Designing TRL 4 Inter-Laboratory Validation Studies for Court-Ready Forensic Methods
This article provides a comprehensive framework for designing inter-laboratory validation studies to advance forensic methods to Technology Readiness Level (TRL) 4.
Designing TRL 4 Inter-Laboratory Validation Studies for Court-Ready Forensic Methods
Abstract
This article provides a comprehensive framework for designing inter-laboratory validation studies to advance forensic methods to Technology Readiness Level (TRL) 4. It guides researchers and forensic professionals through the foundational principles, methodological execution, troubleshooting strategies, and final validation required to demonstrate that a method is robust, standardized, and ready for implementation in casework. Special emphasis is placed on meeting the stringent legal admissibility standards, such as the Daubert Standard and Federal Rule of Evidence 702, which require known error rates, peer review, and general acceptance within the scientific community.
Laying the Groundwork: From TRL 3 to TRL 4 and Legal Admissibility
Defining Technology Readiness Level 4 in Forensic Contexts
Technology Readiness Levels (TRLs) represent a systematic metric for assessing the maturity of a particular technology, typically on a scale from 1 (basic principles observed) to 9 (system proven in operational environment) [1]. In forensic science, this framework helps standardize the development and implementation of new analytical methods, ensuring they meet the rigorous demands of legal proceedings. The transition from promising research to court-admissible evidence requires careful navigation of both technical and legal standards, including the Daubert Standard and Federal Rule of Evidence 702 in the United States, which emphasize testing, peer review, error rates, and general acceptance within the scientific community [2].
Within forensic chemistry publications, a specialized four-level TRL scale is often employed to better reflect the development pathway of analytical methods intended for crime laboratory implementation [3]. This adapted framework places specific emphasis on validation and standardization requirements at each stage. TRL 4 represents a critical milestone where methods transition from preliminary proof-of-concept to being substantiated through multi-laboratory validation, making them candidates for implementation in operational forensic laboratories [3].
Defining TRL 4 in Forensic Contexts
Core Definition and Position in the Development Pathway
In forensic contexts, Technology Readiness Level 4 signifies the stage where a method undergoes refinement, enhancement, and inter-laboratory validation to become a standardized protocol ready for implementation in forensic laboratories [3]. Research at this level generates knowledge that can be "immediately adopted or used in casework" [3]. This represents a significant advancement beyond TRL 3, where techniques are applied to specific forensic applications with measured figures of merit and aspects of intra-laboratory validation, but lack independent verification across multiple laboratories [3].
The fundamental distinction of TRL 4 research is its focus on establishing reproducibility and reliability across different institutional settings, instruments, and operators. This inter-laboratory validation is essential for forensic methods because results must withstand legal scrutiny and be independent of the specific laboratory that generated them. Methods reaching TRL 4 have typically addressed key variables that could affect analytical outcomes and have demonstrated robustness through standardized protocols.
Comparative Analysis of TRL Frameworks
Table 1: TRL 4 Definitions Across Different Frameworks
| Framework | TRL 4 Definition | Key Emphasis | Primary Context |
|---|---|---|---|
| Forensic Chemistry Journal | "Refinement, enhancement, and inter-laboratory validation of a standardized method ready for implementation in forensic laboratories" [3] | Inter-laboratory validation, error rate measurement, database development | Forensic method development for crime laboratories |
| Traditional NASA/ESA Scale | "Component and/or breadboard validation in laboratory environment" [1] | Component integration and testing in laboratory setting | Aerospace and general technology development |
| Canadian Government Scale | "Component and/or validation in a laboratory environment" [4] | Integration of basic technological components in laboratory | Broad technology assessment |
| Medical Countermeasures | "Optimization and Preparation for Assay, Component, and Instrument Development" [5] | Down-selecting targets, finalizing methods, developing detailed plans | Medical device and diagnostic development |
As illustrated in Table 1, the forensic chemistry adaptation of TRL 4 places greater emphasis on collaborative validation and immediate applicability to casework compared to more traditional TRL frameworks. While the NASA/ESA scale focuses on component-level validation in laboratory environments, the forensic context specifically requires multi-laboratory participation to establish method reliability across the forensic community.
Experimental Protocols for TRL 4 Validation Studies
Comprehensive Inter-Laboratory Comparison Design
A representative example of TRL 4 research in forensic science is demonstrated in a 2025 study published in Forensic Chemistry titled "Improving inter-laboratory comparability of tooth enamel carbonate stable isotope analysis (δ13C, δ18O)" [6]. This study exemplifies the systematic approach required for establishing method reliability across multiple laboratories.
The experimental protocol involved:
- Sample Selection and Preparation: Ten "modern" faunal teeth obtained from field recoveries were selected as test samples. Enamel powder subsamples were prepared using standardized protocols across participating laboratories [6].
- Variable Testing: The study systematically compared the effects of multiple methodological variables:
- Chemically pretreated versus untreated samples
- Standardized versus non-standardized acid reaction temperatures
- Samples analyzed with and without baking to remove moisture before analysis [6]
- Data Analysis: Isotopic δ values (δ13C and δ18O) generated by different laboratories were compared using statistical methods to identify systematic differences and their causes [6].
This experimental design allowed researchers to identify that "δ values from the two laboratories were systematically different when samples were chemically pretreated, but that differences were smaller or negligible for untreated samples" [6]. Such findings are crucial for establishing standardized protocols that minimize inter-laboratory variability.
Methodologies for Forensic Chemistry Applications
In forensic chemistry applications such as comprehensive two-dimensional gas chromatography (GC×GC), TRL 4 validation requires specific experimental approaches:
- Intra- and Inter-laboratory Validation: Conducting rigorous testing within a single laboratory followed by collaborative trials across multiple laboratories to establish reproducibility [2].
- Error Rate Analysis: Quantifying methodological uncertainty and potential sources of error through controlled experiments and statistical analysis [2].
- Standardization Development: Creating detailed protocols that can be implemented across different laboratory settings with different instrument configurations [2].
Table 2: Key Experimental Components for TRL 4 Forensic Validation
| Component | Protocol Requirements | Validation Metrics | Outcome Measures |
|---|---|---|---|
| Inter-laboratory Testing | Identical samples analyzed across multiple laboratories using standardized protocols | Statistical comparison of results (e.g., ANOVA, t-tests) | Establishment of reproducibility limits and systematic biases |
| Error Rate Analysis | Controlled introduction of known variables and potential interferents | Quantification of false positive/negative rates, measurement uncertainty | Defined confidence intervals for analytical results |
| Method Robustness | Deliberate variations in analytical conditions (temperature, timing, reagents) | Determination of critical parameters affecting results | Established tolerances for methodological variables |
| Reference Materials | Development and characterization of standardized control materials | Consistency in measurement across laboratories and over time | Quality control framework for ongoing method implementation |
Visualization of TRL 4 Advancement Pathway
The following diagram illustrates the progression from TRL 3 to TRL 4 in forensic contexts and the key components required for validation:
Diagram 1: TRL 4 Advancement Pathway in Forensic Science
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Materials for TRL 4 Forensic Validation Studies
| Item | Function in TRL 4 Research | Application Examples |
|---|---|---|
| Reference Standard Materials | Provide calibrated benchmarks for inter-laboratory comparison and method validation | Characterized control samples with known properties for instrument calibration [6] |
| Certified Reference Materials | Establish traceability and accuracy in quantitative analyses | Materials with certified isotopic compositions or chemical concentrations [6] |
| Standardized Chemical Reagents | Ensure consistency in sample preparation and treatment across laboratories | High-purity acids, solvents, and derivatization agents with specified lot-to-lot consistency [6] |
| Stable Isotope Standards | Enable comparative analysis of isotopic ratios across different instrumental platforms | Internationally recognized isotopic reference materials for forensic isotope analysis [6] |
| Quality Control Materials | Monitor analytical performance over time and across different laboratory environments | Control samples analyzed repeatedly to establish method precision and reproducibility [6] |
Comparative Performance Data for TRL 4 Methods
Quantitative Assessment of Method Performance
TRL 4 validation requires comprehensive quantitative data demonstrating method performance across multiple laboratories. The tooth enamel carbonate stable isotope study provides exemplary data for such assessment:
Table 4: Performance Comparison of TRL 4 Validated Method Versus Pre-Validation State
| Performance Metric | Pre-TRL 4 (Single Laboratory) | Post-TRL 4 (Multi-Laboratory Validated) | Improvement |
|---|---|---|---|
| Inter-laboratory Variability (δ13C) | Significant systematic differences between laboratories (e.g., up to 0.5‰) [6] | Reduced differences (e.g., < 0.1‰) through protocol standardization [6] | >80% reduction in systematic bias |
| Effect of Chemical Pretreatment | Introduced measurable bias in isotopic measurements [6] | Elimination of pretreatment-induced variability through protocol modification [6] | Removal of significant error source |
| Data Comparability | Limited due to methodological heterogeneity [6] | Enabled through standardized protocols and elimination of unnecessary steps [6] | Establishment of reliable cross-study comparisons |
| Method Robustness | Susceptible to variations in sample preparation protocols [6] | Resilient to minor variations in implementation across laboratories [6] | Enhanced reproducibility across different operational environments |
Legal Admissibility Considerations
For forensic methods, TRL 4 validation directly addresses key legal admissibility criteria:
- Testing and Error Rates: TRL 4 research explicitly quantifies methodological uncertainty and error rates through inter-laboratory studies, satisfying Daubert requirements [2].
- Peer Review and Publication: Research at this level is typically published in peer-reviewed journals, providing independent scientific validation [2] [6].
- General Acceptance: Inter-laboratory validation across multiple institutions demonstrates growing acceptance within the relevant scientific community [2].
- Standardization: Development of standardized protocols supports consistent application across forensic laboratories, enhancing reliability [2].
Technology Readiness Level 4 represents a critical transition point in forensic method development where techniques progress from single-laboratory applications to multi-laboratory validated protocols ready for implementation in casework. The defining characteristics of TRL 4 research include systematic inter-laboratory comparison, rigorous error rate analysis, and development of standardized protocols that can be consistently applied across different forensic laboratory environments.
The experimental approaches and validation methodologies required at TRL 4 directly address the legal standards for admissibility of scientific evidence in judicial proceedings, particularly the Daubert Standard and Federal Rule of Evidence 702 in the United States. By establishing method reliability through collaborative validation studies, TRL 4 research provides the necessary foundation for forensic techniques to withstand legal scrutiny while producing scientifically robust evidence. As forensic science continues to evolve toward more quantitative, data-driven approaches [7], the rigorous validation standards embodied by TRL 4 will become increasingly essential for maintaining and enhancing the quality and reliability of forensic evidence in the justice system.
For researchers and scientists developing novel forensic methods, navigating the legal standards for evidence admissibility is a critical final step in the technology transfer pipeline. The admissibility of expert testimony in U.S. courts is governed primarily by two competing standards: the Frye standard established in 1923 and the Daubert standard from 1993, with Federal Rule of Evidence 702 providing the statutory framework for federal courts [8]. For forensic methods at Technology Readiness Level (TRL) 4—where experimental prototypes have been validated in a laboratory environment—understanding these legal frameworks during study design is paramount for eventual courtroom acceptance [2].
Recent amendments to Federal Rule of Evidence 702, effective December 2023, have clarified that the proponent of expert testimony must demonstrate to the court that "it is more likely than not that" the testimony meets all admissibility requirements [9]. This heightened emphasis on the judge's gatekeeping role makes rigorous inter-laboratory validation studies essential for novel forensic techniques like comprehensive two-dimensional gas chromatography (GC×GC) and other analytical methods being developed for forensic applications [2].
Comparative Analysis of Legal Standards
The Frye Standard: General Acceptance Test
The Frye standard, derived from Frye v. United States (1923), establishes that expert testimony is admissible only if the scientific technique on which the opinion is based is "generally accepted" as reliable in the relevant scientific community [10]. This standard essentially makes the scientific community the gatekeeper of evidence admissibility, with courts considering the issue once and not revisiting it in subsequent cases after establishing general acceptance [11].
Under Frye, novel scientific methods that produce "good science" may be excluded if they have not yet reached the level of general acceptance within their field [11]. Conversely, techniques that are generally accepted but poorly applied in a specific case ("bad science") will likely still be admitted, with challenges going to the weight rather than admissibility of the evidence [8].
The Daubert Standard and Federal Rule 702
The Daubert standard, established in Daubert v. Merrell Dow Pharmaceuticals, Inc. (1993), significantly expanded the judge's role as evidentiary gatekeeper [12]. Daubert held that Rule 702 of the Federal Rules of Evidence superseded Frye's general acceptance test, requiring trial judges to ensure that proffered expert testimony rests on a reliable foundation and is relevant to the case [13].
The Daubert decision provided a non-exclusive checklist of factors for trial courts to consider [14]:
- Whether the theory or technique can be and has been tested
- Whether it has been subjected to peer review and publication
- Its known or potential error rate
- The existence and maintenance of standards controlling its operation
- Whether it has attracted widespread acceptance in a relevant scientific community
The 2000 and 2023 amendments to Federal Rule of Evidence 702 codified and clarified these principles, emphasizing that judges must evaluate whether the proponent has demonstrated by a preponderance of the evidence that: (a) the expert is qualified; (b) the testimony is based on sufficient facts or data; (c) the testimony is the product of reliable principles and methods; and (d) the expert has reliably applied the principles and methods to the facts of the case [14] [9].
Jurisdictional Application of Standards
| Jurisdiction Type | Governing Standard | Key Characteristics |
|---|---|---|
| Federal Courts | Daubert + FRE 702 [12] | Judges act as active gatekeepers; flexible, multi-factor analysis [8] |
| Daubert States (Majority) | Daubert/Modified Daubert [11] | Variations include "Shreck/Daubert" (CO), "Porter/Daubert" (CT) [11] |
| Frye States (Minority) | Frye Standard [11] | CA, IL, PA, WA; focuses primarily on "general acceptance" [13] |
| Hybrid Jurisdictions | Mixed Standards [11] | NJ applies different standards depending on case type [11] |
Table 1: Jurisdictional application of expert testimony admissibility standards across United States courts.
Implications for TRL 4 Forensic Research Design
Designing Inter-Laboratory Studies for Legal Admissibility
For forensic methods at TRL 4—where technology components are validated as laboratory prototypes—inter-laboratory validation studies must be designed with specific legal admissibility criteria in mind [2]. Research indicates that GC×GC and other novel forensic applications face significant hurdles in courtroom implementation due to strict legal standards, despite their analytical advantages [2].
A comprehensive review of forensic applications using GC×GC noted that "future directions for all applications should place a focus on increased intra- and inter-laboratory validation, error rate analysis, and standardization" to meet legal admissibility requirements [2]. This aligns directly with Daubert factors emphasizing known error rates and maintenance of standards.
Experimental Protocols for Legal Readiness
Error Rate Determination: Under Daubert, courts consider "the known or potential rate of error" of a technique [12]. TRL 4 research should incorporate protocols that quantitatively assess method reliability across multiple laboratories. For example, a recent inter-laboratory comparison of tooth enamel carbonate stable isotope analysis (δ13C, δ18O) implemented a systematic comparison of isotope delta values measured in two different laboratories, evaluating variations across pretreatment protocols and analytical conditions [6].
Standardization Protocols: The existence and maintenance of standards controlling a technique's operation is another key Daubert factor [12]. Research should establish standardized protocols that can be consistently applied across laboratory environments. The tooth enamel study demonstrated that standardization of acid reaction temperature and baking improved inter-laboratory comparability, while chemical pretreatment introduced unnecessary variability [6].
Blinded Testing: Incorporating blinded testing procedures across multiple laboratories helps establish whether a technique can be tested objectively—another Daubert consideration [12]. This approach minimizes contextual bias and demonstrates methodological rigor.
Data Transparency: Complete documentation of all methodological variations, statistical analyses, and raw data supports peer review and scientific acceptance. The tooth enamel study made their data and R code publicly available on GitHub, facilitating transparency and further validation [6].
The Scientist's Toolkit: Essential Materials for Admissibility-Focused Research
| Research Component | Function in Validation | Relevance to Legal Standards |
|---|---|---|
| Inter-laboratory Protocols | Standardized procedures across multiple labs | Demonstrates "existence of standards" (Daubert) [12] |
| Reference Materials | Certified materials with known properties | Establishes methodology reliability and testing capability [12] |
| Statistical Analysis Packages | Quantify error rates and variability | Addresses "known or potential error rate" (Daubert) [12] |
| Blinded Sample Sets | Controls for analyst bias during testing | Supports objective testability requirement [12] |
| Data Transparency Platforms | Share raw data and analytical code | Facilitates peer review and scientific acceptance [2] |
Table 2: Essential research components for designing TRL 4 validation studies that address legal admissibility criteria.
Case Study: GC×GC Forensic Applications at TRL 4
Comprehensive two-dimensional gas chromatography (GC×GC) represents an illustrative case study of advanced analytical techniques navigating the path toward courtroom admissibility. Current research on GC×GC use for forensic applications was summarized and reviewed for analytical advances and technology readiness, with seven forensic chemistry applications categorized into technology readiness levels based on current literature [2].
These applications face significant admissibility hurdles despite their analytical advantages. As noted in the research, "routine evidence analysis in forensic science laboratories does not currently use GC×GC–MS as an analytical technique due to strict criteria set by legal systems that limit the entrance of scientific expert testimony into a legal proceeding" [2]. This challenge is particularly relevant for analytical chemists developing new methods, as "the standards required of research for eventual admission into the legal system are not set by scientists but rather other stakeholders in the legal system" [2].
Visualizing the Legal-Admissibility Pathway for Forensic Research
Diagram 1: Legal-admissibility pathway for TRL 4 research.
The pathway from laboratory validation to courtroom admissibility for novel forensic methods requires strategic research design that explicitly addresses the legal standards of the relevant jurisdiction. For TRL 4 research, this means designing inter-laboratory studies that not only establish analytical validity but also specifically generate the evidence needed to satisfy Daubert factors or the Frye general acceptance test.
Researchers should prioritize error rate quantification, inter-laboratory standardization, robust sample sizes, and peer-reviewed publication to build the foundation for eventual expert testimony admissibility. As the 2023 amendments to Rule 702 have emphasized, the burden is squarely on the proponent of expert testimony to demonstrate its reliability by a preponderance of the evidence, making rigorous validation studies at the TRL 4 stage more critical than ever for forensic method development.
The Critical Role of Inter-Laboratory Comparisons (ILC) and Proficiency Testing (PT)
In the rigorous field of forensic science, the validation of new analytical methods is paramount to ensuring that results are reliable, reproducible, and defensible in a court of law. For methods at Technology Readiness Level (TRL) 4, characterized by the refinement and inter-laboratory validation of a standardized method ready for implementation, this process is particularly critical [15]. Within this framework, Inter-Laboratory Comparisons (ILC) and Proficiency Testing (PT) emerge as indispensable tools. According to international standards, an Inter-Laboratory Comparison (ILC) is the organization, performance, and evaluation of tests on the same or similar items by two or more laboratories under predetermined conditions. Proficiency Testing (PT), a specific type of ILC, is defined as the evaluation of participant performance against pre-established criteria [16] [17]. While the terms are often used interchangeably, a key distinction exists: PT is a formal, third-party-managed exercise that includes a reference laboratory to determine participant performance, whereas an ILC can be a simpler agreement between laboratories to compare results among themselves [17]. For forensic methods transitioning from development to operational use, these processes provide the external, objective evidence needed to demonstrate that a method is not only functional in a single laboratory but also robust and transferable across multiple facilities, thereby forming the bedrock of methodological credibility [15] [18].
The Strategic Importance of ILC/PT in Forensic Research
Participation in ILC and PT schemes offers strategic benefits that extend far beyond mere regulatory compliance. For a forensic laboratory, these activities are a cornerstone of quality assurance.
Promoting Confidence and Ensuring Compliance: Successful participation in ILC/PT promotes confidence among external stakeholders, including regulators, customers, and the legal system, as well as within the laboratory's own staff and management [16]. Furthermore, it is a direct requirement for accreditation to international standards such as ISO/IEC 17025 [19]. For forensic evidence, which can directly impact individual liberties and legal outcomes, this external validation is not just beneficial—it is essential [18].
Assessing and Improving Laboratory Competence: ILC/PT provides an unparalleled, holistic assessment of a laboratory's entire testing process. It simultaneously evaluates all factors influencing a test result, including the validity of methods, the adequacy of equipment, the correctness of data handling, and the competence of personnel [19]. This comprehensive check offers laboratories an early warning of potential measurement problems, allowing for corrective actions before casework is compromised [20].
Supporting Method Validation and Uncertainty Estimation: From a TRL 4 research perspective, ILC/PT data is vital for method validation. It helps demonstrate method precision, accuracy, and robustness across different laboratory environments [16]. The results provide valuable data for comparing results obtained from different methods and are crucial for the realistic estimation of measurement uncertainty by revealing laboratory-specific bias and generating reproducibility standard deviations that account for all known and unknown sources of error [16] [19].
Cost-Benefit Analysis: The cost of participating in a proficiency test is typically only a few hundred euros. When weighed against the potentially catastrophic costs of unreliable forensic results—which can include miscarriages of justice, loss of reputation, and massive litigation—the investment is overwhelmingly justified [19].
The following diagram illustrates the logical relationship between the core concepts of ILC/PT and their critical outcomes in a forensic research context.
Experimental Protocols for ILC/PT in Validation Studies
Designing and executing a robust ILC or PT study for a forensic method at TRL 4 requires meticulous planning and adherence to established protocols. The process can be broken down into three key phases, with specific considerations for method validation at this stage.
Phase 1: Pre-Testing Preparation and Planning
- Developing the PT Plan: Laboratories must develop and document a formal PT plan. For accreditation, this often entails a four-year plan to ensure annual participation and adequate coverage of the laboratory's full scope of accreditation within the cycle [16]. For a TRL 4 method validation study, the plan should specify the number of participating laboratories, the homogeneity and stability testing of test items, and the statistical model for evaluation.
- Enrollment and Sample Selection: Laboratories must enroll in a CMS-approved PT program (if applicable) or, for a novel method, establish a collaborative agreement with other laboratories [21]. The test materials must be homogeneous, stable, and mimic real casework samples as closely as possible. For a shooting distance determination test, for example, this might involve preparing a series of controlled specimens at various known distances [15].
- Defining Objective Performance Criteria: Before testing begins, a validation plan must define the objective performance criteria for the method. This framework, crucial for developmental validation, should address parameters such as specificity, sensitivity, reproducibility, and false-positive and false-negative rates [18].
Phase 2: Sample Processing and Testing
- Routine Sample Handling: A cardinal rule of PT is that the proficiency test samples must be processed in the same manner as routine casework samples to the extent possible [21]. This means using the same methods, personnel, equipment, and data handling procedures. Testing should be rotated among all analysts who normally perform the test.
- Avoiding Unusual Practices: Laboratories must refrain from repeating tests on PT samples unless such repetition is standard operating procedure for patient or casework samples. Furthermore, inter-laboratory communication regarding the PT samples is prohibited until after the results submission deadline [21].
- Blind Testing: For a rigorous internal validation, the use of blind samples—where the analyst is unaware that the sample is part of a test—can provide the most unbiased assessment of routine performance.
Phase 3: Results Analysis and Reporting
- Data Collection and Submission: Participants submit their results to the coordinating body according to the prescribed format and timeline. For a quantitative test, this includes the measured value and its associated estimate of measurement uncertainty [17].
- Performance Evaluation (Z-Score and Eₙ Number): The coordinating body evaluates performance using statistical metrics. The Z-score indicates how many standard deviations a laboratory's result is from the consensus mean of all participants, with |Z| ≤ 2 considered satisfactory. The Normalized Error (Eₙ) compares the participant's result to the reference value, considering the uncertainty of both, with |Eₙ| ≤ 1 indicating satisfactory performance [17].
- Internal Corrective Action: If unsatisfactory or questionable results are obtained (|Z| ≥ 3 or |Eₙ| > 1), the laboratory must initiate a root cause analysis and implement a corrective action plan. This is a fundamental part of the quality improvement cycle and is essential for demonstrating a commitment to reliability [17].
The workflow for a typical PT scheme, from preparation to corrective action, is visualized below.
Performance Evaluation and Data Analysis
The quantitative data generated through ILC/PT programs are analyzed using standardized statistical methods to provide an objective measure of a laboratory's performance. The two primary metrics used are the Z-score and the Normalized Error (Eₙ).
Table 1: Key Statistical Metrics for Evaluating ILC/PT Results
| Metric | Calculation Formula | Performance Interpretation | Primary Use Case |
|---|---|---|---|
| Z-Score | ( Z = \frac{x{lab} - X}{\sigma} ) Where ( x{lab} ) is the lab's result, ( X ) is the assigned value (e.g., consensus mean), and ( \sigma ) is the standard deviation for proficiency assessment. | Satisfactory: |Z| ≤ 2 Questionable: 2 < |Z| < 3 Unsatisfactory: |Z| ≥ 3 | Comparing a laboratory's result to the population of all participants to identify outliers. |
| Normalized Error (Eₙ) | ( En = \frac{x{lab} - x{ref}}{\sqrt{U{lab}^2 + U{ref}^2}} ) Where ( x{lab} ) and ( x{ref} ) are the lab and reference values, and ( U{lab} ) and ( U_{ref} ) are their expanded uncertainties. | Satisfactory: |Eₙ| ≤ 1 Unsatisfactory: |Eₙ| > 1 | Determining conformance when the reference value and both participants' uncertainties are known and reliable. |
The power of ILC/PT data extends beyond a simple pass/fail grade. For a TRL 4 research project, analyzing results across multiple laboratories allows for the determination of key method performance characteristics, such as the method's repeatability standard deviation (within-lab precision) and reproducibility standard deviation (between-lab precision) [19]. This data is indispensable for defining the reportable range of the method and understanding its limitations under different operating conditions, as required for a rigorous developmental validation [18].
Essential Research Reagent Solutions for ILC/PT
The successful execution of an ILC/PT study, particularly for validating novel forensic methods, relies on a suite of essential materials and reagents. These components ensure the integrity of the test and the validity of the resulting data.
Table 2: Key Materials and Reagents for Forensic ILC/PT Studies
| Item Category | Specific Examples | Critical Function in ILC/PT |
|---|---|---|
| Homogeneous Test Materials | Certified Reference Materials (CRMs), synthetic saliva/drug mixtures, fortified substrates, controlled gunshot residue patterns [15]. | Serves as the consistent, stable, and uniform test item circulated among participants; fundamental for a fair comparison of results. |
| Calibration Standards | Pure analyte standards, internal standards, calibration solutions traceable to national metrology institutes. | Ensures the traceability and accuracy of all measurements performed by participating laboratories. |
| Specialized Assay Components | Specific primers and probes for DNA/RNA targets, antibodies for immunoassays, enzymes, buffers, and extraction kits. | Enables the specific detection, identification, and quantification of the target analytes (e.g., drugs, explosives, biological agents). |
| Quality Control Materials | Positive, negative, and sensitivity controls. | Run concurrently with PT samples to monitor the correct performance of the assay and instrument stability throughout the testing event. |
For forensic methods at TRL 4, standing on the precipice of implementation, Inter-Laboratory Comparisons and Proficiency Testing are not optional exercises but fundamental components of a robust validation framework. They provide the critical, external evidence required to transition a method from a research prototype to an operational tool that can withstand legal scrutiny. Through structured experimental protocols and rigorous data analysis using metrics like Z-scores and the Eₙ number, ILC/PT delivers an objective assessment of a method's precision, accuracy, and reproducibility across multiple laboratory environments. By participating in these programs, forensic researchers and laboratory managers can confidently demonstrate the reliability of their results, fulfill accreditation requirements, and, most importantly, uphold the integrity of the justice system.
Establishing the Scope and Objectives for a TRL 4 Validation Study
Technology Readiness Levels (TRL) are a systematic metric used to assess the maturity of a particular technology. The scale runs from TRL 1 (basic principles observed) to TRL 9 (actual system proven through successful mission operations). TRL 4 represents a critical stage where component validation is performed in a laboratory environment. According to NASA's definition, this level is achieved when a proof-of-concept technology is ready and "multiple component pieces are tested with one another" [22]. In forensic science, this stage bridges foundational research and practical application, establishing that an analytical method functions correctly as an integrated system before advancing to more complex testing environments.
Reaching TRL 4 is particularly significant for forensic methods due to the stringent legal admissibility standards they must eventually meet. At this stage, the scientific research transitions from speculative investigation to practical application, setting the foundation for eventual implementation in casework [2]. For techniques like comprehensive two-dimensional gas chromatography (GC×GC), which is being explored for forensic applications including illicit drug analysis, toxicology, and fire debris analysis, TRL 4 validation provides the initial laboratory evidence that the method can deliver reliable, reproducible results under controlled conditions [2]. This stage establishes the groundwork for the more rigorous inter-laboratory studies required at higher TRLs.
Defining the Scope of a TRL 4 Validation Study
Core Components of Scope
The scope of a TRL 4 validation study must be carefully delineated to demonstrate that the method is "fit for purpose" while acknowledging the limitations of this development stage. The scope should explicitly define the boundaries of the validation, including the specific forensic applications, sample types, and analytical ranges covered. For a GC×GC method, this might include defining the specific compound classes it can detect, the concentration ranges validated, and the sample matrices tested [2].
A properly scoped TRL 4 study also identifies what falls outside its current parameters. While the method should be tested with forensically relevant materials, it may not yet address all the complexities of real casework evidence. The UK Government's guidance on method validation in digital forensics emphasizes that "data for all validation studies have to be representative of the real life use the method will be put to," but at TRL 4, this may involve controlled samples that approximate, rather than perfectly replicate, actual forensic evidence [23]. The scope should clearly state that the validation occurs in a laboratory environment and may not yet account for all the variables encountered in operational forensic settings.
Technology Readiness in Context
Understanding where TRL 4 sits in the broader technology development pathway helps clarify its appropriate scope. The table below outlines the progression from basic research to operational implementation:
Table: Technology Readiness Levels for Forensic Methods
| TRL | Stage Description | Key Activities | Forensic Context |
|---|---|---|---|
| TRL 1-2 | Basic principles observed and formulated | Fundamental research; practical applications conceived | Exploring feasibility of new analytical techniques [22] |
| TRL 3 | Active research and design initiated | Analytical and laboratory studies; proof-of-concept model construction | Experimental proof of concept for forensic application [22] [2] |
| TRL 4 | Component validation in laboratory environment | Multiple component pieces tested together; basic functionality established | Integrated testing of analytical method with controlled forensic samples [22] |
| TRL 5-6 | Validation in relevant environment | Rigorous testing in simulated conditions; prototype development | Testing with realistic forensic evidence; establishing error rates [22] [2] |
| TRL 7-9 | System demonstration in operational environment | Field testing; method qualification; implementation in real cases | Courtroom admissibility; use in casework [22] [2] |
Establishing Core Objectives for TRL 4 Validation
Primary Validation Objectives
The objectives of a TRL 4 validation study should focus on generating objective evidence that the method performs reliably for its intended purpose. The UK Government's validation guidance emphasizes that "validation involves demonstrating that a method used for any form of analysis is fit for the specific purpose intended, i.e. the results can be relied on" [23]. At TRL 4, this translates to several key objectives:
First, the study must demonstrate that all integrated components of the analytical system function together correctly. For a GC×GC-MS method, this would involve verifying that the modulator, columns, detector, and data processing software work seamlessly as a system to produce reliable chromatographic separations [2]. Second, the study should establish basic performance characteristics under controlled laboratory conditions, including sensitivity, specificity, and reproducibility for the target analytes. Third, the validation should identify any significant limitations or failure modes of the method within the tested parameters.
Analytical Performance Metrics
At TRL 4, specific performance metrics should be established to quantitatively evaluate the method. These metrics form the basis for assessing whether the method meets its intended purpose and provide benchmarks for comparison with existing methods. The validation should employ a validation matrix that clearly links performance characteristics with specific metrics, graphical representations, and validation criteria [24].
Table: Essential Performance Metrics for TRL 4 Validation
| Performance Characteristic | Recommended Metrics | TRL 4 Acceptance Criteria | Measurement Approach |
|---|---|---|---|
| Accuracy | Cllr (Log-likelihood ratio cost) | Minimum acceptable value established | Comparison of method results with known ground truth [24] [25] |
| Discriminating Power | EER (Equal Error Rate), Cllrmin | Maximum acceptable error rate defined | Ability to distinguish between similar and non-similar sources [24] |
| Calibration | Cllrcal | Threshold for calibration quality set | Agreement between calculated likelihood ratios and ground truth [24] |
| Robustness | Variation in Cllr, EER under modified conditions | Acceptable performance range established | Testing with deliberate variations in method parameters [24] [26] |
| Reproducibility | Percentage correct decisions, AUC (Area Under Curve) | Minimum reproducibility standard defined | Repeated testing across multiple runs and analysts [25] [26] |
Experimental Protocols for TRL 4 Validation
Core Validation Protocol
A robust TRL 4 validation protocol should be designed to stress test the method under conditions that challenge its reliability while remaining within laboratory parameters. The experimental design must incorporate appropriate controls and reference materials to generate meaningful validation data. The protocol should include:
Controlled Sample Analysis: Testing the method with samples of known composition that represent the expected range of forensic evidence. For drug analysis, this might include certified reference materials at various concentrations in appropriate matrices [2]. The dataset should be carefully designed to "include data challenges that can stress test the method" without overwhelming it with unrealistic complexity at this development stage [23].
Systematic Variation of Critical Parameters: A key objective at TRL 4 is understanding how the method performs when operating conditions change slightly. As outlined in chromatography validation literature, this involves "deliberate variations in procedural parameters listed in the documentation" such as mobile phase composition, temperature, or instrumental settings [26]. This systematic approach helps establish the method's robustness and identifies which parameters require strict control.
Experimental Workflow
The following diagram illustrates the typical experimental workflow for a TRL 4 validation study in forensic science:
TRL 4 Experimental Workflow
Robustness Testing Design
Robustness testing is a critical component of TRL 4 validation that investigates a method's capacity to remain unaffected by small, deliberate variations in method parameters. According to chromatographic validation literature, "robustness traditionally has not been considered as a validation parameter in the strictest sense because usually it is investigated during method development" [26]. However, at TRL 4, formal robustness testing becomes essential.
Effective robustness studies employ multivariate experimental designs rather than one-variable-at-a-time approaches. These designs efficiently identify which factors significantly affect method performance. Common approaches include:
- Full Factorial Designs: All possible combinations of factors are tested (practical for up to 5 factors)
- Fractional Factorial Designs: A carefully chosen subset of factor combinations is tested (efficient for larger numbers of factors)
- Plackett-Burman Designs: Highly efficient screening designs where only main effects are of interest [26]
For a chromatographic method, typical factors to vary might include mobile phase composition, pH, flow rate, temperature, and detection wavelength. The results from robustness testing help establish system suitability parameters and define the operational boundaries for the method.
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful TRL 4 validation requires specific materials and reference standards to ensure the reliability and relevance of the validation data. The following table outlines essential research reagent solutions for forensic method validation:
Table: Essential Research Reagent Solutions for TRL 4 Validation
| Category | Specific Examples | Function in TRL 4 Validation | Forensic Relevance |
|---|---|---|---|
| Certified Reference Materials | Certified drug standards, controlled substance analogs, metabolite standards | Provide ground truth for method accuracy assessment; enable quantification and identification verification [2] [23] | Essential for validating methods against known standards with established properties |
| Quality Control Materials | Internal standards, system suitability test mixtures, proficiency test materials | Monitor method performance during validation; detect instrumental drift or performance issues [23] [26] | Ensure consistent method performance across validation experiments |
| Sample Matrices | Synthetic bodily fluids, fortified substrates, simulated casework samples | Test method performance with forensically relevant materials without operational evidence [2] [23] | Bridge between clean standards and complex real-world evidence |
| Data Quality Tools | Validation software, statistical packages, likelihood ratio calculation tools | Quantify performance metrics; calculate error rates; support objective decision making [24] [25] | Enable statistical rigor required for admissibility standards |
| Chromatographic Supplies | GC×GC columns, modulators, liners, septa, specialty gases | Ensure system components meet specification; test method with different column batches [2] [26] | Critical for separation science methods common in forensic chemistry |
Successful completion of a TRL 4 validation study represents a significant milestone in forensic method development. It transforms a proof-of-concept into a laboratory-validated integrated system with documented performance characteristics and recognized limitations. The data generated at this stage provides the evidentiary foundation for deciding whether to advance the method to higher TRLs, where it will face more rigorous testing in forensically relevant environments.
The scope and objectives established at TRL 4 directly support subsequent validation stages. The performance metrics, robustness data, and operational boundaries defined at this level inform the design of TRL 5-6 studies, which focus on testing the method with realistic forensic evidence and establishing known error rates [2]. By thoroughly addressing the component validation objectives at TRL 4, researchers create a robust platform for the inter-laboratory studies and eventual implementation needed to meet legal admissibility standards such as Daubert and Frye [2] [27].
Building the Blueprint: A Step-by-Step Study Design Protocol
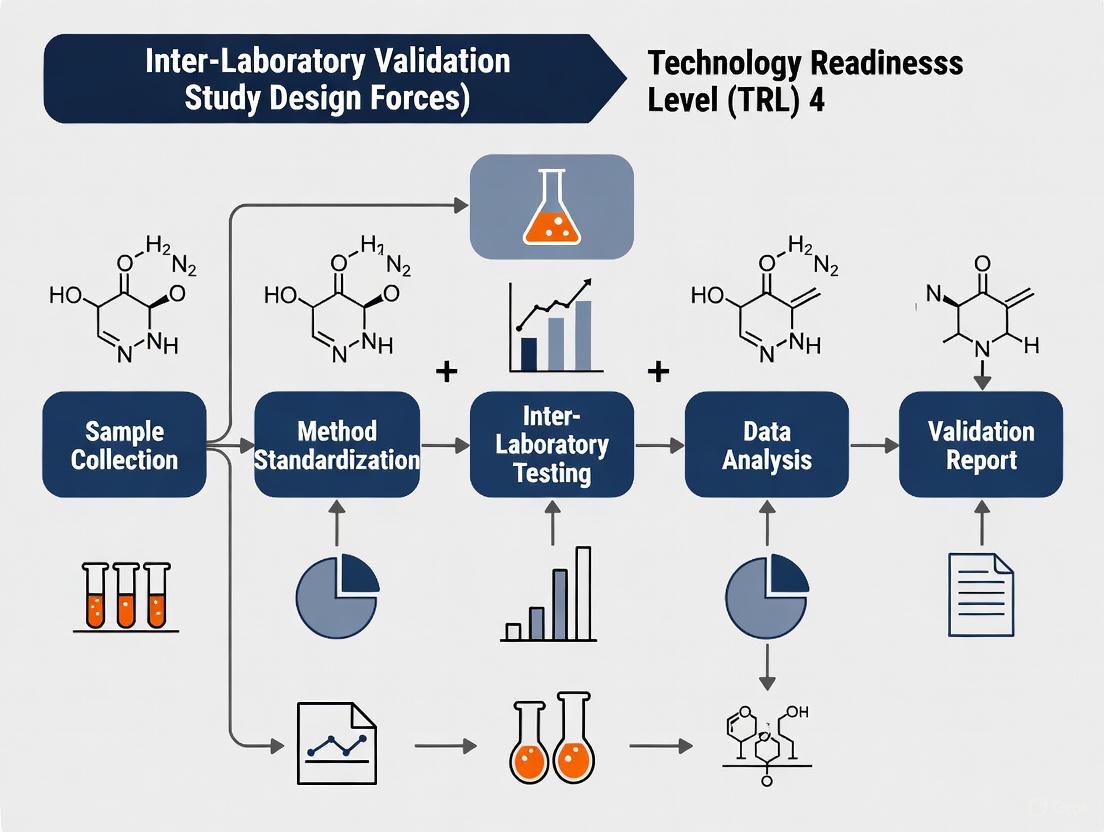
Selecting Participating Laboratories and Defining Sample Logistics
The transition of a forensic analytical method from research to routine casework is a critical juncture. For methods at Technology Readiness Level 4, defined as the refinement, enhancement, and inter-laboratory validation of a standardized method ready for implementation, this transition is predicated on robust inter-laboratory validation studies [3]. The design of these studies, particularly the selection of participating laboratories and the definition of sample logistics, forms the bedrock of generating defensible, reliable, and legally admissible data. This guide objectively compares different approaches to these core design elements, providing a framework for researchers to build studies that meet the stringent requirements of the legal system, including the Daubert Standard and Federal Rule of Evidence 702, which emphasize testing, known error rates, and standardisation [2].
Laboratory Selection Frameworks
The choice of laboratories for a validation study directly impacts the generalizability and acceptance of the results. A poorly selected laboratory cohort can introduce bias and limit the perceived applicability of the method.
Comparative Approaches to Laboratory Selection
The table below outlines three primary models for laboratory selection, comparing their objectives, implementation, and suitability for TRL 4 research.
Table 1: Objective Comparison of Laboratory Selection Frameworks for Validation Studies
| Selection Framework | Primary Objective | Implementation Strategy | Key Performance Metrics | Suitability for TRL 4 |
|---|---|---|---|---|
| Representative Sampling | To reflect the operational conditions and resource levels of the target community of forensic labs. | Recruit labs based on stratified sampling (e.g., by size, funding, geographic location). | Demographics of participating labs; diversity of instrument platforms. | High. Provides data on real-world robustness and implementation ease [3]. |
| Expert Performance-Based | To establish the upper limits of method performance under optimal, expert conditions. | Select labs with proven expertise and state-of-the-art instrumentation in the specific method domain. | Sensitivity; specificity; rate of inconclusive decisions; adherence to protocol [28]. | Medium. Essential for initial benchmark setting but may overestimate typical lab performance. |
| Census-Based Invitation | To achieve maximum uptake and demonstrate broad community consensus. | Invite all accredited forensic laboratories within a jurisdiction or network to participate. | Participation rate as a percentage of the total invited lab population. | Medium-High. Builds widespread acceptance but may be resource-intensive [2]. |
Experimental Protocols for Laboratory Evaluation
Prior to final selection, a lab qualification protocol is recommended. This involves:
- Pre-Study Questionnaire: Distributing a detailed survey to potential labs to catalog their capabilities, including instrument types and models (e.g., GC×GC–MS configurations), analyst qualifications and experience, and current casework volumes [29].
- Method Demonstration Kit: Providing a small set of pre-characterized samples for labs to analyze using the proposed method. This verifies basic competency and instrumental compatibility before the full study begins, providing an early measure of procedural adherence [28].
Sample Logistics and Design
The design and distribution of samples are perhaps the most critical operational aspect of an inter-laboratory study. Flaws here can invalidate the entire dataset.
Sample Design Strategies
A successful sample set must challenge the method across its intended scope while being logistically feasible to produce, distribute, and analyze.
Table 2: Comparison of Sample Set Design and Logistics Models
| Aspect | Blinded Proficiency Model | Collaborative Validation Model | Tiered-Difficulty Model |
|---|---|---|---|
| Core Principle | Mimics routine proficiency testing; labs are unaware of sample identities and expected results. | Open collaboration; all participants know the sample compositions and work together to characterize method performance. | Sample set includes a gradient of difficulty, from straightforward to highly challenging samples. |
| Key Data Outputs | False positive rate; false negative rate; rates of inconclusive decisions; measures reproducibility in a "real-world" context [28]. | Reproducibility standard deviation; collaborative assessment of systematic bias (trueness). | Diagnostic sensitivity and specificity across a spectrum of realistic scenarios; identifies method limitations [29]. |
| Logistics Complexity | High. Requires secure, centralized packaging and distribution to prevent decoding. Blind coding must be impeccable. | Moderate. Simplified logistics as blinding is not required, but sample homogeneity is still critical. | High. Requires careful design and pre-testing to ensure the difficulty gradient is accurate and informative. |
| Statistical Power | Provides direct estimates of error rates suitable for courtroom testimony under the Daubert standard [2]. | Provides high-quality data on precision and trueness for method refinement. | Offers a comprehensive view of method robustness and analyst skill under varying conditions [28]. |
Experimental Protocol for Sample Logistics
- Sample Preparation & Homogeneity Testing: A single, large batch of each sample type is prepared. Random sub-samples are analyzed in replicate using a reference method to statistically confirm homogeneity. This is a non-negotiable step; without it, inter-lab variance cannot be attributed to the method or the labs themselves [29].
- Stability Testing: Samples are stored under accelerated aging conditions (e.g., elevated temperature) to ensure they remain stable for the duration of the study.
- Blinding & Randomization: Each sample is assigned a unique, random code. The sample set for each lab is assembled to include replicates and a randomized order of presentation to control for sequence effects and within-lab bias [28].
- Structured Data Reporting: Participants receive a standardized data sheet or electronic portal for reporting. This sheet must explicitly capture inconclusive responses separately from forced binary choices (match/non-match) to allow for proper data analysis according to best practices in forensic science [28].
The Scientist's Toolkit: Essential Materials for Inter-Laboratory Studies
The following reagents and materials are critical for executing a forensic chemistry inter-laboratory study, particularly for techniques like comprehensive two-dimensional gas chromatography (GC×GC).
Table 3: Key Research Reagent Solutions and Materials
| Item Name | Function/Application | Critical Specifications |
|---|---|---|
| Consecutively Manufactured Tools | Provides a source of known-match and known-non-match samples for toolmark or impression evidence studies. Essential for establishing foundational data on method discrimination [29]. | Tools (e.g., screwdrivers) from the same production batch to minimize intrinsic variation. |
| Certified Reference Materials (CRMs) | To calibrate instruments across all participating laboratories and provide a benchmark for quantifying trueness. | Independently certified purity and concentration, with a valid chain of custody. |
| Stable Isotope-Labeled Internal Standards | Used in quantitative MS-based methods (e.g., toxicology) to correct for analyte loss during sample preparation and instrument variability. | High chemical and isotopic purity; must be spectrally distinct from the target analyte. |
| Inert Sample Storage Vials | To maintain sample integrity during storage and shipping. Prevents adsorption, contamination, or degradation of volatile analytes. | Headspace vials with polytetrafluoroethylene (PTFE)-lined septa, certified for the analytes of interest (e.g., for ignitable liquid residues) [2]. |
| Modulator Cryogen & Consumables | Specific to GC×GC systems, the modulator is critical for separation. A consistent supply of consumables (e.g., liquid nitrogen, CO₂) or modulator parts is needed for methods at this technical level [2]. | Purity and supply reliability to prevent study interruptions. |
Visualizing Study Workflows
The following diagrams, defined using the DOT language and adhering to the specified color and contrast rules, illustrate the logical relationships and workflows in inter-laboratory study design.
Lab Selection and Validation Workflow
This diagram outlines the sequential process for selecting participating laboratories and validating their readiness.
Sample Logistics and Data Analysis Process
This diagram details the pathway for sample preparation, distribution, and the subsequent analysis of returned data.
TRL Progression to Legal Admissibility
This diagram shows the logical relationship between technology readiness, inter-laboratory validation, and the criteria for legal admissibility.
The development of a robust test plan for forensic methods at Technology Readiness Level (TRL) 4 requires rigorous validation frameworks to ensure scientific reliability and legal admissibility. Inter-laboratory studies at this stage must demonstrate that analytical techniques are accurate, reproducible, and fit-for-purpose within the justice system. Research in forensic science must adhere to international standards and legal precedents governing expert testimony and evidence admission [2] [30]. The ISO 21043 standard provides requirements and recommendations designed to ensure the quality of the forensic process, covering vocabulary, recovery, transport, storage of items, analysis, interpretation, and reporting [30]. This guide outlines the comprehensive test plan structure necessary for validating emerging forensic methods through multi-laboratory studies, with particular focus on materials, standardized methodologies, and data reporting protocols that meet both scientific and legal requirements.
Materials and Research Reagent Solutions
A standardized set of materials and reagents is fundamental to any inter-laboratory validation study. The consistent use of certified reference materials and quality-controlled reagents across participating laboratories minimizes variability and ensures comparable results. The following table details essential research reagent solutions for forensic method validation:
Table: Essential Research Reagent Solutions for Forensic Method Validation
| Item Name | Function/Application | Specifications/Standards |
|---|---|---|
| Certified Reference Materials (CRMs) | Calibration and quality control; provides known quantitative values for method accuracy assessment | Traceable to national/international standards; certificate of analysis with stated uncertainty |
| Internal Standards (IS) | Correction for analytical variability in mass spectrometry; improves data accuracy and precision | Stable isotope-labeled analogs of target analytes; high chemical purity (>95%) |
| Quality Control Materials | Monitoring analytical process performance; detecting systematic errors and drift | Characterized pools with established target values and acceptable ranges |
| Mobile Phase Solvents | Liquid chromatography separation; compound elution and ionization | HPLC or LC-MS grade; low UV absorbance; minimal particulate matter |
| Stationary Phase Columns | Compound separation based on chemical properties; critical for resolution and sensitivity | Specified dimensions, particle size, and surface chemistry; from reputable manufacturers |
| Derivatization Reagents | Chemical modification of analytes to enhance detection, volatility, or stability | High purity; demonstrated reaction efficiency with target compounds |
The selection of these materials must be documented with detailed specifications, including manufacturer, lot numbers, storage conditions, and expiration dates. For inter-laboratory studies, central procurement and distribution of critical reagents enhance consistency across participating sites [31].
Methodological Protocols for Inter-laboratory Validation
Experimental Design and Sample Preparation
Inter-laboratory validation studies for TRL 4 forensic methods require meticulously controlled experimental protocols to generate statistically meaningful data. The sample set should include certified reference materials, real-world case-type samples, and negative controls to comprehensively evaluate method performance. Sample preparation protocols must be explicitly detailed, including extraction methods, purification steps, and derivatization procedures where applicable. For comprehensive two-dimensional gas chromatography (GC×GC) applications, which provide advanced chromatographic separation for forensic evidence, method parameters including column selection, temperature programs, and modulation periods must be standardized across participating laboratories [2]. All protocols should specify equipment calibration procedures, acceptance criteria for quality control samples, and contingency plans for protocol deviations.
Key Validation Parameters and Testing Protocols
Forensic method validation requires systematic assessment of multiple performance parameters to establish reliability, accuracy, and robustness. The following experimental protocols outline the core validation tests required for TRL 4 inter-laboratory studies:
Table: Core Validation Parameters and Testing Methodologies
| Validation Parameter | Experimental Protocol | Acceptance Criteria | Data Reporting Requirements |
|---|---|---|---|
| Accuracy and Trueness | Analysis of certified reference materials (n≥5 replicates) and comparison to reference values; recovery studies at multiple concentration levels | Mean accuracy 85-115%; CV <15% for most analytes | Reported as percent recovery or bias; statistical significance testing |
| Precision | Intra-day (n≥5) and inter-day (n≥3 days) replication at low, medium, and high concentrations; inter-laboratory comparison | Intra-laboratory CV <15%; inter-laboratory CV <20% | CV values for each concentration level; ANOVA components of variance |
| Selectivity/Specificity | Analysis of blank matrix samples and samples with potentially interfering compounds; assessment of chromatographic resolution | No significant interference at target analyte retention times; resolution >1.5 between critical pairs | Chromatograms demonstrating separation; peak purity data |
| Linearity and Range | Analysis of calibration standards at 5-7 concentration levels across expected measurement range; triplicate measurements | R² ≥0.990; residual plots without systematic patterns | Regression equation, R² value, residual plots |
| Limit of Detection (LOD) / Limit of Quantification (LOQ) | Serial dilution of low-concentration samples; signal-to-noise ratio of 3:1 for LOD and 10:1 for LOQ | LOD/LOQ appropriate for intended application; sufficient sensitivity for casework | Justification for established limits; supporting chromatograms |
| Robustness | Deliberate, small variations in method parameters (pH, temperature, flow rate); Youden's ruggedness test | Method performance maintained within acceptable criteria under varied conditions | Experimental design matrix; results of parameter variations |
Implementation of these validation protocols across multiple laboratories provides essential data on method transferability and reliability—key factors for legal admissibility under standards such as Frye, Daubert, and Federal Rule of Evidence 702 in the United States, and the Mohan criteria in Canada [2]. These legal frameworks require that scientific techniques be generally accepted in the relevant scientific community, peer-reviewed, testable, and have known error rates [2] [31].
Data Analysis and Reporting Requirements
Statistical Analysis Framework
Inter-laboratory validation studies require sophisticated statistical analysis to evaluate method performance across multiple sites. Data should be analyzed using both descriptive statistics (mean, standard deviation, coefficient of variation) and inferential statistics (ANOVA, regression analysis, outlier tests). The use of the likelihood-ratio framework for interpretation of evidence provides a logically correct framework that is consistent with the forensic-data-science paradigm [30]. Statistical packages should be specified in the test plan, along with predetermined significance levels (typically α=0.05). Data normalization procedures should be documented, and all statistical tests should be justified based on data distribution characteristics.
Standardized Reporting Protocols
Comprehensive documentation is essential for forensic method validation. The test plan must specify standardized reporting templates that include all elements required by ISO 21043, particularly Part 4 (interpretation) and Part 5 (reporting) [30]. Reports should transparently document all procedures, software versions, logs, and chain-of-custody records [31]. Error rate analysis is particularly critical for legal proceedings and must be explicitly reported with confidence intervals [2] [31]. All reports should include statements of uncertainty for quantitative measurements and clearly distinguish between observational data and interpretive conclusions.
Workflow Visualization for Inter-laboratory Validation
The following diagram illustrates the complete workflow for developing and executing an inter-laboratory test plan for forensic method validation at TRL 4:
Legal Admissibility Criteria Mapping
The successful implementation of a forensic test plan requires alignment with legal admissibility standards. The following diagram maps the relationship between validation activities and legal criteria:
Statistical Framework for Data Analysis and Determining Consensus
The validation of forensic methods at Technology Readiness Level (TRL) 4 represents a critical juncture in the transition of analytical techniques from proof-of-concept to operational implementation. At this stage, methods undergo inter-laboratory validation to demonstrate reliability across different institutional settings, instrumentation, and personnel. The statistical frameworks used to analyze this validation data and determine consensus are fundamental to establishing the scientific rigor and legal admissibility of forensic techniques. This guide compares predominant statistical frameworks applied in TRL 4 forensic research, evaluating their performance characteristics, implementation requirements, and applicability to various evidence types.
Within forensic science, TRL 4 is defined by the refinement and inter-laboratory validation of a standardized method ready for implementation in forensic laboratories [3]. Achieving this requires robust statistical approaches to demonstrate that a method produces consistent, reproducible, and reliable results across multiple laboratories—a process essential for meeting the admissibility standards outlined in legal precedents such as Daubert and Frye [2].
Comparative Analysis of Statistical Frameworks
The following analysis compares three statistical frameworks with demonstrated applicability to forensic validation studies and consensus determination.
Table 1: Comparison of Statistical Frameworks for Data Analysis and Consensus Determination
| Framework Feature | Median Aggregation with ICC Validation [32] | Functional Linear Mixed Models (FLMM) [33] | Histogram-Based Classification [34] |
|---|---|---|---|
| Primary Application | Multi-rater evaluation systems without objective ground truth | Analysis of trial-level temporal dynamics (e.g., photometry) | Categorizing opinion distributions (e.g., survey data) |
| Core Methodology | Robust median estimation; Intraclass Correlation Coefficient (ICC2k) | Functional regression exploiting signal autocorrelation; joint confidence intervals | Bin-counting algorithm; pre-defined category thresholds |
| Consensus Metric | Inter-rater reliability (ICC2k ≥ 0.955 reported) | Statistical significance of covariate effects across time-points | Qualitative categories: Perfect Consensus, Consensus, Polarization, Clustering, Dissensus |
| Handling of Variance | Quantifies individual rater alignment via consistency metrics (R², variance) | Accounts for within-trial, between-trial, and between-animal variance | Uses bin count thresholds (T₁, T₂) to discriminate signal from noise |
| Key Performance | 67% reduction in computational cost with minimal reliability loss | Identifies significant effects obscured by trial-averaging | Captures evolution of qualitative states via transition tables |
| Technology Readiness | High (validated on ~14,384 samples) | Emerging (primarily in neuroscience) | Moderate (validated on World Values Survey data) |
| Implementation Complexity | Low to Moderate | High | Moderate |
Experimental Protocols for Framework Evaluation
The following section details the experimental methodologies employed in the cited studies to generate the performance data summarized in Table 1.
Protocol for Median Aggregation and ICC Validation
This protocol is designed to assess inter-laboratory consensus when objective ground truth is unavailable [32].
- Step 1: Data Collection. Multiple evaluators (e.g., laboratories, instruments, algorithms) analyze an identical set of samples. In the cited study, 17 Large Language Models evaluated ~14,384 samples for semantic similarity.
- Step 2: Consensus Estimation. Calculate the median value across all evaluators for each individual sample. The median provides a robust consensus estimate with a 50% breakdown point, resistant to outliers.
- Step 3: Reliability Assessment. Compute the Intraclass Correlation Coefficient (ICC2k) using the median consensus as a reference standard. The two-way random, absolute agreement model (ICC2k) is appropriate for assessing the reliability of multiple raters.
- Step 4: Core Set Optimization. Apply algorithms to select a minimal subset of evaluators that maintains high reliability (e.g., ICC > 0.95), thereby reducing computational or operational costs.
Protocol for Functional Linear Mixed Models (FLMM)
This protocol is optimized for analyzing complex, time-series data from repeated-measures experiments, common in instrumental analysis [33].
- Step 1: Model Specification. Formulate a functional linear mixed model. The model incorporates fixed effects for experimental conditions (e.g., treatment/control) and random effects to account for variability between subjects (e.g., different laboratories) and within subjects across repeated trials.
- Step 2: Hypothesis Testing. Test the association between covariates and the signal at every time-point within the trial, rather than condensing the signal into a single summary statistic.
- Step 3: Confidence Interval Construction. Exploit the inherent autocorrelation in the time-series signal to calculate joint 95% confidence intervals. These intervals account for multiple comparisons across the entire trial without being overly conservative.
- Step 4: Visualization and Interpretation. Generate plots showing covariate effect estimates and their statistical significance at each time-point, unifying hypothesis testing with data visualization.
Protocol for Histogram-Based Classification
This protocol provides a structured method for categorizing quantitative data into qualitative consensus states [34].
- Step 1: Data Binning. Partition the data range (e.g., -1 to +1 for opinion data) into M bins of equal width. For 10-point Likert scale data, M=10 is typical.
- Step 2: Bin Classification. Normalize bin counts to 100%. Classify each bin as:
- Green: > T₁% (e.g., T₁=50, indicating a majority)
- Blue: < T₂% (e.g., T₂=5, indicating residual noise)
- Red: Values between T₂ and T₁.
- Step 3: Group Formation. Define a "group" as consecutive green or red bins.
- Step 4: Category Assignment. Apply classification criteria:
- Perfect Consensus: A single green bin exists.
- Consensus: One group of ≤ B bins containing >50% of responses.
- Polarization: Two groups, separated by ≥ K bins, containing >50% of responses combined.
- Clustering: More than two groups containing >50% of responses.
- Dissensus: No grouping contains a majority.
Figure 1: Workflow for the histogram-based classification algorithm, illustrating the logical sequence from data input to final consensus category assignment [34].
The Scientist's Toolkit: Essential Reagents & Materials
Successful implementation of statistical frameworks for inter-laboratory validation requires both computational and experimental resources. The following table details key solutions and their functions.
Table 2: Key Research Reagent Solutions for Inter-Laboratory Validation
| Reagent/Material | Function in Validation Study | Example Application |
|---|---|---|
| Standardized Reference Materials | Provides a common, homogeneous sample for all participating laboratories to analyze, enabling direct comparison of results. | Ten "modern" faunal teeth used across labs in isotope analysis [6]. |
| Validated Calibrants & Controls | Ensures analytical instruments across different laboratories are producing accurate and comparable measurements. | GMP-compliant pilot lots in drug development [35]. |
| Open-Source Data Analysis Platforms (e.g., R, GitHub Code) | Promotes transparency, reproducibility, and allows all laboratories to apply the exact same statistical algorithms. | R code provided on GitHub for inter-laboratory comparison [6]. |
| Statistical Reference Datasets | Serves as a benchmark for testing and validating new statistical frameworks and software implementations. | World Values Survey data for testing opinion formation models [34]. |
| Documented Standard Operating Procedures (SOPs) | Guarantees that all sample preparation, analysis, and data collection steps are performed identically across labs. | ISO 21043 standards for forensic analysis [36]. |
The selection of an appropriate statistical framework is paramount for robust inter-laboratory validation at TRL 4. The Median Aggregation with ICC Validation framework offers a robust, computationally efficient solution for establishing consensus in subjective evaluation tasks, directly addressing legal standards for reliability and known error rates [2] [32]. For forensic disciplines generating complex temporal or spectral data, FLMM provides superior power to detect significant effects by leveraging full datasets without coarsening information [33]. Finally, the Histogram-Based Classification framework provides a transparent and intuitive method for translating quantitative results into actionable, qualitative categories, facilitating decision-making [34].
Future directions should emphasize the development of standardized, discipline-specific validation frameworks that incorporate these statistical principles, enabling more efficient adoption of novel forensic methods into operational casework.
Defining Performance Metrics and Acceptance Criteria for Success
The transition of a forensic method from research to routine casework is a critical juncture. For methods at Technology Readiness Level 4 (TRL 4), defined as the stage for "refinement, enhancement, and inter-laboratory validation of a standardized method ready for implementation in forensic laboratories," establishing robust performance metrics and acceptance criteria is the cornerstone of success [15]. The core objective of a TRL 4 validation study is to demonstrate that an analytical method is not only functionally effective but also reliable, reproducible, and legally defensible across multiple independent laboratories.
This process is governed by a stringent framework of legal and scientific standards. Before forensic evidence can be admitted in court, the underlying analytical method must satisfy specific legal precedents, such as the Daubert Standard in the United States or the Mohan Criteria in Canada [2]. These standards require that a method has been tested, subjected to peer review, has a known error rate, and is generally accepted in the scientific community [2]. Therefore, the performance metrics and acceptance criteria defined during inter-laboratory validation are not merely scientific exercises; they are essential for ensuring the method's admissibility and the integrity of subsequent justice outcomes.
Performance Metrics Framework for Forensic Validation
The validation of a forensic method requires a multi-faceted approach to performance assessment. The following metrics are universally critical for evaluating a method's fitness for purpose.
Table 1: Core Performance Metrics and Their Definitions in Forensic Validation
| Metric | Definition | Significance in Forensic Context |
|---|---|---|
| Trueness (Accuracy) | The closeness of agreement between the average value obtained from a large series of test results and an accepted reference value [37]. | Ensures that evidence is correctly identified and quantified, preventing miscarriages of justice. |
| Precision | The closeness of agreement between independent test results obtained under stipulated conditions [37]. | Can be measured as repeatability (within-lab) and reproducibility (between-lab). |
| Specificity | The ability of the method to distinguish the target analyte from other substances in a complex mixture [37]. | Critical for analyzing trace evidence or complex mixtures where contaminants may be present. |
| Limit of Detection (LOD) | The lowest concentration of an analyte that can be detected, but not necessarily quantified, under the stated experimental conditions [37]. | Defines the sensitivity of the method for analyzing minimal or degraded samples. |
| Limit of Quantification (LOQ) | The lowest concentration of an analyte that can be quantified with acceptable levels of trueness and precision [37]. | Essential for reliable quantitative analysis, such as determining drug concentrations. |
| Robustness | A measure of the method's capacity to remain unaffected by small, deliberate variations in method parameters. | Indicates the method's reliability during routine use in different laboratory environments. |
| Error Rate | The observed or estimated rate at which a method produces false positives or false negatives [2]. | A key requirement under the Daubert Standard for courtroom admissibility of evidence [2]. |
Application in Different Forensic Disciplines
The application of these core metrics varies across forensic disciplines, informing the design of inter-laboratory studies:
- Digital Forensics: For AI-driven social media analysis, studies report metrics like sensitivity (98%) and specificity (96%) in classifying relevant evidence, such as in cyberbullying or misinformation campaigns [38]. The high-dimensional data requires rigorous testing of algorithm robustness against adversarial attacks and data evolution [38].
- Chemical Forensics: In techniques like Comprehensive Two-Dimensional Gas Chromatography (GC×GC), precision is measured through the reproducibility of chromatographic profiles across labs, while specificity is demonstrated by separating co-eluting analytes in complex mixtures like illicit drugs or ignitable liquids [2].
- Toolmark and Firearm Forensics: Objective algorithmic comparisons of bullet signatures have demonstrated high performance, with one study achieving a cross-validated sensitivity of 98% and specificity of 96% [29]. Establishing the known match and known non-match densities and deriving likelihood ratios are critical for defining acceptance criteria [29].
Designing the Inter-Laboratory Validation Study
A well-designed inter-laboratory study is fundamental to generating the data required to define acceptance criteria.
Experimental Protocol for Inter-Laboratory Validation
The following workflow outlines the standard operating procedure for a TRL 4 inter-laboratory validation study, integrating best practices from forensic and bioanalytical guidelines [39] [37].
Establishing Data-Driven Acceptance Criteria
Acceptance criteria must be practical, statistically derived, and tailored to the method's intended use. A retrospective analysis of bioanalytical cross-validation studies suggests that criteria should account for inter-laboratory variability, which can arise from differences in sample preparation, reagent batches, and environmental conditions [39]. The following table provides a template for defining these criteria based on the core performance metrics.
Table 2: Template for Defining Acceptance Criteria Based on Performance Metrics
| Performance Metric | Recommended Acceptance Criterion for TRL 4 | Example from Forensic Disciplines |
|---|---|---|
| Trueness (Accuracy) | Mean recovery of 80-120% for quantitative assays; >99% true positive identification for qualitative methods. | In GMO testing, quantitative PCR methods require demonstrated trueness across the validated dynamic range [37]. |
| Precision (Reproducibility) | Relative Standard Deviation (RSD) between laboratories ≤ 15-20% for quantitative analysis. | For bioanalytical methods using LC/MS/MS, inter-lab precision is a key criterion for cross-validation success [39]. |
| Specificity | No false positives or false negatives when testing against a panel of closely related interferents. | In GC×GC for drug analysis, the method must resolve the target drug from cutting agents and metabolites [2]. |
| LOD / LOQ | Consistent detection/quantification at the claimed target concentration across all participating labs. | For DNA analysis, the LOQ must be set to ensure reliable results from low-template or degraded samples [40]. |
| Error Rate | A documented and acceptably low rate of false positives and false negatives, as required by the Daubert Standard [2]. | In objective bullet comparison, algorithms must demonstrate a false positive rate < 4% based on known non-match densities [29]. |
The Scientist's Toolkit: Essential Research Reagent Solutions
The reliability of a validated method is dependent on the quality and consistency of the materials used. The following table details key reagents and their critical functions in forensic method development and validation.
Table 3: Essential Research Reagent Solutions for Forensic Method Validation
| Reagent / Material | Function | Considerations for Inter-Laboratory Studies |
|---|---|---|
| Certified Reference Materials (CRMs) | Provides an authentic, well-characterized standard for method calibration and trueness assessment. | Must be traceable to a national or international standard. The same batch should be used by all labs in a study to minimize variability [37]. |
| Quality Control (QC) Samples | Used to monitor the precision and stability of the analytical method during a validation run. | Typically prepared at low, medium, and high concentrations covering the dynamic range of the assay [39]. |
| DNA Oligonucleotides (Primers/Probes) | Essential for PCR-based forensic methods, including DNA sequencing and quantitative PCR for GMO testing [37]. | Require validation for specificity and sensitivity. Batch-to-batch consistency is critical; a single supplier is recommended for inter-lab studies. |
| Sample Preparation Kits (e.g., DNA/RNA Extraction) | Standardizes the isolation and purification of the target analyte from a complex matrix. | Kit lot numbers and protocols should be consistent across laboratories to ensure comparable results [40]. |
| Matrix-Matched Standards | Analytical standards prepared in a sample matrix that mimics real evidence (e.g., blood, soil, food). | Accounts for matrix effects that can suppress or enhance the analytical signal, providing a more realistic measure of performance [2]. |
Case Study: Validation of AI for Social Media Forensics
The integration of artificial intelligence (AI) and machine learning (ML) into digital forensics provides a contemporary case study for TRL 4 validation. In this domain, the "experimental protocol" involves using defined datasets to test models like BERT for natural language processing (NLP) and Convolutional Neural Networks (CNNs) for image analysis [38].
- Performance Metrics: Key quantitative metrics include the accuracy, precision, and recall in classifying evidence (e.g., identifying cyberbullying language or fake news). One study demonstrated the effectiveness of these models in empirical studies on cyberbullying and fraud detection [38].
- Acceptance Criteria: Beyond pure accuracy, acceptance criteria must address algorithmic bias and model interpretability. For courtroom admissibility, it is crucial to use explainable AI (XAI) techniques, such as SHAP or LIME, to make the model's decisions transparent to judges and juries [38]. The known error rate, a Daubert requirement, must be established through rigorous testing on diverse, representative datasets to uncover and mitigate biases, particularly in facial recognition [38].
- Inter-Laboratory Challenge: A key challenge is that AI models can be "context-agnostic," potentially compromising reliability. The validation study must therefore prove that the model performs robustly across different data subsets and scenarios, as would be the case when deployed in different law enforcement agencies [38].
Defining performance metrics and acceptance criteria is the definitive step that bridges promising forensic research and its reliable application in the justice system. A meticulously designed inter-laboratory validation study, grounded in the framework presented here, generates the empirical data needed to set these criteria. By rigorously evaluating trueness, precision, error rates, and other key metrics against legally and scientifically sound benchmarks, researchers can elevate a method to TRL 4. This process ensures that new forensic technologies are not only analytically powerful but also standardized, reproducible, and ready for implementation in casework, ultimately upholding the integrity of forensic science and the legal process it serves.
Navigating Challenges: Ensuring Robustness and Reliability
Inter-laboratory variance refers to the variability in results obtained when different laboratories analyze the same samples using ostensibly the same methods. This variability presents significant challenges in forensic science, drug development, and basic research, as it can undermine the reliability, reproducibility, and comparability of scientific data. A recent inter-laboratory study by the ReAct group demonstrated considerable variability in DNA recovery between forensic laboratories, highlighting the pervasive nature of this issue [41]. Similarly, in neuroscience, methodological differences in patch-clamp electrophysiology experiments have been shown to contribute significantly to study-to-study variability in measurements of fundamental electrophysiological parameters [42].
Addressing inter-laboratory variability is particularly crucial for the validation of forensic methods at Technology Readiness Level (TRL) 4, where controlled laboratory validation establishes proof of principle. At this stage, understanding and controlling for sources of variability ensures that subsequent development and implementation across laboratories yield consistent and reliable results. The forensic community has increasingly recognized that physical fit examinations, while generally accurate, are not exempt from errors, necessitating standardized approaches to minimize potential sources of error and bias [43].
Methodological and Procedural Differences
Variability in experimental protocols and procedures represents a fundamental source of inter-laboratory differences. In patch-clamp electrophysiology, for example, a comprehensive analysis of 509 published articles revealed that "very few articles used the exact same experimental solutions as any other," with differences stemming from "recipe inheritance from advisor to advisee as well as changing trends over the years" [42]. These methodological differences can explain up to 43% of the study-to-study variance in electrophysiological parameters, leaving the majority of variability unexplained and suggesting additional unreported factors contribute significantly [42].
Forensic sciences face similar challenges, where differences in sample handling, interpretation criteria, and examination techniques can introduce variability. In duct tape physical fit examinations, factors such as "the quality grade of the tape, separation method, and level of stretching influence the edge similarity score" [43]. Without standardized protocols, these procedural differences can lead to inconsistent results between laboratories examining the same evidence.
Table 1: Common Methodological Sources of Variance Across Disciplines
| Source of Variance | Impact Area | Example from Literature |
|---|---|---|
| Solution Composition | Electrophysiology | Differences in artificial cerebrospinal fluid and internal pipette solutions [42] |
| Sample Preparation | Forensic Science | Separation method (hand-torn vs. scissor-cut) affecting duct tape edge characteristics [43] |
| Data Interpretation Criteria | Multiple Fields | Subjective assessment of physical fits without quantitative metrics [43] |
| Instrument Calibration | Multiple Fields | Inter-laboratory variability in DNA recovery efficiency [41] |
Reagent and Material Variability
The quality, composition, and source of reagents and materials contribute significantly to inter-laboratory variance. In DNA analysis, differences in recovery efficiency between laboratories present substantial challenges when one laboratory needs to use data produced by another [41]. This variability affects the ability to compare results directly and necessitates careful calibration when integrating historical data from different sources.
The impact of reagent variability extends to basic research as well. In electrophysiology, specific solution components such as "internal anions or extracellular Ca2+ and Mg2+" have been experimentally shown to influence measurements, yet these factors are typically studied in isolation within a single laboratory, making it difficult to generalize findings across different experimental contexts [42]. This problem is compounded by the fact that complete chemical dissociation is often assumed when preparing solutions, as "dissociation constants were unavailable for many chemical components at typical recording temperatures" [42].
Human Factors and Interpretation
Human expertise, training, and subjective judgment introduce another layer of variability in inter-laboratory studies. Even when following standardized protocols, differences in examiner experience and interpretation can affect outcomes. In duct tape physical fit examinations, the development of "standardized qualitative and quantitative metrics" was necessary to "support the examiner's opinion" and provide consistent results between participants [43].
The evolution of assessment methods demonstrates how structured approaches can mitigate human factors. Initial studies on duct tape physical fits showed that while analysts had relatively high accuracy rates, they were "not exempt from errors" [43]. Through inter-laboratory studies involving 38 practitioners from 23 laboratories, researchers refined examination protocols, reporting tools, and training materials, resulting in improved inter-examiner agreement and overall accuracy increasing from 95% to 99% between the first and second exercises [43].
Experimental Approaches for Identifying Variance
Interlaboratory Study Design
Well-designed interlaboratory studies represent the gold standard for identifying and quantifying sources of variability. These studies typically involve a coordination body that creates the experimental design, prepares standardized samples, and distributes them to multiple participating laboratories while maintaining blind conditions. In the forensic duct tape study, samples were prepared from medium-quality grade duct tape with hand-torn separations to create "casework-like fits and non-fits," with ground truth maintained blind to participants [43].
Effective study design requires careful consideration of sample selection and consensus values. For the duct tape studies, samples were "divided into seven groups of three similar pairs each, to prepare three distribution kits," with grouping criteria ensuring each kit contained pairs representing a range of edge similarity scores from high-confidence fits to more challenging comparisons [43]. This approach allowed for systematic assessment of examiner performance across different difficulty levels.
Statistical Frameworks for Quantifying Variance
Robust statistical analysis is essential for interpreting inter-laboratory data and distinguishing systematic differences from random variation. The duct tape studies employed both performance rates based on participant conclusions and quantitative comparison to consensus edge similarity scores (ESS) established prior to administering the studies by an independent panel [43]. By assessing ESS data using z-scores, researchers could identify participants whose results fell outside acceptable ranges, with most results being satisfactory but with "eight cautionary and two insufficient results in the first study, and seven cautionary and no insufficient results in the second trial" [43].
For DNA recovery studies, Bayesian statistical approaches have been proposed to "incorporate inter-laboratory variability within an evaluation" when calibration data between laboratories is unavailable [41]. These approaches allow evaluations to continue while ensuring "that the strength of findings is appropriately represented," even when utilizing data produced by other laboratories with different recovery characteristics [41].
Table 2: Quantitative Metrics for Assessing Inter-Laboratory Variance
| Metric | Application | Interpretation |
|---|---|---|
| Edge Similarity Score (ESS) | Duct tape physical fits | Quantitative assessment of fit quality; higher scores indicate better alignment [43] |
| Z-Scores | Method performance evaluation | Identifies results falling outside acceptable ranges relative to consensus values [43] |
| Accuracy Rates | Overall method performance | Percentage of correct identifications in ground-truth studies [43] |
| Inter-laboratory Recovery Variability | DNA analysis | Measures differences in efficiency of DNA recovery between laboratories [41] |
Mitigation Strategies and Standardization
Collaborative Method Validation
The traditional model of individual laboratories independently validating methods creates significant redundancy and inefficiency. A collaborative validation model proposes that "FSSPs performing the same task using the same technology are encouraged to work together cooperatively to permit standardization and sharing of common methodology" [44]. This approach increases efficiency while promoting higher standards across laboratories.
The collaborative model operates through a structured process: originating laboratories publish comprehensive validation data in peer-reviewed journals; subsequent laboratories adopting the exact methodology can then perform verification rather than full validation; and ongoing performance monitoring ensures continued reliability. This process "increases efficiency through shared experiences and provides a cross check of original validity to benchmarks established by the originating FSSP" [44]. The substantial cost savings of this approach can be demonstrated through "salary, sample and opportunity cost bases" [44].
Quantitative Metrics and Standardized Reporting
The development and implementation of quantitative assessment tools significantly reduce subjective interpretation variances. In duct tape physical fit examinations, the edge similarity score (ESS) provides "a metric for the quality of the fit" by estimating "a relative percentage of corresponding scrim bins along the total width of a fracture between two tapes" [43]. This objective measure creates a common framework for comparison across laboratories and examiners.
Standardized reporting templates further enhance consistency by ensuring all relevant information is documented transparently. The duct tape studies found that providing participants with "standardized reporting criteria" facilitated "consistent results between participants" and "demonstrable conclusions" [43]. The reporting template required examiners to document "bin-by-bin observations" systematically, creating a clear record supporting their conclusions and enabling meaningful peer review [43].
Calibration and Data Adjustment Methods
When inter-laboratory variability cannot be eliminated through standardization alone, calibration exercises and statistical adjustments provide alternative solutions. For DNA recovery variability, one proposed option involves laboratories carrying out "a calibration exercise so that appropriate adjustments between laboratories can be made" [41]. This approach directly addresses systematic differences in recovery efficiency.
For situations where historical data must be incorporated or calibration is impractical, statistical methods can account for inter-laboratory differences. Recent research has presented "a method to utilise data produced in other laboratories that takes into account inter-laboratory variability within an evaluation" [41]. This allows for more appropriate use of existing data while acknowledging its limitations, though incorporating such variation necessarily "reduces discrimination power" [41].
Experimental Protocols and Data Presentation
Standardized Experimental Workflow
The following workflow diagram illustrates a systematic approach to inter-laboratory study design, incorporating elements from successful implementations in forensic science [43] and collaborative method validation [44]:
Collaborative Method Validation Process
The transition from traditional to collaborative validation models involves multiple phases with distinct responsibilities, as illustrated below:
Research Reagent Solutions for Standardization
Table 3: Essential Materials and Reagents for Inter-Laboratory Studies
| Reagent/Material | Function in Standardization | Considerations for Inter-Laboratory Use |
|---|---|---|
| Standard Reference Materials | Provides uniform baseline for comparison across laboratories | Should be characterized by certifying body; stable and homogeneous [43] |
| Control Samples | Monitors analytical performance and detects drift | Include positive, negative, and sensitivity controls; same lot numbers preferred [43] |
| Standardized Solution Formulations | Reduces variability from chemical composition differences | Exact recipes with specified grades and sources of chemicals [42] |
| Quantitative Assessment Tools | Provides objective metrics replacing subjective judgment | Software tools for calculating similarity scores, statistical measures [43] |
Inter-laboratory variance presents a multifaceted challenge affecting the reliability and reproducibility of scientific data across disciplines. Through systematic identification of variability sources—including methodological differences, reagent variability, and human factors—and implementation of targeted mitigation strategies such as collaborative validation, quantitative metrics, and calibration protocols, laboratories can significantly improve consistency and comparability of results. The continued development and refinement of these approaches, particularly for forensic methods at TRL 4, will strengthen the scientific foundation of analytical techniques and enhance their utility in both research and applied settings.
Strategies for Resolving Discrepancies and Outlier Results
In forensic method validation and drug development, the integrity of analytical results is paramount. Outliers—data points that deviate markedly from other observations—can significantly skew results, leading to inaccurate conclusions, flawed method validation, and potentially compromising scientific or legal outcomes. Effectively managing these discrepancies is a critical component of inter-laboratory validation study design, particularly for Technology Readiness Level (TRL) 4 research where methods are refined and prepared for implementation [15]. The strategic handling of outliers ensures that forensic methods produce reliable, defensible data fit for purpose in legal contexts [44].
This guide compares predominant outlier management strategies, evaluating their methodological rigor, implementation requirements, and suitability for forensic and pharmaceutical research contexts. We provide experimental protocols and quantitative comparisons to guide researchers in selecting appropriate approaches for resolving discrepancies in validation studies.
Table 1: Strategic Approaches to Outlier Management
| Strategy | Key Principle | Typical Use Case | Advantages | Limitations |
|---|---|---|---|---|
| Iterative Outlier Removal (IOR) | Repeated exclusion of data points >3 SD from mean difference until no outliers remain [45] | Laboratory recalibration studies; Method alignment across datasets | Identifies more extraneous outliers than single removal; Reduces standard deviation more effectively [45] | Potential over-removal if not carefully validated; Requires multiple computational iterations |
| Single-Round Removal | Single exclusion of data points >3 SD from mean difference [45] | Initial data screening; Datasets with minimal expected outliers | Simple implementation; Minimal computational requirements | May leave relevant outliers; Less effective at reducing error inflation [45] |
| Data Transformation | Application of mathematical functions (e.g., logarithms) to normalize distribution [46] | Data with non-constant error proportional to analyte value; Asymmetric distributions | Can normalize distributions without deleting data; Handles proportional error structures | Introduces nonlinearity; Requires back-transformation; May complicate interpretation |
| Robust Statistical Methods | Use of median instead of mean; Weighted calculations to minimize outlier influence [46] | Exploratory analysis; Datasets where preservation of all data points is critical | Resistant to outlier effects; No data loss | Non-standard implementations for multivariate data; May require specialized software |
| Root Cause Analysis | Investigation of fundamental scientific causes of discordant values [46] | Pharmaceutical quality control; Discovery research | Can reveal new scientific phenomena; Addresses underlying methodological issues | Time-consuming; Requires specialized investigative expertise |
Experimental Protocols for Key Strategies
Protocol 1: Iterative Outlier Removal (IOR) Methodology
The IOR protocol provides a systematic approach for identifying outliers likely unrelated to laboratory measurement procedure error [45]. This method is particularly valuable in recalibration studies where different assays, instruments, or specimen types are used across timepoints.
Materials and Equipment:
- Paired dataset of original and reference measurements
- Statistical computing software (R, Python, or equivalent)
- Data visualization tools for Bland-Altman plots
Procedural Steps:
- Calculate Initial Difference Metrics: For each participant or sample, calculate the difference between original and reference values (original - reference) [45].
- Compute Mean and Standard Deviation: Determine the mean difference and standard deviation (SD) of these differences [45].
- First Iteration Exclusion: Identify and exclude data points where the difference is >3 SD away from the mean difference [45].
- Iterative Recaluation: Recalculate the mean difference and SD using the remaining data points. Again identify and exclude data points >3 SD from this new mean difference [45].
- Process Completion: Continue iterations until no data points exceed the 3 SD threshold from the current mean difference [45].
- Validation: Compare pre- and post-IOR metrics including mean difference, SD, and prevalence rates for key biomarkers or analytes [45].
Application Note: When non-constant bias is suspected (regression slope significantly different from 1.0), apply this method to regression residuals rather than simple differences [45].
Protocol 2: Data Transformation Methodology
Transformation approaches address underlying distributional issues that may manifest as apparent outliers [46].
Materials and Equipment:
- Full dataset including suspected outliers
- Statistical software with distribution fitting capabilities
- Graphical tools for assessing distribution normality
Procedural Steps:
- Distribution Assessment: Visualize data distribution using histograms, Q-Q plots, and density plots.
- Error Structure Evaluation: Determine if error is proportional to analyte value by plotting error magnitude against concentration.
- Transformation Selection:
- For log-normal distributions or proportional error: Apply logarithmic transformation [46].
- For other distributional issues: Consider square root, Box-Cox, or other appropriate transformations.
- Model Development: Perform calibration calculations using transformed values.
- Back-Transformation: After analysis, apply inverse transformation to return results to original scale.
- Validation: Assess transformation effectiveness through residual analysis and goodness-of-fit metrics.
Protocol 3: Robust Methodological Accommodation
Robust methods reduce outlier influence without complete removal [46].
Materials and Equipment:
- Full dataset including all values
- Statistical software supporting robust regression methods
- Weighting algorithms appropriate for data structure
Procedural Steps:
- Central Tendency Assessment: Replace mean with median for central tendency estimation [46].
- Weight Determination: Assign weights based on extremity of relevant characteristics [46].
- Weighted Analysis: Implement weighted versions of standard algorithms (MLR, PCR, PLS) [46].
- Model Validation: Compare robust model performance with standard approaches using cross-validation.
Quantitative Comparison of Strategy Performance
Table 2: Experimental Performance Data from Uric Acid Recalibration Study
| Performance Metric | Before Outlier Removal | After Single-Round Removal | After IOR (4 iterations) |
|---|---|---|---|
| Sample Size (n) | 200 [45] | 196 [45] | 191 [45] |
| Mean Original Value (mg/dL) | 6.41 (SD=1.44) [45] | 6.35 (SD=1.39) [45] | 6.36 (SD=1.38) [45] |
| Mean Reference Value (mg/dL) | 5.17 (SD=1.30) [45] | 5.13 (SD=1.23) [45] | 5.12 (SD=1.23) [45] |
| Mean Difference (mg/dL) | 1.25 (SD=0.62) [45] | 1.22 (SD=0.51) [45] | 1.23 (SD=0.45) [45] |
| Outliers Identified (n) | 0 | 4 [45] | 9 [45] |
| Hyperuricemia Prevalence (>7 mg/dL) | 28.5% [45] | 7.5% [45] | 8.5% [45] |
Table 3: Simulation Results Comparing Outlier Removal Approaches (1,000 observations)
| Simulation Parameter | Standard Single-Round Removal | Iterative Outlier Removal |
|---|---|---|
| Outlier Detection Rate (1% contamination) | <1% identified | >1% identified [45] |
| Outlier Detection Rate (5% contamination) | <5% identified | ~5% identified [45] |
| Outlier Detection Rate (10% contamination) | <10% identified | ~10% identified [45] |
| Reduction in Standard Error | Moderate [45] | Significant [45] |
| Slope Estimation Accuracy | Moderately improved | Substantially improved [45] |
Visual Workflows for Outlier Resolution Strategies
Figure 1: Decision workflow for selecting appropriate outlier resolution strategies based on data characteristics and research context.
Figure 2: Step-by-step iterative outlier removal (IOR) protocol for systematic outlier identification and exclusion.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Essential Materials for Outlier Resolution in Validation Studies
| Item | Function/Application | Specification Considerations |
|---|---|---|
| Statistical Software Platform | Implementation of IOR, data transformation, and robust statistical methods | R, Python with scikit-learn, SAS, or MATLAB with statistical toolboxes |
| Reference Control Materials | Method calibration and outlier assessment baseline | Certified reference materials with established target values and uncertainty ranges |
| Data Visualization Tools | Generation of Bland-Altman plots, distribution visualizations | Software capable of producing publication-quality figures (ggplot2, Matplotlib, etc.) |
| Quality Control Samples | Monitoring analytical performance throughout validation | Samples representing low, medium, and high concentration levels of analyte |
| Documentation System | Recording outlier decisions and methodological details | Electronic laboratory notebook (ELN) or standardized documentation templates |
| Collaborative Validation Framework | Inter-laboratory comparison of outlier management approaches | Standardized protocols shared across Forensic Science Service Providers (FSSPs) [44] |
Selecting appropriate strategies for resolving discrepancies and outlier results requires careful consideration of research context, data characteristics, and methodological goals. The experimental data presented demonstrates that Iterative Outlier Removal provides more effective outlier identification and error reduction compared to single-round removal in recalibration studies [45]. However, transformation and robust methods offer valuable alternatives when data preservation is prioritized or distributional issues underlie apparent outliers [46].
For TRL 4 forensic research, where method refinement and inter-laboratory validation are crucial, establishing standardized protocols for outlier management enhances reproducibility and reliability across institutions [44]. The workflows, protocols, and comparative data presented here provide researchers with evidence-based guidance for implementing these critical methodological safeguards in validation study design.
Using ILC/PT Data for Continuous Method Improvement and Training
Within the rigorous framework of forensic science, the transition of a novel analytical method from research to routine casework is governed by its Technology Readiness Level (TRL). TRL 4 represents a critical stage where a standardized method undergoes refinement, enhancement, and inter-laboratory validation, making it ready for implementation in forensic laboratories [3]. At this juncture, Inter-Laboratory Comparisons (ILC) and Proficiency Testing (PT) cease to be mere accreditation requirements and become powerful tools for continuous improvement. Successful participation in these programs provides external validation, promotes confidence among stakeholders, and generates invaluable data that can be leveraged to refine methods, estimate measurement uncertainty, and target staff training [16]. This guide objectively examines the role of ILC/PT data in advancing forensic methods, focusing on its application within a TRL 4 validation study design.
ILC/PT in the Forensic Validation Framework
Technology Readiness Levels (TRL) and Legal Admissibility
For a forensic method to be admissible in legal proceedings, it must satisfy stringent legal standards, such as the Daubert Standard in the United States or the Mohan Criteria in Canada [2]. These standards emphasize that a technique must be tested, peer-reviewed, have a known error rate, and be generally accepted in the scientific community [2]. A TRL 4 method, by definition, addresses these requirements through intra-laboratory validation and initial inter-laboratory trials. ILC/PT participation directly provides evidence for calculating method error rates and demonstrating precision and accuracy, which are fundamental for meeting these legal benchmarks [16] [2].
Core Concepts: ILC and PT
- Inter-Laboratory Comparison (ILC): The organization, performance, and evaluation of tests on the same or similar test items by two or more laboratories in accordance with pre-determined conditions [16].
- Proficiency Testing (PT): An evaluation of participant performance against pre-established criteria through ILC; it is a core tool for demonstrating laboratory competence [16].
A well-documented PT plan is essential for forensic laboratories, requiring annual participation to ensure adequate coverage of the scope of accreditation within a four-year cycle [16].
Experimental Protocols for Utilizing ILC/PT Data
The following protocols outline how to experimentally incorporate ILC/PT data into a TRL 4 method validation study.
Protocol 1: Method Performance Benchmarking
Objective: To compare the performance of a laboratory's internal method (Method A) against the performance of peer laboratories using the same or different methods on a standardized PT sample.
Methodology:
- PT Participation: Select and participate in a relevant PT scheme that provides detailed summary reports, including data on participant methods and statistical performance metrics (e.g., z-scores, assigned values, standard deviations).
- Data Extraction: From the PT report, extract the summary statistics for all participants and, if available, for subgroups using methods similar to your own (Method A) and other common methods (e.g., Method B, Method C).
- Performance Calculation: Calculate key performance indicators for your laboratory and the peer groups, including:
- Accuracy: Deviation from the assigned value.
- Precision: Standard deviation or relative standard deviation.
- z-score: A standardized measure of performance (
z = (lab result - assigned value) / standard deviation). A |z| ≤ 2.0 is generally considered satisfactory.
Data Utilization: The compiled data allows for a direct, objective comparison of your method's performance against the market average and alternative techniques, highlighting relative strengths and potential weaknesses.
Protocol 2: Internal Method Optimization and Uncertainty Estimation
Objective: To use ILC data to validate a new method against an existing one and to provide experimental data for the estimation of measurement uncertainty.
Methodology:
- Split-Sample Analysis: Analyze a homogeneous, stable sample split into multiple aliquots using both the established method and the new/optimized method.
- Inter-Laboratory Data Collection: Source ILC data from multiple laboratories that have analyzed the same or a directly comparable material. This data can be from formal PT schemes or collaboratively organized ILCs.
- Statistical Comparison: Perform statistical tests (e.g., t-tests, F-tests) to compare the accuracy and precision of the two methods. The variability observed in the ILC data can be used as a component in calculating the method's standard measurement uncertainty.
Data Utilization: This protocol provides a robust basis for method validation, demonstrating that the new method performs as well as or better than the existing one. The ILC data provides a "real-world" estimate of method precision across different environments, instruments, and operators, which is crucial for a defensible uncertainty budget [16].
Comparative Performance Data
The data generated from the experimental protocols above should be synthesized into clear tables for objective comparison.
Table 1: Comparative Method Performance from a Hypothetical PT Scheme for Drug Quantification
| Method | Number of Labs | Assigned Value (mg/g) | Mean Result (mg/g) | Standard Deviation (mg/g) | Average | z | -score |
|---|---|---|---|---|---|---|---|
| Method A (LC-MS/MS) | 25 | 100.5 | 100.2 | 1.8 | 0.72 | ||
| Method B (GC-MS) | 18 | 100.5 | 99.8 | 2.5 | 1.12 | ||
| Method C (HPLC-UV) | 32 | 100.5 | 101.0 | 3.1 | 0.95 | ||
| All Participants | 75 | 100.5 | 100.4 | 2.4 | 0.91 |
Table 2: ILC Data for Measurement Uncertainty Estimation (Trace Element Analysis)
| Laboratory ID | Result (ppm) | Deviation from Mean (ppm) | Squared Deviation |
|---|---|---|---|
| Lab 01 | 12.5 | -0.3 | 0.09 |
| Lab 02 | 13.2 | 0.4 | 0.16 |
| Lab 03 | 12.8 | 0.0 | 0.00 |
| Lab 04 | 12.6 | -0.2 | 0.04 |
| Lab 05 | 13.1 | 0.3 | 0.09 |
| Mean & Standard Deviation | 12.8 ppm | s = 0.31 ppm |
Data Interpretation and Training Implications
Interpreting ILC/PT data goes beyond checking for a satisfactory z-score. Trends in the data can directly inform continuous improvement and targeted training programs.
- Identifying Methodologic Weaknesses: If data from Table 1 consistently shows that a particular method (e.g., Method C) has a larger standard deviation, it may indicate the method is more susceptible to matrix effects or requires more stringent calibration control. This insight directs research and development efforts.
- Operator Performance and Training: ILC/PT results can be used to compare operator capabilities, providing operator-specific repeatability data [16]. A consistent bias or high variation linked to a specific operator highlights a direct training need, such as refresher courses on sample preparation or instrument calibration.
- Uncertainty Estimation: The standard deviation derived from ILC data, as shown in Table 2, provides a robust, empirically-based component for the estimation of measurement uncertainty, moving beyond theoretical models to data-driven assessments [16].
The Scientist's Toolkit: Essential Reagents and Materials
The following table details key materials and reagents essential for conducting validation experiments and participating in ILC/PT programs.
Table 3: Research Reagent Solutions for Forensic Method Validation
| Item | Function in Experiment |
|---|---|
| Certified Reference Materials (CRMs) | Provides a traceable and definitive value for a specific analyte in a defined matrix, used for method calibration, accuracy determination, and assigning values in PT schemes [16]. |
| Proficiency Test (PT) Samples | Commercially available samples with homogenized and stable properties, designed to simulate casework samples and assess a laboratory's testing performance against peers [16]. |
| Internal Standards (Isotope-Labeled) | Compounds with nearly identical chemical properties to the analyte but different mass; used in mass spectrometry to correct for sample loss and instrument variability, improving accuracy and precision. |
| Quality Control (QC) Materials | Stable, well-characterized materials run alongside test samples to monitor the ongoing performance and stability of the analytical method during a validation study or ILC. |
Workflow and Logical Relationships
The following diagram illustrates the continuous improvement cycle driven by ILC/PT data within a TRL 4 forensic method validation framework.
ILC/PT Driven Improvement Cycle
Signaling Pathways for Data Utilization
This diagram maps the decision-making pathway for translating specific ILC/PT data patterns into targeted training and method improvement actions.
Data-Driven Decision Pathway
Addressing Instrumentation and Reagent Variability Across Sites
In forensic method development, the Technology Readiness Level 4 (TRL 4) stage represents a critical transition where proof-of-concept technologies mature into integrated laboratory prototypes. At this juncture, components that functioned optimally in isolation are tested together to validate them as a cohesive system [47]. This integration phase presents substantial challenges for inter-laboratory studies, where instrumentation and reagent variability across different sites can significantly impact the reproducibility and reliability of analytical results. For forensic researchers and drug development professionals, understanding and controlling these sources of variability is essential for successful method validation and eventual adoption.
The inherent diversity of analytical platforms, each with distinct performance characteristics, coupled with the potential for contamination and batch effects in reagents and consumables, creates a complex validation landscape. This article systematically examines these variability sources, provides comparative performance data across instrumentation platforms, and outlines robust experimental protocols to standardize inter-laboratory validation studies at the TRL 4 stage.
Technology Readiness Level 4 in Context
Defining TRL 4 in Forensic Method Development
Technology Readiness Levels (TRL) provide a systematic measurement framework for assessing technology maturity, with TRL 1 representing initial basic research and TRL 9 indicating proven mission-ready systems [22]. TRL 4 occupies a pivotal position in this continuum, serving as the bridge between theoretical promise and practical application. At this stage, technology components transition from isolated proof-of-concept experiments to integrated system validation in laboratory environments [47].
The fundamental objective at TRL 4 is to demonstrate that integrated components function cohesively as a complete system under controlled conditions. This involves verifying that all elements—instrumentation, reagents, protocols, and analytical methodologies—work in concert to produce reliable, reproducible results. For forensic analytical chemistry, this often means validating that a method can reliably detect and quantify target analytes in complex matrices across multiple laboratory environments.
Strategic Importance for Multi-Site Validation
TRL 4 represents the final development stage conducted entirely within controlled laboratory environments before progressing to more realistic simulation environments at TRL 5 [47] [48]. This makes it the last opportunity to identify and resolve fundamental compatibility issues before facing the additional complexities of real-world operational settings. The components that worked perfectly in isolation often reveal unexpected interactions when integrated, highlighting the necessity of rigorous testing at this stage [48].
For multi-site studies, TRL 4 provides the ideal framework for establishing standardized protocols and performance benchmarks that can be implemented across participating laboratories. Success at this stage builds confidence in the technology's reliability and generates the preliminary data needed to justify further investment and development toward operational deployment.
Figure 1: TRL 4 Multi-Site Validation Workflow. This diagram illustrates the sequential process from component validation through multi-site verification that characterizes Technology Readiness Level 4 in forensic method development.
Instrumentation Platform Comparison
Mass Spectrometry Platforms: Performance Characteristics
Liquid chromatography-mass spectrometry (LC-MS) platforms represent cornerstone technologies in modern forensic toxicology, yet their varying performance characteristics introduce significant variability in inter-laboratory studies [49]. A recent systematic comparison of four LC-MS platforms for zeranol analysis in urine provides objective performance data essential for platform selection in multi-site validation studies [50] [51].
The study evaluated two low-resolution (linear ion trap) and two high-resolution (Orbitrap and time-of-flight) platforms, revealing substantial differences in sensitivity, precision, and selectivity that directly impact quantitative results. These performance variations stem from fundamental differences in instrument design and detection principles, which must be accounted for when establishing cross-platform validation criteria.
High-resolution mass spectrometry (HRMS) platforms, particularly Orbitrap technology, demonstrated superior capability to differentiate between coeluting compounds with similar mass-to-charge ratios—a common challenge in complex biological matrices [51]. For example, in zeranol analysis, HRMS could distinguish a concomitant peak at 319.1915 from the target analyte at 319.1551, while low-resolution instruments could not resolve these species, potentially leading to inaccurate quantification [50].
Quantitative Performance Metrics
Table 1: Performance Comparison of LC-MS Platforms for Zeranol Analysis [50] [51]
| Performance Metric | Orbitrap | Linear Ion Trap (LTQ) | Linear Ion Trap (LTQXL) | Time-of-Flight (G1 V Mode) | Time-of-Flight (G1 W Mode) |
|---|---|---|---|---|---|
| Sensitivity Ranking | 1 (Highest) | 2 | 3 | 4 | 5 (Lowest) |
| Precision (%CV) | Lowest | Moderate | Moderate | High | Highest |
| Mass Accuracy | Highest | Low | Low | High | High |
| Resolution | >100,000 | <2,000 | <2,000 | ≥10,000 | ≥10,000 |
| Linear Dynamic Range | 3-4 orders | 2-3 orders | 2-3 orders | 2-3 orders | 2-3 orders |
| Ability to Resolve Coeluting Compounds | Excellent | Poor | Poor | Good | Good |
Analytical Figures of Merit and Implications
The calibration curves across all platforms demonstrated strong linearity (r = 0.989 ± 0.012) for all zeranol analytes, indicating that instrument choice does not fundamentally compromise quantitative capability when properly validated [51]. However, the limits of detection (LOD) and quantification (LOQ) followed a consistent ranking pattern, with Orbitrap technology showing superior sensitivity, followed by the linear ion traps and time-of-flight instruments [50].
This performance hierarchy has practical implications for method transfer between laboratories employing different platforms. A method developed on a high-resolution Orbitrap system may require modification when implemented on a low-resolution linear ion trap, particularly for analytes present at trace concentrations or in complex matrices with significant background interference.
For inter-laboratory studies, these findings underscore the necessity of establishing platform-specific acceptance criteria that account for inherent performance differences while maintaining overall data quality standards. This may involve adjusting LOD/LOQ requirements, implementing additional sample cleanup procedures for less sensitive platforms, or establishing compound-specific qualification thresholds based on instrument capabilities.
Experimental Protocols for Platform Comparison
Sample Preparation and Extraction Methodology
Standardized sample preparation is fundamental to minimizing variability in cross-platform comparisons. The zeranol comparison study employed a robust solid-phase extraction (SPE) protocol optimized for recovery and reproducibility across instruments [51]. The methodology involved:
- Matrix-matched calibration standards prepared in pooled urine at concentrations ranging from 0-20 ng/mL
- Enzymatic deconjugation using β-glucuronidase from Helix pomatia in sodium acetate buffer (pH 4.65) during overnight incubation at 37°C
- Dual-stage SPE cleanup using unbuffered Chem Elut cartridges followed by Discovery DSC-NH2 columns
- Extraction with methyl tert-butyl ether (MTBE), evaporation under nitrogen, and reconstitution in methanol followed by LC-compatible solvent (50:25:25 H2O:MeOH:ACN)
This comprehensive sample preparation protocol effectively minimized matrix effects that could differentially impact instrument performance, thereby ensuring that observed differences truly reflected platform capabilities rather than preparation artifacts.
Instrumentation and Analytical Conditions
Each platform was operated with optimized conditions specific to its design while maintaining comparable chromatographic separation to ensure valid comparisons [51]:
- Chromatography: Reversed-phase separation using a phenyl-hexyl column with water/acetonitrile/methanol mobile phase
- Low-resolution MS: Thermo Scientific LTQ and LTQ XL linear ion traps with atmospheric pressure chemical ionization (APCI)
- High-resolution MS: Q Exactive HF Hybrid Quadrupole-Orbitrap and Waters Synapt G1 with electrospray ionization (ESI)
- Data acquisition: Full scan mode with targeted extraction for quantification
The study design incorporated quality control samples at mid-range concentrations (10 ng/mL) analyzed throughout the sequence to monitor instrument performance stability, a critical consideration for extended multi-site validation studies where analytical runs may span several days or weeks.
Figure 2: Experimental Protocol for Multi-Site Platform Comparison. This workflow outlines the standardized procedures for evaluating analytical platforms across multiple laboratory sites, highlighting critical points requiring strict protocol adherence.
Reagent and Consumable Quality Control
Quality Assurance for Reagents
Reagent quality represents a frequently underestimated source of variability in multi-site studies. Implementing robust quality control procedures for reagents is essential for detecting and preventing contamination that could compromise analytical results [52]. Key considerations include:
- Regular negative controls and reagent blanks to detect contamination introduced during reagent preparation or storage
- Pre-use quality control checks on all reagent batches before employment in casework or validation studies
- Documentation of reagent lot numbers, preparation dates, and expiration dates to facilitate investigation of batch-specific issues
- Acknowledgment of sporadic contamination that may not affect all samples equally, necessitating ongoing monitoring rather than single-point verification
These measures are particularly critical in forensic applications where trace-level detection is required, and minute contaminant introductions could generate false positive results or inaccurate quantification.
Consumables Management
Consumables represent another potential variability source, with certain items posing greater contamination risks than others. Laboratories should implement lot-specific quality control procedures for critical consumables [52]:
- Pre-treatment protocols for autoclave-compatible or UV-light-tolerant consumables to reduce contamination potential
- Percentage-based sampling strategy for evaluating consumables from each lot number prior to use in casework
- Documentation of consumable profiles to create reference databases for investigating suspected contamination events
- Special attention to non-treatable items like centrifugal filter units and filtered pipette tips that may harbor contaminants
This systematic approach to consumables management helps identify lot-specific issues before they impact study results and provides traceability for troubleshooting anomalous findings across multiple laboratory sites.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Research Reagent Solutions for Forensic LC-MS Analysis
| Reagent/Consumable | Function | Quality Control Considerations |
|---|---|---|
| Matrix-Matched Standards | Calibration reference in biologically relevant matrices | Verify absence of target analytes in blank matrix; document source and handling |
| Stable Isotope-Labeled Internal Standards | Correction for extraction efficiency and matrix effects | Assess isotopic purity; monitor for cross-talk with native analytes |
| Solid-Phase Extraction Cartridges | Sample cleanup and analyte concentration | Test blank elutions for contamination; validate recovery rates for each lot |
| Chromatography Solvents | Mobile phase components and sample reconstitution | LC-MS grade purity; monitor for background interference in blank injections |
| Enzymatic Deconjugation Reagents | Hydrolysis of conjugated metabolites | Verify activity with control compounds; monitor for non-specific hydrolysis |
| Filtered Pipette Tips | Precise liquid handling while preventing aerosol contamination | Evaluate filtrate for analyte adsorption; test for particulate introduction |
Addressing instrumentation and reagent variability across sites requires a systematic, comprehensive approach that begins at TRL 4 and continues throughout method development. The comparative performance data presented here demonstrates that platform selection directly impacts analytical capabilities, particularly for methods requiring high sensitivity and selectivity in complex matrices. By implementing the standardized protocols, quality control procedures, and performance benchmarks outlined in this guide, researchers can significantly enhance the reliability and reproducibility of multi-site validation studies.
Successful navigation of the TRL 4 stage establishes a solid foundation for progression to more advanced readiness levels, where technologies are tested in increasingly realistic environments [48]. The rigorous attention to variability sources at this critical juncture ultimately determines whether promising forensic methods will achieve widespread adoption or remain confined to research settings. Through deliberate platform evaluation, reagent standardization, and protocol harmonization, the forensic research community can accelerate the translation of innovative analytical technologies from laboratory prototypes to operational tools.
Demonstrating Courtroom Readiness: From Data to Legal Compliance
Calculating Method Error Rates and Measurement Uncertainty
In the development and validation of forensic methods, the rigorous assessment of measurement uncertainty and method error rates is not merely a technical formality but a foundational requirement for scientific and legal defensibility. For techniques at Technology Readiness Level (TRL) 4, defined as the application of an established technique with measured figures of merit, some measurement uncertainty, and developed aspects of intra-laboratory validation [3], this assessment is a critical milestone. It signifies the transition from a promising research concept to a method undergoing refinement for eventual implementation in forensic laboratories.
The legal frameworks governing the admissibility of scientific evidence, such as the Daubert Standard and Federal Rule of Evidence 702 in the United States, explicitly require knowledge of a method's potential error rate [2]. Consequently, understanding and quantifying the sources and magnitude of uncertainty in measurement results is paramount. This guide objectively compares the two principal methodologies for evaluating measurement uncertainty: the Guide to the Expression of Uncertainty in Measurement (GUM) and the Monte Carlo Method (MCM). Supported by experimental data and detailed protocols, this comparison aims to inform researchers and professionals in drug development and forensic science on selecting the most appropriate approach for their inter-laboratory validation studies at TRL 4.
Comparative Analysis of Uncertainty Evaluation Methods
The GUM provides an analytical framework for uncertainty evaluation, propagating uncertainties from input quantities through a measurement model using first-order Taylor series approximations and combining them into a single standardized metric [53]. In contrast, MCM is a computational approach that employs random sampling from the probability distributions of input quantities to simulate a large number of possible outcomes, thereby constructing a numerical representation of the output quantity's distribution [53].
A comparative study evaluating the gauge factor (GF) of high-temperature strain gauges provides quantitative data on the performance of both methods [53]. The experiment was conducted from 25°C to 900°C, and the uncertainty of the GF calibration was assessed using both GUM and MCM. The table below summarizes the key comparative findings.
Table 1: Performance Comparison of GUM and MCM in Uncertainty Evaluation
| Feature | GUM (Guide to Uncertainty in Measurement) | MCM (Monte Carlo Method) |
|---|---|---|
| Core Principle | Analytical propagation of uncertainties using a first-order Taylor series approximation [53] | Numerical propagation of distributions via random sampling from input probability distributions [53] |
| Model Flexibility | Best suited for linear or mildly nonlinear models; performance can degrade with strong nonlinearity [53] | Handles highly nonlinear models effectively without additional complexity [53] |
| Computational Demand | Low; relies on analytical formulas [53] | High; requires a large number of model evaluations (e.g., hundreds of thousands to millions) [53] |
| Output Provided | A combined standard uncertainty and expanded uncertainty interval (e.g., 95% confidence) [53] | A full numerical representation of the output's probability distribution [53] |
| Key Finding from Experimental Data | The uncertainty interval provided was less aligned with the real situation compared to MCM [53] | The uncertainty interval was closer to the real situation, proving superior for this specific application [53] |
| Best Suited For | Relatively simple, linear measurement models where computational simplicity is desired | Complex, nonlinear models where a more realistic estimation of the output distribution is critical [53] |
Experimental Protocol for Uncertainty Comparison
The following detailed methodology is adapted from a study comparing GUM and MCM for calibrating high-temperature strain gauges, providing a template for a TRL 4 intra-laboratory validation study [53].
Materials and Equipment
Table 2: Research Reagent Solutions and Essential Materials
| Item Name | Function / Specification |
|---|---|
| Pt-W High-Temperature Strain Gauges | The sensor under test; used for strain monitoring on aero-engine hot-end components [53]. |
| Calibration Specimen | A beam structure onto which the strain gauge is installed [53]. |
| Plasma-Sprayed Ceramic (Al₂O₃) | Used as an insulating adhesive to install the strain gauge onto the specimen for high-temperature operation [53]. |
| High-Temperature Furnace | Provides a controlled temperature environment from room temperature up to 900°C [53]. |
| Strain Meter/Indicator | Measures the change in electrical resistance of the strain gauge and converts it to a strain reading [53]. |
| Mechanical Loading System | Applies and removes a precise mechanical load to the calibration specimen [53]. |
Step-by-Step Procedure
- Specimen Preparation and Gauge Installation: Install the high-temperature strain gauges onto the midpoint of the calibration specimen using the plasma-sprayed ceramic Al₂O�3 [53].
- Setup Assembly: Place the specimen and its support structure into the high-temperature furnace. Connect the installed strain gauges to the strain meter [53].
- Temperature Profiling: Heat the furnace according to a defined temperature ramp rate and specific calibration points (e.g., from 25°C to 900°C in intervals). Hold each target temperature for a set duration to achieve thermal stability [53].
- Data Acquisition at Temperature: At each stabilized calibration temperature point:
a. Apply and remove the mechanical load three times.
b. For each loading cycle, record the specimen's midpoint deflection (
f_l/2) and the corresponding strain reading from the strain meter (Δε) [53]. - Gauge Factor Calculation: Calculate the reference strain (
ε_l/2) using Hooke's law from the measured deflection (Equation 1). Then, compute the gauge factor (GF) for each measurement using the established mathematical model (Equation 3) [53].
The core mathematical model used in this calibration is summarized below, illustrating the relationship between the inputs and the output GF.
Figure 1: Logical workflow for calculating the Gauge Factor (GF).
Uncertainty Evaluation and Error Rate Analysis
- Identify Input Uncertainties: List all sources of uncertainty, including the Type A uncertainty from the repeated strain readings (
Δε) and Type B uncertainties from instrument calibration (e.g., dimensionsh,l,aof the specimen) [53] [54]. - Quantify and Characterize Uncertainties:
- For
Δε, calculate the standard uncertainty of the mean (u(Δε_bar)) from 18 measured values (6 strain gauges × 3 load cycles) using Equations 4 and 5 (Type A) [53]. - For other inputs, determine their standard uncertainties based on calibration certificates or manufacturer specifications, using Equation 6 (Type B) [53].
- For
- Apply GUM and MCM:
- GUM Path: Use the law of propagation of uncertainty to combine the standard uncertainties of all inputs into the combined standard uncertainty for
GF. Multiply by a coverage factor (e.g., k=2) to obtain the expanded uncertainty at approximately 95% confidence [53] [54]. - MCM Path: For each input quantity, define its probability distribution based on the characterization in the previous step. Run a large number of simulations (e.g., 1,000,000), each time randomly sampling from the input distributions and computing a corresponding
GFvalue. The resulting distribution ofGFvalues directly provides the estimate, standard uncertainty, and a coverage interval [53].
- GUM Path: Use the law of propagation of uncertainty to combine the standard uncertainties of all inputs into the combined standard uncertainty for
- Compare and Validate: Compare the uncertainty intervals and standard uncertainties obtained from both methods. The experimental study found that the MCM interval was "closer to the real situation" for this specific, nonlinear application [53].
- Determine Chief Uncertainty Sources: To identify the most significant contributors to the overall uncertainty, the concept of a weight coefficient (
W) can be employed. This coefficient quantitatively analyzes the influence of each input's uncertainty on the uncertainty of the outputGF[53]. In the strain gauge study, the uncertainty in the strain reading (Δε) was identified as the main source.
Implications for TRL 4 Inter-Laboratory Validation
At TRL 4, the focus is on intra-laboratory validation and establishing figures of merit, with inter-laboratory studies on the horizon [3]. The choice between GUM and MCM has direct implications for this stage.
Table 3: Uncertainty Method Selection at TRL 4
| Consideration | GUM | MCM |
|---|---|---|
| Implementation at TRL 4 | Suitable for establishing initial, defensible uncertainty estimates for simpler methods. | Recommended for complex methods where nonlinearity is a concern, providing a more robust foundation for inter-laboratory trials. |
| Error Rate for Legal Standards | Provides an uncertainty value that contributes to understanding measurement reliability. | Can provide a more comprehensive and realistic probabilistic foundation for estimating error rates, which is a key Daubert criterion [2]. |
| Path to TRL 4 and Beyond | A GUM-based uncertainty budget satisfies the "measurement of uncertainty" requirement for TRL 4 [3]. | An MCM evaluation may offer greater confidence and resilience during inter-laboratory validation (TRL 4+) and future courtroom scrutiny under the Daubert Standard [2]. |
The following diagram outlines the decision-making process for selecting an uncertainty evaluation method within a TRL 4 validation framework.
Figure 2: Decision workflow for selecting an uncertainty evaluation method at TRL 4.
Documenting Method Robustness and Reproducibility for Peer Review
For forensic science research, particularly at Technology Readiness Level (TRL) 4, demonstrating robustness and reproducibility is a critical gateway for method acceptance and progression toward implementation in casework [3]. TRL 4 is characterized by the refinement, enhancement, and inter-laboratory validation of a standardized method, making it ready for implementation in forensic laboratories [3]. At this stage, new knowledge should be immediately adoptable for casework, necessitating a level of documentation in peer-reviewed articles that is comprehensive, transparent, and structured to withstand scrutiny from both the scientific and legal communities. This guide provides a framework for comparing product performance and documenting experimental data to meet the rigorous demands of peer review for TRL 4 forensic research.
The paradigm is shifting in forensic science, moving from methods based on human perception and subjective judgment toward those grounded in relevant data, quantitative measurements, and statistical models [36]. This new paradigm requires methods that are not only transparent and reproducible but also intrinsically resistant to cognitive bias and empirically validated under casework conditions. Proper documentation of robustness and reproducibility is the cornerstone of this shift, providing the evidence base needed for a method to be considered "generally accepted" under legal standards like Daubert and Mohan [2].
Defining Reproducibility and Robustness in the Forensic Context
A clear understanding of key terms is essential for accurate documentation. In computational science, these concepts are often defined hierarchically [55]:
- Reproducibility is achieved when the same results are obtained using the same data and the same code or methods. It is the fundamental first step in validating any scientific finding.
- Robustness is demonstrated when the same results are obtained using the same data but with different code or a different implementation of the method (e.g., a different software environment or programming language) [55].
- Replicability refers to obtaining consistent results when using different data but the same code or methods.
For forensic science, these concepts extend beyond the laboratory bench. The legal system imposes additional requirements, and forensic methods must be legally reliable [2]. This involves meeting criteria such as known error rates, peer review, and general acceptance within the relevant scientific community, as outlined in the Daubert standard [2].
The following workflow outlines the key stages for establishing and documenting reproducibility and robustness in a TRL 4 study, from initial design to peer-review submission:
Experimental Protocols for Assessing Robustness and Reproducibility
Designing experiments to test the limits of a method is crucial for TRL 4. The following protocols provide a template for generating the necessary data to support claims of robustness and reproducibility.
Protocol for Inter-Laboratory Comparability
This protocol is designed to assess whether different laboratories can reproduce the same results using the same standardized method, a key requirement for TRL 4 [3].
- Objective: To evaluate the inter-laboratory comparability of quantitative results (e.g., δ13C and δ18O isotope ratios) by having multiple laboratories analyze identical sample sets.
- Materials:
- Samples: A set of 10 or more homogeneous, well-characterized samples (e.g., faunal teeth for isotope analysis) [6].
- Reagents: All necessary chemicals and standards, with identical batches and suppliers provided to all participating laboratories.
- Instrumentation: Similar class instruments (e.g., GC×GC–MS, isotope ratio mass spectrometers) across laboratories.
- Method:
- Sample Preparation: Provide all laboratories with an identical, detailed sample preparation protocol (e.g., whether to use chemical pretreatment, baking procedures, acid reaction temperature) [6].
- Blinded Analysis: Each laboratory receives a randomized, blinded set of sample subsamples.
- Data Collection: Each laboratory follows the identical protocol to analyze the samples and reports the raw data and calculated results to a central coordinator.
- Statistical Analysis:
- Use intraclass correlation coefficients (ICC) to measure agreement between laboratories.
- Perform Bland-Altman analysis to assess systematic bias between laboratory results.
- Use analysis of variance (ANOVA) to partition total variance into within-laboratory and between-laboratory components.
Protocol for Robustness Testing via Parameter Variation
This protocol tests how sensitive the method's outcomes are to deliberate, minor changes in key parameters, establishing its operational boundaries.
- Objective: To determine the impact of deliberate variations in key method parameters on the final results, thereby defining the method's robustness.
- Materials:
- A single, well-characterized test sample.
- The standard reagents and instrumentation for the method.
- Method:
- Identify Critical Parameters: Select parameters for testing (e.g., linkage methods in hierarchical clustering, graph regulator factors, incubation temperature, pH of a buffer) [55].
- Define Variation Range: Systematically vary each parameter one at a time around the specified optimum.
- Replicate Measurements: For each parameter setting, perform a minimum of three replicate measurements of the test sample.
- Statistical Analysis:
- Calculate the mean, standard deviation, and relative standard deviation (RSD) for results at each parameter setting.
- Use control charts to visually identify parameter ranges where results remain within pre-defined acceptance criteria (e.g., ±2SD of the result at optimum conditions).
Quantitative Data Presentation and Comparison
Structured presentation of data is vital for peer review. The following tables provide templates for summarizing key experimental results.
Table 1: Example Data Summary for an Inter-Laboratory Comparability Study of Tooth Enamel Isotope Analysis (adapted from [6])
| Sample ID | Laboratory A δ13C (‰) | Laboratory B δ13C (‰) | Laboratory A δ18O (‰) | Laboratory B δ18O (‰) | Inter-Lab Difference δ13C (‰) | Inter-Lab Difference δ18O (‰) |
|---|---|---|---|---|---|---|
| T-001 | -14.8 | -14.5 | -4.3 | -4.1 | 0.3 | 0.2 |
| T-002 | -12.1 | -12.4 | -3.8 | -4.0 | -0.3 | -0.2 |
| T-003 | -15.3 | -15.6 | -5.1 | -5.3 | -0.3 | -0.2 |
| ... | ... | ... | ... | ... | ... | ... |
| Mean Difference | -0.05 | -0.07 | ||||
| Standard Deviation of Differences | 0.15 | 0.12 |
Table 2: Example Data Summary for a Robustness Test of a Hierarchical Clustering Method (inspired by [55])
| Tested Parameter | Standard Value | Varied Value | Impact on Cluster Purity (%) | Impact on Rand Index | Meets Acceptance Criteria? |
|---|---|---|---|---|---|
| Linkage Method | UPGMA (MATLAB) | Single (SciPy) | -22.5 | -0.31 | No [55] |
| Linkage Method | UPGMA (MATLAB) | UPGMA (SciPy) | -1.2 | -0.02 | Yes |
| Graph Factor | 1800 (Optimal) | 1.0 | -35.1 | -0.45 | No [55] |
| Graph Factor | 1800 (Optimal) | 1600 | -3.5 | -0.05 | Yes |
| Random Seed | 12345 | 54321 | +0.8 | +0.01 | Yes |
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table details key materials and tools essential for conducting and documenting robustness and reproducibility studies in forensic chemistry.
Table 3: Essential Research Reagent Solutions for Forensic Validation Studies
| Tool/Reagent | Specific Function in Validation | Example Use-Case in Documentation |
|---|---|---|
| Stable Isotope Reference Materials | Calibrates mass spectrometers and ensures accuracy and comparability of isotope ratio data across laboratories. | Document the specific reference materials (e.g., NIST standards) and their measured values to establish traceability [6]. |
| Certified Reference Materials (CRMs) | Provides a ground truth for method accuracy for specific evidence types (e.g., drug mixtures, ignitable liquids). | Report recovery rates and accuracy metrics when the method is applied to a CRM. |
| Open-Source Software (e.g., R, Python) | Enforces transparency and robustness by allowing re-implementation and re-analysis without proprietary license barriers [55]. | Provide a link to the full analysis code in a repository like GitHub. Specify version numbers (e.g., Python 3.8, R 4.1.0) and key packages (e.g., SciPy, NumPy) [55] [6]. |
| Containerization Software (e.g., Docker) | Captures the entire computational environment (OS, libraries, code) to guarantee long-term reproducibility. | Include a Dockerfile in the submission materials to allow reviewers and readers to recreate the exact analysis environment [55]. |
| Interactive Notebooks (e.g., Jupyter) | Combines code, textual explanations, and results in a single document, ideal for creating transparent tutorials and workflows. | Submit a Jupyter notebook as supplementary material that walks through the key data processing steps [55]. |
A Framework for Documentation in Peer-Reviewed Manuscripts
To ensure transparency and facilitate peer review, manuscripts should explicitly include the following sections, which go beyond a simple description of the methods.
Statement on Robustness and Reproducibility
A dedicated section should summarize the efforts undertaken to establish and test the method's reproducibility and robustness. This section should:
- Explicitly state the TRL of the research (self-assigned as TRL 4) [3].
- Briefly describe the inter-laboratory study and robustness tests performed.
- Reference the availability of data, code, and detailed protocols.
Data, Code, and Materials Availability
Adherence to the FAIR Principles (Findable, Accessible, Interoperable, and Reusable) is paramount [56]. This section must provide direct links to:
- Raw and Processed Data: Publicly deposited in a reputable repository such as Figshare, Zenodo, or Dryad [6] [56].
- Analysis Code and Scripts: Deposited on a version-controlled platform like GitHub, with a clear link and DOI provided [55] [6].
- Detailed Computational Environment: This can be achieved by providing a Docker container or by listing all software packages with their exact version numbers [55].
Detailed Description of Statistical Analyses and Uncertainty
Merely stating the statistical tests used is insufficient. Authors must provide a full account of their statistical approaches, including [57]:
- The rationale for sample sizes and statistical power, if applicable.
- A complete account of statistical outputs (e.g., effect sizes, confidence intervals), not just p-values.
- Measures of uncertainty for all key results.
- A description of how missing data or outliers were handled.
For forensic methods at TRL 4, demonstrating robustness and reproducibility through well-designed inter-laboratory studies and rigorous documentation is not merely an academic exercise—it is a fundamental requirement for gaining the trust of the scientific community and the legal system. By adopting the structured approach outlined in this guide, researchers can provide the transparent, comprehensive evidence needed during peer review to show that a method is truly ready for implementation in casework. This commitment to rigor and transparency is the foundation for advancing reliable and defensible forensic science.
Compiling Evidence for Standard Development Organizations (SDOs)
In forensic science, the admissibility and reliability of evidence often hinge on the methodological rigor and standardized application of analytical techniques. Technology Readiness Level (TRL) 4 represents a critical stage where a method is refined, enhanced, and subjected to inter-laboratory validation, making it ready for implementation in forensic laboratories [15] [58]. Research at this level generates new knowledge that can be immediately adopted in casework [15]. For Standard Development Organizations (SDOs), compiling evidence at this stage is paramount to establishing protocols that ensure results are reproducible, comparable across different laboratories, and meet the stringent admissibility standards set by legal systems, such as the Daubert Standard and Mohan Criteria [2]. This guide objectively compares experimental approaches and data for designing robust inter-laboratory validation studies, providing a foundational framework for SDOs to develop actionable and legally defensible standards.
Comparative Analysis of Key Inter-Laboratory Validation Studies
The following section presents a comparative analysis of selected studies, highlighting their experimental designs, key findings, and relevance to SDOs developing standards for forensic methods.
Table 1: Comparison of Inter-Laboratory Validation Study Designs and Findings
| Study Focus / Technique | Experimental Design & Protocol | Key Comparative Findings | Implications for SDOs |
|---|---|---|---|
| Tooth Enamel Carbonate Stable Isotope Analysis (δ13C, δ18O) [6] | • Samples: 10 "modern" faunal teeth.• Protocol: Subsamples from the same specimens were analyzed in two different laboratories.• Variables Tested: Chemical pretreatment (applied vs. not applied), sample baking (with vs. without), acid reaction temperature (standardized vs. not). | • Chemical Pretreatment: Caused systematic differences in δ values between labs. Untreated samples showed smaller or negligible differences.• Baking: Improved inter-lab comparability under certain conditions.• Acid Temperature: Had little-to-no impact on comparability. | Standards should deemphasize chemical pretreatment for enamel samples. Protocols can allow flexibility in acid reaction temperature but should provide guidelines on baking procedures. |
| Comprehensive Two-Dimensional Gas Chromatography (GC×GC) [2] | • Design: A review of current literature across seven forensic applications (e.g., illicit drugs, toxicology, decomposition odor).• Validation Metrics: Applications were categorized by Technology Readiness Level based on analytical and legal readiness. | • Found a need for increased intra- and inter-laboratory validation, error rate analysis, and standardization across all GC×GC applications.• Few techniques have reached the maturity required for routine casework. | SDOs should prioritize creating standards that mandate inter-lab trials, establish protocols for error rate calculation, and align with legal admissibility criteria (e.g., Daubert). |
| Collaborative Method Validation Model [44] | • Proposal: A model where an originating lab publishes a full validation in a peer-reviewed journal. Subsequent labs can perform an abbreviated verification if they adhere strictly to the published method.• Data Analysis: Cost-benefit analysis of collaborative vs. traditional independent validation. | • Proposed model demonstrates significant cost and time savings.• Facilitates direct cross-comparison of data between laboratories using identical methods.• Increases efficiency and establishes benchmarks for method performance. | SDOs should endorse and formalize this collaborative model. Standards can reference published, peer-reviewed validations as foundational documents for other labs to verify against. |
Detailed Experimental Protocols for Validation
This section elaborates on the methodologies behind the key experiments cited in the comparison guide, providing a template for designing TRL 4 validation studies.
Protocol for Inter-Laboratory Comparison of Stable Isotope Analysis
This protocol is derived from the study on tooth enamel carbonate, which achieved TRL 4 by demonstrating inter-laboratory comparability [6].
1. Sample Selection and Preparation:
- Obtain a set of homogeneous samples. The referenced study used 10 "modern" faunal teeth obtained from field recoveries [6].
- Clean the samples physically to remove surface contaminants.
- Pulverize the enamel to a homogeneous powder using a agate mortar and pestle or a dental drill.
- Split the powdered enamel into representative subsamples for distribution to participating laboratories.
2. Variable Testing (Experimental Factors):
- Chemical Pretreatment: Divide subsamples into two groups. One group undergoes chemical pretreatment (e.g., with dilute acetic acid to remove diagenetic carbonates), while the other group receives no chemical pretreatment [6].
- Baking: For selected subsamples, implement a baking step (e.g., at a specific temperature for a set duration) to remove absorbed moisture prior to analysis [6].
- Acid Reaction Temperature: Analyze some subsamples with a standardized acid reaction temperature and others without this standardization [6].
3. Inter-Laboratory Analysis:
- Distribute the subsamples from all test groups to at least two independent laboratories.
- Each laboratory analyzes the samples using their own standard operating procedures for isotope ratio mass spectrometry (IRMS) to measure δ13C and δ18O values.
- Laboratories should report raw data, including internal standards and measurement uncertainties.
4. Data Analysis and Comparability Metrics:
- Perform statistical analysis (e.g., t-tests, ANOVA) to identify systematic differences (offsets) between laboratories for each test condition.
- Calculate the pooled standard deviation or similar metrics to assess the dispersion of results across labs.
- The key metric is the magnitude and statistical significance of the difference in isotope values between labs under each protocol variation [6].
Protocol for Establishing Legal Readiness (Daubert/Mohan Compliance)
For a method to be admissible in court, validation must address specific legal criteria. The following workflow, based on a review of GC×GC techniques, outlines the necessary steps [2].
Diagram 1: Pathway from method development to legal admissibility, illustrating how validation activities map to legal criteria.
The Scientist's Toolkit: Essential Reagents and Materials
The following table details key reagents, materials, and instrumental components essential for conducting the types of validation experiments described in this guide.
Table 2: Key Research Reagent Solutions and Materials for Forensic Validation
| Item Name / Category | Function / Purpose in Validation |
|---|---|
| Homogeneous Reference Materials (e.g., powdered tooth enamel, standard drug mixtures, certified reference materials) | Serves as a consistent and well-characterized sample for distribution to multiple laboratories. This is fundamental for assessing inter-laboratory comparability and precision [6] [44]. |
| Isotope Ratio Mass Spectrometer (IRMS) | The core instrument for high-precision measurement of stable isotope ratios (e.g., δ13C, δ18O) in materials like tooth enamel carbonate. Its calibration and performance are critical for data validity [6]. |
| Comprehensive Two-Dimensional Gas Chromatograph (GC×GC) | Provides superior separation power for complex mixtures (e.g., drugs, ignitable liquids, biological samples). Validation involves establishing modulation parameters, column combinations, and temperature programs [2]. |
| Quality Control (QC) Standards & Calibrants | Includes internal standards (e.g., isotopically labeled compounds) and calibration solutions used to monitor instrument performance, correct for drift, and ensure quantitative accuracy throughout the validation process [44]. |
| Statistical Software & Data Analysis Tools (e.g., R, Python with specialized libraries) | Used for calculating key validation metrics such as error rates, repeatability/reproducibility standard deviations, confidence intervals, and for performing multivariate analysis on complex datasets [6] [59]. |
The compilation of evidence from rigorous inter-laboratory studies is the cornerstone of developing effective standards for forensic methods at TRL 4. The comparative data and protocols presented in this guide underscore several critical principles for SDOs. First, the pursuit of simplicity in sample preparation—as demonstrated by the superior comparability of untreated tooth enamel—can often yield more reproducible results than complex, multi-step protocols [6]. Second, the adoption of a collaborative validation model, where one laboratory's published validation serves as the benchmark for others, promises significant gains in efficiency and consistency across the forensic community [44]. Finally, from the initial design phase, validation studies must be structured to answer the specific questions posed by legal standards, including the calculation of known error rates and the demonstration of reliability through inter-laboratory trials [2]. By anchoring standards in this empirical, collaborative, and legally-aware framework, SDOs can ensure that new forensic methods are not only scientifically sound but also robust and readily admissible in a court of law.
Preparing the Final Validation Report for Laboratory Implementation and Legal Scrutiny
Validation studies are foundational to establishing the reliability, admissibility, and scientific integrity of analytical methods, particularly in forensic science and pharmaceutical development. These studies provide the empirical evidence required to demonstrate that a method consistently produces accurate, precise, and reproducible results under specified conditions. In the context of Technology Readiness Level (TRL) 4 research, which corresponds to the validation in a laboratory environment, the final report must be robust enough to withstand both scientific peer-review and legal scrutiny under standards such as those outlined in the Daubert ruling [27]. This guide objectively compares two common analytical techniques—Capillary Electrophoresis (CE) and High-Performance Liquid Chromatography (HPLC)—using experimental data from published studies. The framework is situated within inter-laboratory validation study design, emphasizing protocols, performance metrics, and reporting standards essential for admissibility in legal proceedings and implementation across laboratory networks.
Analytical Technique Comparison: CE vs. HPLC
Experimental Protocols and Methodologies
To ensure a fair comparison, identical or highly similar sample types and validation criteria were used across studies evaluating CE and HPLC.
- CE Method Protocol: The CE-DAD method for moniliformin (MON) in maize employed a fused-silica capillary with an inner diameter of 50 µm and a total length of 48.5 cm. The background electrolyte was a 30 mM sodium tetraborate buffer (pH 9.3). Samples were injected hydrodynamically at 50 mbar for 5 seconds, and the separation was performed at an applied voltage of 20 kV with the capillary temperature maintained at 25°C. Detection was performed at 260 nm [60].
- HPLC Method Protocol: The HPLC-DAD analysis for MON used a C18 column (150 mm x 4.6 mm, 5 µm). The mobile phase consisted of a gradient of 10 mM ammonium acetate in water (pH 5.0 with acetic acid) and acetonitrile. The flow rate was 0.8 mL/min, the column temperature was maintained at 30°C, and the injection volume was 20 µL. Detection was also performed at 260 nm [60].
- Sample Preparation: For both techniques, maize samples were ground and homogenized. One gram of sample was extracted with 4 mL of a mixture of acetonitrile and water (84:16, v/v) by shaking vigorously for 60 minutes. The extract was centrifuged, diluted with water, and purified using a solid-phase extraction (SPE) cartridge before analysis [60].
- Method Validation Parameters: Both methods were validated according to ICH guidelines, assessing specificity, linearity, accuracy, precision, limit of detection (LOD), and limit of quantification (LOQ) [61].
Comparative Experimental Data
The following tables summarize the quantitative performance data for CE and HPLC based on the analysis of MON in maize and a pharmaceutical compound (mirtazapine).
Table 1: Comparison of key validation parameters for MON analysis in maize using CE-DAD and HPLC-DAD [60].
| Validation Parameter | CE-DAD | HPLC-DAD | HPLC-MS/MS |
|---|---|---|---|
| Linear Range (ng/mL) | 50-5000 | 100-5000 | 10-5000 |
| Correlation Coefficient (R²) | >0.999 | >0.998 | >0.999 |
| LOD (ng/mL) | 15 | 30 | 3 |
| LOQ (ng/mL) | 50 | 100 | 10 |
| Accuracy (% Recovery) | 95-102 | 92-98 | 96-104 |
| Precision (% RSD) | <5% | <5% | <4% |
Table 2: Validation data for the determination of mirtazapine and related substances using CE and a reference HPLC method [62].
| Parameter | CE Performance | HPLC Performance |
|---|---|---|
| Injection Precision (RSD) | 2-3% (required internal standard) | <1% |
| Analysis Time | ~13 minutes | >35 minutes |
| Selectivity | Optimized with experimental design | Comparable selectivity |
| Running Costs | Low (minimal solvent use) | Higher (significant solvent consumption) |
Performance Analysis and Legal Relevance
The data demonstrates that CE provides comparable sensitivity and selectivity to HPLC for the analysis of polar compounds like MON, with the added advantages of shorter analysis times and lower running costs [60]. However, CE can exhibit poorer injection precision, often necessitating an internal standard for reliable quantification [62]. From a forensic and legal perspective, the error rates (implicit in precision and accuracy data) and the empirical validation of both techniques are critical. Courts acting as "gatekeepers" under Daubert and Federal Rule of Evidence 702 must examine the empirical foundation of proffered expert testimony [27]. The documented LOD, LOQ, and precision metrics provide the necessary evidence of a method's reliability. The choice between CE and HPLC can be guided by the specific application: CE is suitable for high-throughput, cost-effective analysis of ionic/polar molecules, whereas HPLC-MS/MS is indispensable for ultra-trace level confirmation and complex matrices.
Framework for Forensic Validation and Performance Measurement
Guidelines for Validating Forensic Feature-Comparison Methods
Inspired by the Bradford Hill Guidelines for causal inference in epidemiology, a parallel framework of four guidelines has been proposed to establish the validity of forensic comparison methods [27]:
- Plausibility: The scientific basis for the method must be sound and grounded in a logical theory.
- Soundness of Research Design and Methods: The research must demonstrate construct validity (does it measure what it intends to?) and external validity (are the results generalizable?).
- Intersubjective Testability: The method and its results must be replicable and reproducible by different examiners and laboratories.
- Methodology for Individualization: There must be a valid methodology to reason from group-level data (e.g., "this bullet matches this class of gun") to specific, individual source statements (e.g., "this bullet was fired from this specific gun").
These guidelines help courts and researchers move beyond the limited "checklist" approach of Daubert and provide a structured way to evaluate the scientific rigor of techniques like firearm and toolmark examination [27].
Measuring Expert Performance Using Signal Detection Theory
Evaluating the performance of human experts in forensic pattern matching (e.g., fingerprints, firearms, toolmarks) requires methods that distinguish between true accuracy and response bias. Signal Detection Theory (SDT) provides a robust framework for this purpose [28] [25].
- Core Concepts: In SDT, a "signal" is a same-source pair (e.g., a fingerprint and a print from the same person), and "noise" is a different-source pair. The expert's ability to distinguish between them is discriminability.
- Key Metrics:
- Sensitivity (d'): A measure of the examiner's ability to distinguish between same-source and different-source evidence, independent of their bias.
- Response Bias: The tendency to favor one decision outcome over another (e.g., to call more samples "matches").
- Experimental Design Considerations: Studies measuring expert performance should [25]:
- Include an equal number of same-source and different-source trials.
- Record inconclusive responses separately from definitive "match" or "non-match" decisions.
- Include a control group (e.g., novices) for comparison.
- Counterbalance or randomly sample trials for each participant.
- Present as many trials as practical to ensure statistical reliability.
The diagram below illustrates the workflow for designing and interpreting a study on expert forensic performance using SDT.
Figure 1: Workflow for expert performance study design and analysis.
The Scientist's Toolkit: Essential Reagents and Materials
The following table details key reagents, materials, and instruments essential for conducting validation studies for CE and HPLC, along with their critical functions.
Table 3: Essential research reagents and solutions for analytical method validation.
| Item Name | Function/Application | Key Consideration |
|---|---|---|
| Background Electrolyte (BGE) | Medium for electrophoretic separation in CE. Composition affects selectivity, resolution, and analysis time [62]. | pH and ionic strength must be optimized for the target analyte. |
| HPLC Mobile Phase | Solvent system that carries the sample through the chromatographic column. Can be isocratic or gradient [61]. | Purity is critical; often requires degassing to prevent air bubbles. |
| Solid-Phase Extraction (SPE) Cartridge | Purifies and concentrates the sample extract, removing matrix interferences before instrumental analysis [60]. | Select sorbent phase (e.g., C18, ion-exchange) based on analyte properties. |
| Internal Standard | A known compound added to the sample to correct for variability in injection volume and sample preparation, especially in CE [62]. | Must be chemically similar to the analyte but resolvable during separation. |
| Certified Reference Material | Provides a known concentration of the target analyte with documented purity, used for method calibration and accuracy determination [61]. | Essential for establishing traceability and measurement uncertainty. |
Visualization of the Validation Pathway for Legal Admissibility
The pathway from method development to legal admissibility is complex and requires meticulous documentation. The following diagram outlines the critical stages and decision points, incorporating the guidelines for forensic methods [27] and the requirements of legal standards [27].
Figure 2: Validation pathway from method development to legal admissibility.
Conclusion
A meticulously designed TRL 4 inter-laboratory validation study is the critical final step in transitioning a forensic method from a research-grade technique to an operationally reliable and legally defensible tool. Success hinges not only on achieving technical proficiency and statistical agreement across laboratories but also on explicitly addressing the legal criteria for admissibility. By systematically generating evidence on error rates, reproducibility, and standardization, these studies bridge the gap between scientific innovation and the practical needs of the justice system. Future efforts must focus on expanding these validation frameworks to emerging forensic technologies and fostering a culture of open data and collaborative standard-setting to ensure the continuous evolution and reliability of forensic science.
References
- 1. Technology readiness level [https://en.wikipedia.org/wiki/Technology_readiness_level]
- 2. Are We Ready for It? A Review of Forensic Applications and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12012292/]
- 3. Guide for authors - Forensic Chemistry - ISSN 2468-1709 [https://www.sciencedirect.com/journal/forensic-chemistry/publish/guide-for-authors]
- 4. Technology readiness levels [https://ised-isde.canada.ca/site/innovation-canada/en/technology-readiness-levels]
- 5. TECHNOLOGY READINESS LEVELS (TRLs) FOR ... [https://medicalcountermeasures.gov/trl/trls-for-medical-devices]
- 6. Improving inter-laboratory comparability of tooth enamel ... [https://www.sciencedirect.com/science/article/pii/S2468170925000414]
- 7. Advancing a paradigm shift in evaluation of forensic evidence [https://www.sciencedirect.com/science/article/pii/S2589871X22000559]
- 8. Legal Intelligencer: Battle of the Experts (Standards): 'Frye,' ... [https://www.khflaw.com/news/legal-intelligencer-battle-of-the-experts-standards-frye-daubert-and-federal-rule-of-evidence-702/]
- 9. Federal Rule of Evidence 702 [https://harvardlawreview.org/print/vol-138/federal-rule-of-evidence-702/]
- 10. Standards For Testimony: Frye Vs Daubert [https://mcdowellowens.com/about-us/articles/testimony-or-frye-v-daubert]
- 11. . Daubert : A State-by-State vs Frye Comparison [https://www.expertinstitute.com/resources/insights/daubert-versus-frye-a-national-look-at-expert-evidentiary-standards/]
- 12. Daubert Standard | Wex | LII / Legal Information Institute [https://www.law.cornell.edu/wex/daubert_standard]
- 13. Daubert standard [https://en.wikipedia.org/wiki/Daubert_standard]
- 14. Rule 702. Testimony by Expert Witnesses - Cornell Law School [https://www.law.cornell.edu/rules/fre/rule_702]
- 15. Forensic Chemistry | Technology Readiness Level 4 [https://www.sciencedirect.com/special-issue/101HG68D51W]
- 16. Benefits of ILC/PT (Inter-Laboratory Comparisons ... [https://blog.ansi.org/anab/benefits-ilc-pt-inter-laboratory-testing/]
- 17. Proficiency Testing and Interlaboratory Comparisons : The Ultimate... [https://www.isobudgets.com/proficiency-testing-and-interlaboratory-comparisons/]
- 18. Criteria for Validation of Methods in Microbial Forensics [https://pmc.ncbi.nlm.nih.gov/articles/PMC2547046/]
- 19. Why participate to an inter laboratory comparison ? [https://www.compalab.org/pages/what-is-an-ilc/why-participate-to-an-interlaboratory-comparison.html]
- 20. Collaborative Testing Services Inc. -- Proficiency and ... [https://collaborative-testing.com/index-collaborative-testing.php]
- 21. Use of Proficiency Testing as a Tool to Improve Quality in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4696484/]
- 22. Technology Readiness Levels - NASA [https://www.nasa.gov/directorates/somd/space-communications-navigation-program/technology-readiness-levels/]
- 23. Method validation in digital forensics (accessible) - GOV.UK [https://www.gov.uk/government/publications/method-validation-in-digital-forensics/method-validation-in-digital-forensics-accessible]
- 24. Likelihood ratio data to report the validation of a forensic fingerprint evaluation method - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5144651/]
- 25. A guide to measuring expert performance in forensic pattern matching | Behavior Research Methods [https://link.springer.com/article/10.3758/s13428-024-02354-y]
- 26. Method Validation and Robustness [https://www.chromatographyonline.com/view/method-validation-and-robustness]
- 27. Scientific guidelines for evaluating the validity of forensic feature-comparison methods - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10576079/]
- 28. A guide to measuring expert performance in forensic ... [https://pubmed.ncbi.nlm.nih.gov/38485882/]
- 29. An algorithm for forensic toolmark comparisons [https://www.sciencedirect.com/science/article/pii/S2589871X24000901]
- 30. A guide to ISO 21043 Forensic Sciences from the ... [https://www.sciencedirect.com/science/article/pii/S1355030625000887]
- 31. What is forensic validation and how is it used? [https://www.envistaforensics.com/knowledge-center/insights/articles/forensic-validation-pay-attention-to-the-man-behind-the-curtain/]
- 32. A Statistical Framework for Consensus-Based Reliability ... [https://sciety.org/articles/activity/10.21203/rs.3.rs-8093408/v1?utm_source=sciety_labs_article_page]
- 33. A Statistical Framework for Analysis of Trial-Level ... [https://elifesciences.org/reviewed-preprints/95802]
- 34. A framework to analyze opinion formation models [https://www.nature.com/articles/s41598-022-17348-z]
- 35. Integrated Technology Readiness Levels (TRLs) [https://medicalcountermeasures.gov/trl/integrated-trls]
- 36. Geoffrey Stewart Morrison [https://geoff-morrison.net/]
- 37. (PDF) Verification of analytical methods for GMO testing when... [https://www.academia.edu/8071647/Verification_of_analytical_methods_for_GMO_testing_when_implementing_interlaboratory_validated_methods_Guidance_document_from_the_European_Network_of_GMO_laboratories_ENGL]
- 38. Investigating methods for forensic analysis of social media ... [https://www.frontiersin.org/journals/computer-science/articles/10.3389/fcomp.2025.1566513/full]
- 39. Retrospective data analysis and proposal of a practical acceptance ... [https://pubmed.ncbi.nlm.nih.gov/25124547/]
- 40. The future of forensic DNA analysis - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC4580997/]
- 41. Accounting for inter-laboratory DNA recovery data ... [https://www.sciencedirect.com/science/article/pii/S1872497325000638]
- 42. Modeling sources of interlaboratory variability in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5966732/]
- 43. Forensic interlaboratory evaluations of a systematic ... [https://www.sciencedirect.com/science/article/abs/pii/S2468170923000231]
- 44. Collaborative versus traditional method validation approach [https://pmc.ncbi.nlm.nih.gov/articles/PMC7452674/]
- 45. Iterative outlier removal: A method for identifying ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4927349/]
- 46. Outliers, Part III: Dealing with Outliers [https://www.spectroscopyonline.com/view/outliers-part-iii-dealing-outliers]
- 47. TRL 4 (Technology Readiness Level), from laboratory to ... [https://techfinders.io/project/trl-4-technology-readiness-level-explained-from-lab-prototype-to-simulation/]
- 48. TRL 4-5: Laboratory and Relevant Environment Validation [https://www.mcginniscommawill.com/posts/2025-02-28-trl-validation-stages/]
- 49. Modern Instrumental Methods in Forensic Toxicology - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2745311/]
- 50. A comparison of four liquid chromatography-mass ... - PubMed [https://pubmed.ncbi.nlm.nih.gov/37432442/]
- 51. A comparison of four liquid chromatography–mass ... [https://link.springer.com/article/10.1007/s00216-023-04791-8]
- 52. Quality Control Testing of Reagents and Consumables [https://nij.ojp.gov/nij-hosted-online-training-courses/dna-amplification/contamination/prevention/quality-control-testing-reagents-and-consumables]
- 53. Comparison of Guide to Expression of Uncertainty in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11902841/]
- 54. 7 Steps to Calculate Measurement Uncertainty [https://www.isobudgets.com/7-steps-to-calculate-measurement-uncertainty/]
- 55. Experimenting with reproducibility: a case study of robustness ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6054242/]
- 56. Reproducibility and robustness [https://armgpublishing.com/journals/hem/reproducibility-and-robustness/]
- 57. Improving transparency and scientific rigor in academic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6346653/]
- 58. Subscribe to Forensic Chemistry - 2468-1709 [https://shop.elsevier.com/journals/forensic-chemistry/2468-1709]
- 59. In the context of forensic casework, are there meaningful ... [https://www.sciencedirect.com/science/article/pii/S2589871X21000279]
- 60. Comparison of Capillary Electrophoresis and HPLC-Based ... [https://www.mdpi.com/2304-8158/14/15/2623]
- 61. Choices of chromatographic methods as stability indicating ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8027279/]
- 62. Development and validation of a capillary electrophoresis ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967397000903]