.05% TMS
- Utilize consistent concentration protocols across all samples (typically 1-5 mg/mL)
- Transfer 600 μL aliquots to standardized NMR tubes
Spectral Acquisition:
- Collect 1H NMR spectra for all 360 reference compounds using standardized parameters
- Maintain constant temperature (25°C) throughout acquisition
- Implement automated shimming and calibration procedures for reproducibility
Spectral Processing:
- Apply consistent baseline correction across all spectra
- Implement phase correction using automated algorithms with manual verification
- Perform chemical shift correction relative to TMS reference (0 ppm)
- Execute automated peak picking with manual curation
Database Construction:
- Import corrected spectra and chemical structures into MestReNova software
- Annotate key spectral features and structural assignments
- Establish searchable database with metadata including compound class, structural features, and acquisition parameters
Validation Protocol:
- Analyze 50 independent seized drug samples using identical acquisition parameters
- Perform database searching using multiple algorithms (purity peak search, full spectrum correlation)
- Compare top five hit results for identification confidence
- Calculate confidence metrics including match scores and discrimination power
Quality Control Measures:
- Regular instrument performance verification using standard reference materials
- Intra-day and inter-day reproducibility assessment for representative compounds
- Cross-validation with certified reference materials when available
- Continuous database expansion with newly characterized compounds
Validation Frameworks and Quality Assurance
Analytical Performance Metrics
Robust validation of testing panels requires rigorous assessment of analytical performance metrics. High-throughput screening (HTS) approaches adapted from pharmaceutical discovery provide valuable frameworks for assessing assay quality [37]. The Z′ factor serves as a critical metric for evaluating assay quality in HTS applications, calculated as: Z′ = 1 - (3 × (σp + σn)) / |μp - μn| where μp and σp represent the mean and standard deviation of positive control wells, and μn and σn represent the mean and standard deviation of negative controls [37]. Interpretation guidelines specify Z′ ≥ 0.5 as excellent, 0 ≤ Z′ < 0.5 as acceptable with caution for complex assays, and Z′ < 0 as unacceptable [37].
Additional quality metrics include the signal window (SW) calculated as SW = (μp - μn)/σn, and the coefficient of variation (CV) for technical replicates, with targets of CV < 10% for biochemical assays, allowing for higher CV in cell-based systems with appropriate documentation of biological variability [37]. These statistical parameters provide quantitative acceptance criteria essential for reproducible HTS operations and should be enforced at plate-level thresholds prior to data release.
Normalization and Hit-Calling Strategies
Spatial biases (row/column effects) and skewed plate distributions necessitate robust normalization strategies in validation workflows. The B-score algorithm implements median polish on rows and columns followed by scaling by median absolute deviation (MAD), providing a default approach for plates with additive spatial effects [37]. The pseudocode implementation includes:
- Calculate overall median from plate matrix P (r rows × c columns)
- Iterate until convergence:
- Compute roweffect[i] = median(P[i,] - coleffect - overall)
- Subtract roweffect from P
- Compute coleffect[j] = median(P[,j] - roweffect - overall)
- Subtract coleffect from P
- Calculate residuals = P - (overall + roweffect + coleffect)
- Compute B-score = residuals / MAD(residuals)
For hit identification after robust normalization, standardized residual thresholds (typically T = ±3 MAD units) provide primary screening criteria [37]. However, statistical multiple testing correction and experimental replication are essential—implementing Benjamini-Hochberg false discovery rate (FDR) control where p-values are computed, with mandatory confirmation of hits in independent replicates and orthogonal assays [37].
Orthogonal Confirmation and Specificity Assessment
Comprehensive validation requires orthogonal confirmation to eliminate false positives from analytical artifacts. Recommended replicate workflow includes:
- Primary single-concentration screen with retention of top 1-2% of compounds
- Retest of retained set in duplicates/triplicates at same concentration
- Progression of replicated hits to 8-12 point dose-response curves (4PL/5PL fits)
- Orthogonal counterscreens using different detection principles or label-free technologies to exclude artifacts
Specificity assessment must include evaluation against pan-assay interference compounds (PAINS) and other artifactual responses [37]. Automated substructure filters flagging reactive groups (Michael acceptors, isothiocyanates), metals, and likely aggregators should be implemented, combined with detergent counterscreens (e.g., 0.01% Triton X-100) to identify colloidal aggregators [37]. These measures ensure that identified hits represent genuine interactions rather than analytical artifacts.
Experimental Workflow Visualization
NPS Identification Workflow
Data Analysis and Confidence Criteria
Quantitative Assessment Metrics
For confident NPS identification, analytical data must meet stringent confidence criteria. LC-MS/MS-based approaches typically employ four main confidence parameters [36]:
- Mass Error (M): Precursor mass accuracy typically within ±5 ppm of theoretical value
- Retention Time (R): Consistent retention time matching within narrow window (±0.1 min)
- Isotope Ratio Difference (I): Isotopic pattern matching between reference and sample
- Library Score (L): Spectral similarity scoring against reference fragmentation patterns
In benchtop NMR approaches, database search algorithms employ similarity metrics including purity peak search and full spectrum correlation, with chemical shift correction and solvent peak removal identified as critical factors influencing accuracy [27]. The top five hit results from database searches provide tiered confidence levels, with the highest-ranking match requiring manual verification for novel or unusual compounds.
Statistical Rigor in Hit Confirmation
Robust statistical approaches are essential for minimizing false positives in NPS identification. After initial screening, dose-response modeling using four-parameter logistic (4PL) or five-parameter logistic (5PL) curves provides quantitative potency assessment [37]. The 4PL equation is defined as:
Y = Bottom + (Top - Bottom) / (1 + 10^((LogIC50 - X) × HillSlope))
where X is log10(concentration), Top and Bottom are asymptotes, HillSlope defines steepness, and LogIC50 is log10(IC50) [37]. Weighted nonlinear least squares fitting is recommended when variance is heteroscedastic, with reporting of 95% confidence intervals for IC50 and Hill slope parameters.
Implementation of reproducible HTS pipelines with standardized QC checklists ensures consistent performance across screening campaigns [37]. Essential components include LIMS and plate mapping with barcode-driven tracking, instrument orchestration with scheduling and device integration, data warehouses with raw plate files and audit logs, and containerized analytics implementing standardized algorithms with version control.
Essential Research Reagents and Materials
Table 1: Essential Research Reagents for NPS Screening Validation
| Reagent/Material | Specification | Application Purpose | Supplier Examples |
|---|---|---|---|
| NPS Reference Standards | 360+ compounds across structural classes | Method development, calibration, library building | Cerilliant Corporation, Cayman Chemical Company |
| Deuterated Solvents | DMSO-d6 with 0.05% TMS | NMR sample preparation, chemical shift reference | Cambridge Isotope Laboratories |
| LC-MS Mobile Phase Additives | Ammonium formate, formic acid | Chromatographic separation, ionization enhancement | Various chemical suppliers |
| Solid Phase Extraction Cartridges | Mixed-mode polymeric sorbents | Sample clean-up, analyte concentration | Various manufacturers |
| Quality Control Materials | Certified reference materials | Method validation, quality assurance | NIST, LGC Standards |
| HPLC Columns | Phenyl-Hexyl, 50 × 4.6 mm, 2.6 µm | Compound separation, isomer resolution | Phenomenex |
| Internal Standards | Deuterated analogs of target compounds | Quantification, process monitoring | Cerilliant Corporation |
| Protein Precipitation Solvents | Methanol/acetonitrile (50:50) | Matrix protein removal, sample preparation | Various chemical suppliers |
Technological Integration and Workflow Optimization
Complementary Technique Integration
Effective NPS identification increasingly requires integration of complementary analytical techniques to address limitations of individual methodologies. While LC-MS/MS provides excellent sensitivity and specificity for targeted analysis, and NMR offers unparalleled structural elucidation capabilities for unknowns, combining these with additional techniques creates a more robust identification framework. Infrared spectroscopy (IR) and Raman spectroscopy can provide complementary structural information, particularly for isomeric compounds that may challenge mass spectrometry-based approaches [27].
The development of multi-technique databases that incorporate NMR, IR, and MS data creates powerful identification resources that can overcome limitations of individual techniques [27]. Such integrated approaches are particularly valuable for isomeric differentiation, where mass spectrometry alone may be insufficient, and for complex mixture analysis where chromatographic separation may not fully resolve all components. Implementation of pattern recognition algorithms and multivariate statistical analysis enhances the utility of these complementary datasets for rapid classification of unknown substances.
Automated Workflows and Data Integration
Automation represents a critical advancement for comprehensive NPS screening in high-volume laboratory environments. Automated liquid handling systems streamline sample preparation, improving reproducibility while reducing manual labor requirements and potential for human error [36]. Automated data processing workflows integrate peak detection, spectral matching, and result reporting, with customizable confidence thresholds and review criteria for anomalous results [36].
The implementation of laboratory information management systems (LIMS) with barcode-driven tracking of samples, reagents, and instrumentation parameters ensures complete audit trails and data provenance [37]. Instrument orchestration platforms that schedule analyses across multiple systems with job recovery routines maximize operational efficiency, while data warehouses with checksum-verified storage protect data integrity for regulatory compliance and retrospective analysis [37].
The evolving landscape of novel psychoactive substances demands continuous advancement in analytical validation strategies. Future developments will likely focus on in silico prediction of spectral properties for unsynthesized compounds, machine learning-enhanced identification of structural analogs from partial spectral matches, and miniaturized field-deployable instruments for rapid onsite screening. Additionally, international collaboration for data sharing across laboratories and jurisdictions will enhance early warning systems for emerging threats.
Validation frameworks must remain adaptable to incorporate emerging technologies while maintaining rigorous quality standards. The integration of high-throughput synthesis approaches with rapid biological screening, coupled with computational toxicology prediction, will enable more proactive responses to the NPS threat. By implementing comprehensive validation protocols that emphasize orthogonal confirmation, statistical rigor, and continuous method improvement, research laboratories can maintain effective surveillance capabilities against the constantly evolving landscape of novel psychoactive substances.
The rapid proliferation of New Psychoactive Substances (NPS) represents a critical public health challenge and a complex analytical problem for forensic toxicologists and research scientists. According to the United Nations Office on Drugs and Crime definition, NPS constitute "new narcotic or psychotropic drugs that are not controlled by the United Nations drug conventions, but which may pose a public health threat comparable to controlled substances" [30]. The European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) reports approximately 50-60 new NPS are detected annually on the drug market, with potentially more remaining undetected due to analytical limitations [30]. This technical review examines longitudinal data on NPS prevalence from 2023 to 2025, detailing advanced analytical methodologies essential for foundational research in substance identification and toxicological assessment.
The fundamental challenge in NPS research stems from the deliberate chemical modifications designed to circumvent legislative controls while maintaining psychoactive effects. Clandestine laboratories systematically make minor structural alterations to regulated substances—such as terminal fluorine replacement or indole-to-indazole substitutions—creating new compounds that escape conventional targeted analytical methods [30]. This continuous structural evolution necessitates developing increasingly sophisticated non-targeted detection protocols and benchmarking prevalence across diverse populations to inform public health responses and regulatory policies.
Longitudinal NPS Prevalence Data (2023-2025)
Epidemiological tracking of NPS consumption patterns provides crucial data for understanding substance use trends and directing analytical resources. Recent studies employing advanced detection methodologies have yielded population-specific prevalence rates essential for benchmarking purposes.
2023 Prevalence Data
Table 1: NPS Prevalence Benchmarking Data (2023)
| Study Population | Sample Size | Positive for Traditional Drugs | Positive for NPS | Most Prevalent NPS | Polydrug Use |
|---|---|---|---|---|---|
| SUD Patients (Italy) | 88 | 88.1% | 11.9% | Ketamine (8.0%), Buphedrone | 67.9% |
| General Population (Retrospective) | 307,935 (2007-2014) | N/A | Increasing trend | Hallucinogens, Stimulants | Prevalent pattern |
A 2025 study analyzing 2023 data from substance use disorder (SUD) patients in Bologna, Italy, provided granular insights into NPS prevalence within high-risk populations. Among 88 patients, 95.5% tested positive for at least one substance, with the majority (88.1%) positive for traditional drugs of abuse only, while 11.9% tested positive for NPS in addition to traditional drugs [38]. This study demonstrated a strong association between polydrug consumption patterns and NPS use, with 67.9% of positive samples containing more than two drugs [38]. Ketamine emerged as the most prevalent substance within the NPS category, detected in 8.0% of all samples, with mean hair concentrations of 49.68 pg/mg (range: 8.55-81.90 pg/mg) [38]. Notably, ketamine consumption frequently occurred alongside cocaine use (85.7% of cases), highlighting a distinct polysubstance use pattern [38].
Demographic analysis revealed significant age-based distribution in NPS consumption patterns. The combination of traditional drugs of abuse and NPS was statistically more frequent in younger age groups (<21-30 years old) compared to older cohorts (31-70 years, p = 0.025) [38]. This age stratification aligns with broader epidemiological patterns identified in population-level studies.
2024-2025 Prevalence Trends and Methodological Advances
The period of 2024-2025 has witnessed critical methodological innovations in NPS detection, particularly through non-targeted screening approaches. Analytical techniques have evolved significantly to address the limitations of targeted methods, which frequently miss novel analogues and metabolites not included in reference databases.
Recent research emphasis has shifted toward suspect screening using diagnostic fragment ions, enabling preliminary identification of structurally related NPS without requiring reference standards [30]. This approach has proven particularly valuable for detecting emerging synthetic opioids like fentanyl analogues (detected in 3.4% of samples in the Italian study) and cathinone derivatives such as buphedrone [38]. The continuous refinement of high-resolution mass spectrometry techniques has substantially improved detection capabilities for NPS metabolites, which often present longer detection windows than parent compounds in biological matrices [30].
Experimental Protocols for NPS Identification
Sample Preparation Methodologies
Table 2: Sample Preparation Protocols for Biological Matrices
| Biological Matrix | Primary Preparation Method | Key Considerations | Extraction Technique |
|---|---|---|---|
| Hair | Segmentation & Washing | Decontamination protocols critical | Solid-Phase Extraction |
| Blood/Plasma | Protein Precipitation | Short detection window for parent compounds | Liquid-Liquid Extraction |
| Urine | Enzymatic Hydrolysis | Metabolite detection predominant | Solid-Phase Extraction |
Sample preparation represents a critical first step in NPS analysis, with methodology varying significantly by biological matrix. For hair analysis, samples are typically segmented (0-2 cm, 2-4 cm, etc.), washed with appropriate solvents (e.g., dichloromethane) to remove external contamination, and subsequently pulverized or cut into fine segments to enhance extraction efficiency [38]. For blood and urine matrices, protein precipitation using acetonitrile or methanol is commonly employed, followed by sophisticated extraction techniques [39].
Solid-phase extraction (SPE) has emerged as the predominant sample preparation technique for NPS analysis in biological matrices, offering superior clean-up efficiency and analyte recovery compared to traditional liquid-liquid extraction [39]. Recent methodological developments have focused on creating more agile, straightforward preparation procedures with reduced toxicity, particularly through decreased consumption of hazardous solvents [39]. For urine analysis, enzymatic hydrolysis using β-glucuronidase/sulfatase enzymes is frequently necessary to detect phase II metabolites (glucuronides), which often constitute the primary excretion form for many NPS classes [30].
Analytical Detection Techniques
Advanced instrumental techniques form the cornerstone of modern NPS identification, with liquid chromatography coupled to tandem mass spectrometry representing the current gold standard.
Liquid Chromatography Separation: Reverse-phase chromatography employing C18 columns has demonstrated optimal performance for NPS separation, providing sufficient retention and resolution for structurally diverse compounds [39]. Mobile phase systems typically consist of water or aqueous buffers (with modifiers such as formic acid or ammonium formate) and organic solvents (acetonitrile or methanol) in gradient elution programs optimized for the specific NPS class under investigation.
Mass Spectrometric Detection: The emergence of high-resolution mass spectrometry (HRMS) has revolutionized NPS detection, with two primary platforms dominating current applications:
- Quadrupole-time-of-flight (Q-TOF) instruments offer high mass accuracy (<5 ppm) and rapid acquisition speeds suitable for untargeted screening.
- Orbitrap-based systems provide ultra-high resolution (>60,000 FWHM) and excellent mass accuracy for confident molecular formula assignment.
Table 3: Mass Spectrometry Configuration for NPS Analysis
| Parameter | Recommended Configuration | Rationale |
|---|---|---|
| Ionization Source | Electrospray Ionization (ESI) | Optimal for broad NPS classes |
| Polarity Mode | Positive & Negative Switching | Comprehensive coverage |
| Resolution | >30,000 FWHM | Sufficient for formula assignment |
| Mass Accuracy | <5 ppm | Confident molecular identification |
| Collision Energy | Stepped HCD (20-50 eV) | Comprehensive fragmentation |
Tandem mass spectrometry employing collision-induced dissociation (CID) or higher-energy collisional dissociation (HCD) generates characteristic fragment ions essential for structural elucidation [30]. The application of diagnostic fragment ion screening enables the detection of structurally related NPS analogues sharing common core structures, even in the absence of reference standards [30].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Essential Research Reagents for NPS Identification
| Reagent/Material | Function | Application Specifics |
|---|---|---|
| Certified Reference Standards | Method validation & quantitative analysis | Limited availability for novel NPS; structural analogues used as substitutes |
| β-Glucuronidase/Sulfatase Enzymes | Hydrolysis of phase II metabolites | Essential for urine analysis; enables detection of glucuronidated metabolites |
| Solid-Phase Extraction Cartridges | Sample clean-up & analyte concentration | Reverse-phase (C18) & mixed-mode polymers most effective |
| LC-MS Grade Solvents | Mobile phase preparation; minimal background interference | Acetonitrile/methanol with aqueous modifiers (formic acid, ammonium formate) |
| HRMS Calibration Solutions | Mass axis calibration & instrument performance verification | Ensures <5 ppm mass accuracy critical for molecular formula assignment |
| Quality Control Materials | Method validation & accuracy/precision assessment | Certified negative/positive matrices for quality assurance |
The analytical workflow for NPS identification requires specialized reagents and materials to ensure accurate and reproducible results. The critical challenge remains the limited availability of certified reference standards for newly emerging substances, necessitating innovative approaches to compound identification [30]. Method development increasingly relies on diagnostic fragment ion analysis and retrospective data mining of high-resolution mass spectrometry data as new reference materials become available [30].
Quality assurance protocols require the implementation of comprehensive quality control materials, including certified negative matrices (drug-free hair, blood, and urine) and positive controls for monitored substances. The continuous updating of in-house spectral libraries and expanded databases incorporating fragment ion information represents an essential component of the modern NPS research laboratory [30].
Signaling Pathways and Metabolic Considerations
Understanding the metabolic pathways of NPS is crucial for developing effective detection strategies, as metabolites often provide longer detection windows and valuable consumption markers.
NPS undergo extensive biotransformation through phase I (functionalization) and phase II (conjugation) metabolic pathways [30]. Phase I transformations typically include hydroxylation, dealkylation, carbonylation, dehydrogenation, and oxidative defluorination, while phase II metabolism primarily involves glucuronidation and sulfation [30]. These metabolic pathways significantly impact detection strategies:
- Urine analysis primarily targets metabolites rather than parent compounds, as most NPS are extensively transformed and excreted as conjugates [30].
- Hair analysis predominantly detects the parent compound, though metabolites can provide valuable confirmation of active consumption versus external contamination [30].
- Blood and oral fluid typically contain both parent compounds and metabolites, but with shorter detection windows compared to urine and hair matrices [30].
The strategic selection of analytical targets must consider the biological matrix, time since consumption, and metabolic pathways specific to each NPS class. This understanding informs the development of comprehensive screening approaches that account for both parent compounds and characteristic metabolites.
Longitudinal benchmarking of NPS prevalence from 2023 to 2025 reveals consistent patterns of polydrug use, demographic stratification, and continuous chemical evolution that challenge conventional analytical approaches. The data indicate that while overall NPS prevalence remains relatively low compared to traditional drugs of abuse in specific populations (11.9% in SUD patients), certain substances like ketamine demonstrate significant penetration in recreational drug markets [38].
The advancement of non-targeted screening methodologies using high-resolution mass spectrometry represents the most promising approach for addressing the analytical challenges posed by NPS. Future research directions should prioritize:
- Expansion of spectral libraries to include fragment ion data for structural analogues
- Development of rapid metabolite prediction algorithms to accelerate method development
- Implementation of retrospective data mining protocols for identifying previously undetected substances
- Standardization of analytical workflows across laboratories to improve data comparability
The dynamic nature of the NPS market necessitates continuous methodological innovation and international collaboration between forensic laboratories, research institutions, and public health agencies. Only through integrated approaches combining advanced analytical techniques, epidemiological surveillance, and clinical toxicology can effective responses to the evolving threat of new psychoactive substances be developed and implemented.
The rapid proliferation of novel psychoactive substances (NPS) presents a critical global health challenge, with new compounds emerging faster than traditional research methodologies can characterize their risks. Addressing this threat requires unprecedented cross-sector collaboration among forensic, clinical, and public health domains. This technical guide establishes a framework for validating NPS findings through integrated approaches, leveraging the unique capabilities of each sector to create a comprehensive understanding of these substances. The NPS Discovery initiative exemplifies this approach, producing quarterly trend reports based on analysis of authentic forensic samples to provide near real-time information on NPS prevalence and positivity [18]. Such coordinated efforts are essential for developing effective public health responses and foundational research for substance identification and harm reduction.
Quantitative Landscape of Cross-Sector Collaboration
Current State of Collaborative Engagement
Systematic analysis reveals significant variations in how different sectors engage in public health activities related to substance surveillance. The breadth and depth of collaboration can be quantified across organizational types, demonstrating both strengths and gaps in current NPS response networks.
Table 1: Breadth and Depth of Social Service Engagement in Public Health Activities
| Type of Social Service Organization | Extensive Margins (%) | Intensive Margins (%) |
|---|---|---|
| Basic Needs | ||
| Housing, shelter, utilities | 73.8 | 24.3 |
| Food and nutrition | 70.2 | 10.1 |
| Transportation | 64.8 | 16.6 |
| Special Populations | ||
| Veterans | 63.7 | 19.7 |
| Child and family support | 62.5 | 5.7 |
| People with disabilities | 60.5 | 19.6 |
| Criminal Justice | ||
| Law enforcement | 46.0 | 13.0 |
| Corrections, criminal, and juvenile justice | 32.5 | 5.0 |
| Legal assistance | 32.3 | 9.4 |
Data extracted from a national longitudinal survey of public health systems demonstrates that engagement is most prevalent with organizations providing basic needs such as housing (73.8% of communities) and food assistance (70.2% of communities) [40]. However, collaboration with justice-related organizations remains notably limited, with less than 33% of communities engaging law enforcement, corrections, or legal assistance organizations in public health activities [40]. This collaboration gap represents a critical vulnerability in NPS response, as these entities often encounter emerging substance trends before traditional healthcare systems.
Forensic Science Contribution to NPS Surveillance
The forensic sector provides indispensable data through systematic analysis of substances obtained from law enforcement operations, overdose scenes, and other legal contexts. The NPS Discovery Q3 2025 Trend Reports focus on four main NPS classifications: benzodiazepines, opioids, stimulants and hallucinogens, and synthetic cannabinoids [18]. This forensic data creates the foundational chemical identification necessary for clinical and public health responses.
Table 2: Cross-Sector Data Contributions to NPS Research
| Sector | Primary Data Contribution | Validation Role | Methodologies |
|---|---|---|---|
| Forensic | Chemical identification, purity analysis, structural elucidation | Confirms substance identity and composition | Mass spectrometry, chromatography, spectroscopy |
| Clinical | Patient presentation, toxicity patterns, treatment outcomes | Correlates chemical data with human effects | Medical toxicology assessment, patient monitoring, biomarker analysis |
| Public Health | Population-level prevalence, trend analysis, risk communication | Contextualizes findings across communities | Epidemiological surveillance, syndromic monitoring, data integration |
The interconnection of these data streams creates a robust validation framework where forensic chemical analysis confirms substance identity, clinical observation correlates these findings with human toxicity, and public health monitoring tracks population-level spread and impact [18] [41].
Methodological Framework for Cross-Sector NPS Research
Experimental Protocols for NPS Identification and Validation
Effective cross-sector collaboration requires standardized protocols that enable data sharing and methodological alignment across domains. The following experimental workflows ensure consistent application of techniques and interpretation of results.
Figure 1: Integrated Workflow for Cross-Sector NPS Identification and Validation. This diagram illustrates the sequential process of NPS characterization across forensic, clinical, and public health domains, demonstrating how findings are validated through multi-sector analysis.
Cross-Sector Validation Methodology
The validation of NPS findings requires a systematic approach that integrates complementary methodologies from each sector:
Forensic Analytical Protocols
- Sample Preparation: Implement standardized extraction procedures using solid-phase extraction (SPE) techniques with quality controls including blanks, duplicates, and certified reference materials.
- Instrumental Analysis: Employ liquid chromatography-quadrupole time-of-flight mass spectrometry (LC-QTOF-MS) for broad-spectrum toxicological screening, with gas chromatography-mass spectrometry (GC-MS) confirmation.
- Data Interpretation: Utilize advanced library matching algorithms coupled with manual verification to identify novel compounds not present in existing databases.
Clinical Correlation Protocols
- Patient Assessment: Implement standardized toxicology assessment tools to document physiological and psychological effects, including vital signs, neurological status, and psychiatric symptoms.
- Biomarker Analysis: Collect and analyze biological samples (blood, urine) to correlate substance exposure with clinical presentation, including quantitative analysis of parent compounds and metabolites.
- Outcome Documentation: Track patient course through healthcare systems, including emergency department presentations, hospital admissions, and fatalities with toxicological confirmation.
Public Health Integration Protocols
- Data Aggregation: Establish secure data sharing platforms that integrate information from forensic laboratories, healthcare facilities, and community-based organizations while protecting privacy.
- Trend Analysis: Apply statistical process control methods to detect aberrations in substance occurrences, clinical presentations, and population-level indicators.
- Risk Communication: Develop evidence-based alerts and guidance for healthcare providers, public safety personnel, and the public based on validated cross-sector findings.
Essential Research Reagent Solutions for NPS Analysis
The accurate identification and characterization of NPS requires specialized reagents and reference materials that enable precise chemical analysis and biological assessment.
Table 3: Research Reagent Solutions for NPS Analysis
| Reagent Category | Specific Examples | Research Application | Sector Utilization |
|---|---|---|---|
| Certified Reference Standards | Fentanyl analogs, synthetic cannabinoids, cathinones | Quantitative analysis, method validation, instrument calibration | Forensic, Clinical, Public Health |
| Immunoassay Reagents | Synthetic opioid panels, benzodiazepine class antibodies | Rapid screening, high-throughput testing | Clinical, Public Health |
| Metabolite Reference Materials | Phase I and Phase II metabolite standards | Metabolic pathway elucidation, biomarker identification | Forensic, Clinical |
| Molecular Biology Kits | CYP450 enzyme panels, receptor binding assay systems | Mechanism of action studies, toxicity prediction | Clinical, Research |
| Chromatography Supplies | HPLC columns, GC liners, SPE cartridges | Sample preparation, compound separation | Forensic, Clinical |
The selection of appropriate research reagents must consider the emerging nature of NPS, requiring flexible platforms capable of identifying unanticipated compounds. Mass spectrometry-based approaches particularly depend on high-quality reference materials for accurate substance identification [18]. Additionally, the development of targeted immunoassays for novel substance classes enables more efficient clinical and public health screening.
Data Integration and Visualization Framework
Principles for Effective Data Presentation
Cross-sector collaboration generates complex datasets that require careful presentation to facilitate interpretation and decision-making. Effective data tables for NPS research should adhere to the following design principles:
- Focus on Essential Data: Include only the data that the audience needs to focus on, removing extraneous information that can distract from key findings [42].
- Intentional Formatting: Use table titles, column titles, and color/boldness strategically to emphasize key takeaways from the data [42].
- Structural Logic: Order data to match the purpose of the table, facilitating how it will be used by researchers and decision-makers [43].
- Visual Hierarchy: Create clear visual hierarchies through typography to guide the reader's eye to the most important information [43].
- Accessibility Compliance: Ensure sufficient color contrast (at least 4.5:1 for normal text) to make data readable for all users, including those with visual impairments [44].
Cross-Sector Data Integration Model
Successful NPS surveillance requires not only data collection within sectors but also sophisticated integration across domains. This integration enables the validation of findings through triangulation of evidence from multiple sources.
Figure 2: Cross-Sector Data Integration Model for NPS Research. This diagram illustrates how data from forensic, clinical, and public health domains converges into an integrated platform that supports multiple outputs including validated alert systems, research databases, and policy development.
The data integration platform enables several critical functions for NPS research:
Triangulation Validation: Findings from one sector (e.g., identification of a new substance in forensic analysis) can be correlated with data from other sectors (e.g., novel clinical presentations or population-level usage trends) to confirm significance and urgency.
Temporal Trend Analysis: Integrated data allows researchers to track the emergence, spread, and decline of specific substances across time and geography, providing insights into market dynamics and intervention effectiveness.
Risk Stratification: By combining chemical data with clinical outcomes, the integrated platform enables development of risk prediction models that can prioritize resources toward the most dangerous emerging substances.
Implementation Challenges and Solutions
Barriers to Effective Cross-Sector Collaboration
Despite the clear theoretical benefits, implementing effective cross-sector collaboration for NPS research faces significant practical challenges:
- Data Sharing Restrictions: Legal and privacy concerns often prevent sharing of critical information between forensic, clinical, and public health entities. Solution: Develop standardized data use agreements that define specific elements for sharing while protecting sensitive information.
- Methodological Incompatibility: Different sectors often use disparate classification systems, analytical methods, and data formats. Solution: Establish harmonized technical standards through interdisciplinary working groups.
- Resource Limitations: Cross-sector collaboration requires dedicated personnel, technical infrastructure, and sustained funding. Solution: Leverage existing surveillance infrastructure and pursue targeted funding opportunities that require interdisciplinary approaches.
- Communication Gaps: Professional vocabulary and operational priorities differ significantly across sectors. Solution: Create cross-sector liaison positions and develop shared glossaries that translate terminology across domains.
Metrics for Evaluating Collaborative Effectiveness
To ensure continuous improvement in cross-sector NPS research, the following metrics should be tracked:
- Temporal Efficiency: Measure the time from initial substance identification in forensic settings to validated public health communication.
- Completeness of Data Integration: Track the percentage of identified NPS with correlated data across all three sectors.
- Outcome Impact: Monitor trends in morbidity and mortality associated with NPS before and after implementation of collaborative interventions.
- Stakeholder Engagement: Assess the breadth and depth of organizational participation across sectors using established metrics [40].
The evolving threat of novel psychoactive substances demands a sophisticated cross-sector approach that integrates capabilities from forensic, clinical, and public health domains. By implementing structured validation frameworks, standardized experimental protocols, and integrated data systems, researchers can transform isolated findings into comprehensive understanding. The quarterly trend reports produced by initiatives like NPS Discovery represent important steps toward this collaborative model [18]. As cross-sector relationships continue to develop, particularly with currently underutilized partners in justice and social service sectors [40], the capacity to rapidly identify, characterize, and respond to emerging NPS will significantly enhance public health and safety. Future research should focus on quantifying the impact of these collaborative approaches on substance-related harms and refining the technical methodologies that enable seamless data integration across sectors.
Conclusion
Foundational research into NPS identification reveals a public health ecosystem in constant flux, characterized by the rapid emergence of potent synthetic opioids, the infiltration of veterinary sedatives and industrial chemicals into the drug supply, and the persistent challenge of legislative evasion. The key takeaway is the indispensable need for agile, near real-time monitoring systems, comprehensive analytical methodologies, and robust cross-sector collaboration to keep pace with these threats. Future research must prioritize the development of rapid, accessible testing methods, expanded reference material libraries, and deeper investigation into the clinical toxicology and long-term health impacts of emerging adulterants. Only through a coordinated and proactive research agenda can biomedical and clinical science hope to mitigate the risks posed by the evolving NPS landscape.
References
- 1. What are NPS? [https://www.unodc.org/LSS/Page/NPS]
- 2. Recent advances in the identification and quantification of ... [https://pubmed.ncbi.nlm.nih.gov/41085046/]
- 3. The Shifting Landscape of a Fentanyl Adulterant [https://pubmed.ncbi.nlm.nih.gov/40990306/]
- 4. 2025 Mid-Year Update on Novel Psychoactive Substances ... [https://www.aegislabs.com/clinical-update/2025-mid-year-update-on-nps-trends/]
- 5. Medetomidine: What you need to know about the new ... [https://www.substanceusephilly.com/medetomidine]
- 6. Overdoses Involving Medetomidine Mixed with Opioids [https://www.cdc.gov/mmwr/volumes/74/wr/mm7415a1.htm]
- 7. What are fentanyl and nitazenes? Explaining the rise and ... [https://adf.org.au/insights/fentanyl-and-nitazenes/]
- 8. Public Alerts [https://www.cfsre.org/nps-discovery/public-alerts]
- 9. At-a-glance: New psychoactive substances in Canada - 2023 [https://www.canada.ca/en/health-canada/services/publications/healthy-living/new-psychoactive-substances-canada-2023.html]
- 10. Development of a high-resolution paper-spray mass ... [https://pubs.rsc.org/en/content/articlehtml/2025/an/d5an00086f]
- 11. New psychoactive substances: a multidisciplinary review of ... [https://pubmed.ncbi.nlm.nih.gov/40773075/]
- 12. PFAS Legislative and Regulatory Developments — First ... [https://www.paulhastings.com/insights/phast-track-legal-insights-on-environment-energy-and-infrastructure/pfas-legislative-and-regulatory-developments-first-quarter-2025]
- 13. Recent Federal Developments for August 2025 [https://www.lawbc.com/recent-federal-developments-for-august-2025/]
- 14. Uncovering new psychoactive substances research trends ... [https://www.sciencedirect.com/science/article/pii/S2772416625001123]
- 15. New psychoactive substances: a review and updates - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7750892/]
- 16. The novel psychoactive substances epidemic [https://www.sciencedirect.com/science/article/pii/S2772392522000554]
- 17. Differentiation of methyl-substituted fentanyl analogs using ... [https://www.sciencedirect.com/science/article/abs/pii/S2468170925000724]
- 18. NPS Discovery Q3 2025 Trend Reports [https://www.ojp.gov/library/publications/nps-discovery-q3-2025-trend-reports-nps-benzodiazepines-nps-opioids-nps]
- 19. NPS Discovery Q2 2025 TREND REPORTS [https://nij.ojp.gov/library/publications/nps-discovery-q2-2025-trend-reports-nps-benzodiazepines-nps-opioids-nps]
- 20. ortho-Methylfentanyl [https://www.cfsre.org/nps-discovery/monographs/ortho-methylfentanyl]
- 21. Analysis of cis and trans 3-methylfentanyl by liquid ... [https://pubmed.ncbi.nlm.nih.gov/29801193/]
- 22. Detecting novel psychoactive substances around the world [https://pubmed.ncbi.nlm.nih.gov/38818825/]
- 23. 2024 Novel Psychoactive Substance Summary [https://www.aegislabs.com/services/novel-psychoactive-substances/2024-nps-summary/]
- 24. Evaluation of long‐term detection trends of new psychoactive ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7080623/]
- 25. Provisional Drug Overdose Death Counts [https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm]
- 26. Epidemiology of New Psychoactive Substances in Relation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11730279/]
- 27. Automated rapid screening of new psychoactive ... [https://www.sciencedirect.com/science/article/abs/pii/S0379073825002026]
- 28. What's Trending: NPS Discovery Webinar Series - July 2025 [https://www.cfsre.org/education/continuing-professional-education/archival-on-demand-education/what-s-trending-nps-discovery-webinar-series-july-2025]
- 29. Clinical value of analytical testing in patients presenting ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7080633/]
- 30. Identification of new psychoactive substances and their ... [https://www.sciencedirect.com/science/article/abs/pii/S0039914023005672]
- 31. 2025 Mid-Year NPS Summary [https://www.aegislabs.com/services/novel-psychoactive-substances/2025-mid-year-nps-update/]
- 32. Trend Reports [https://www.cfsre.org/nps-discovery/trend-reports]
- 33. Recent Trends in Analytical Methods to Determine New ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5771044/]
- 34. Novel Psychoactive Substances (NPS) analysis [https://sciex.com/applications/forensics-testing/forensic-toxicology/novel-psychoactive-substances-nps-analysis]
- 35. Artificial intelligence (AI) in New Psychoactive Substances ... [https://pubmed.ncbi.nlm.nih.gov/40668255/]
- 36. Expanding NPS screening capabilities in the forensic ... [https://sciex.com/tech-notes/forensic/toxicology/expanding-nps-screening-capabilities-in-the-forensic-toxicology]
- 37. High Throughput Screening [https://www.chemdiv.com/high-throughput-screening-hts/]
- 38. Prevalence of new psychoactive substances and drugs ... [https://pubmed.ncbi.nlm.nih.gov/39655618]
- 39. Analytical detection of New Psychoactive Substances in ... [https://revista.rbc.org.br/index.php/rbc/article/view/943]
- 40. Breadth and Depth of Cross-Sector Collaboration - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7362695/]
- 41. Forensic Nurse Practitioner: Bridging Healthcare and the ... [https://collaboratingdocs.com/forensic-nurse-practitioner-bridging-healthcare-and-the-legal-system/]
- 42. Data Visualization Toolkit: Data Tables Design Principles [https://dasycenter.org/datavis-toolkit/data-tables/]
- 43. Designing Data Tables [https://understandinggraphics.com/design/data-table-design/]
- 44. Contrast Checker [https://webaim.org/resources/contrastchecker/]