Advanced Post-Blast Residue Analysis: Modern Sampling, Concentration, and Analytical Techniques for Forensic Investigations
This article provides a comprehensive overview of modern strategies for the sampling and concentration of post-blast residues, a critical step in forensic explosive investigation.
Advanced Post-Blast Residue Analysis: Modern Sampling, Concentration, and Analytical Techniques for Forensic Investigations
Abstract
This article provides a comprehensive overview of modern strategies for the sampling and concentration of post-blast residues, a critical step in forensic explosive investigation. It explores the foundational challenges of residue collection from complex matrices and environmental conditions. The scope extends to detailed methodological applications of both classical and advanced instrumental techniques, including Gas Chromatography-Mass Spectrometry (GC-MS), Ion Chromatography (IC), and Fourier Transform Infrared (FTIR) spectroscopy. The article further addresses troubleshooting and optimization protocols for challenging scenarios, such as rainy environments or oversized evidence, and offers a comparative validation of analytical methods, including the growing role of chemometrics and machine learning for enhanced classification and data interpretation, providing a complete framework for researchers and forensic professionals.
Understanding Post-Blast Residues: Composition, Challenges, and Forensic Significance
In the realm of forensic chemistry and counter-terrorism, post-blast residue analysis serves as a critical investigative tool for determining the origin and composition of explosive devices. Post-blast residues are the complex chemical mixtures that remain after an explosive detonation, comprising both unreacted explosive materials and a wide array of decomposition products [1]. These residues present significant analytical challenges due to their trace quantities, complex matrices, and potential for environmental degradation. The accurate characterization of these materials requires sophisticated sampling methodologies, meticulous extraction protocols, and advanced analytical techniques to overcome the inherent difficulties of matrix interference and analyte instability [1] [2].
This technical guide examines the fundamental nature of post-blast residues within the broader context of research on sampling and concentration techniques. It establishes a comprehensive framework for understanding residue composition, outlines standardized analytical approaches, and presents recent advancements in detection methodologies that enhance forensic capabilities for investigating explosive incidents.
Composition and Classification of Post-Blast Residues
Post-blast residues originate from the incomplete reaction of an explosive charge during detonation. Their chemical profile is influenced by multiple factors including the explosive composition, detonation efficiency, and environmental conditions surrounding the blast event [3].
Primary Components
The principal constituents of post-blast residues can be categorized into three distinct groups:
- Unexploded Materials: Microscopic particles of the original explosive that survive detonation without reacting. These particles represent the most forensically valuable evidence as they directly reflect the pre-detonation composition. Studies indicate that low-order explosions (characterized by incomplete detonation due to device failure or material deterioration) yield substantially higher quantities of unexploded materials compared to high-order explosions [3].
- Decomposition Intermediates: Partial reaction products formed through incomplete combustion or thermal degradation of the original explosive material. For TNT (2,4,6-trinitrotoluene), these typically include 2,4-dinitrotoluene (DNT) and 1,3-dinitrobenzene (DNB) [4].
- Transformation Products: Compounds generated through environmental or microbial action on post-blast residues over time. The aging process of TNT residues in soil, for instance, produces 2-aminodinitrotoluene (2-ADNT) and 4-aminodinitrotoluene (4-ADNT) as principal microbial degradation products [4].
Distribution and Aging Characteristics
The dispersal and persistence of post-blast residues in the environment follow predictable patterns that inform sampling strategies:
- Spatial Distribution: Research on controlled detonations of aluminised ammonium nitrate and RDX-based compositions demonstrates that post-blast residue mass distributes according to an approximate inverse-square law model, though distribution trends vary significantly between individual analytes [5]. The dominant mechanism affecting dispersal is the wind-directed smoke plume from the detonation, rather than blast wave mechanics alone [5].
- Aging Dynamics: The chemical signature of post-blast residues evolves over time through environmental degradation. Studies quantifying TNT landmine post-blast residues in soil have established degradation half-lives of approximately 40 days for 2,4-DNT and 100 days for TNT itself under controlled moisture conditions [4]. These transformation processes fundamentally alter residue profiles, with DNT/TNT ratios in post-blast residue typically measuring between 1 and 0.01, distinctly different from pre-blast leakage patterns [4].
Table 1: Key Characteristics of Common Explosive Residues
| Explosive | Primary Residue Markers | Decomposition Products | Aging Characteristics |
|---|---|---|---|
| TNT | Unexploded TNT particles | 2,4-DNT, 1,3-DNB | Degrades to 2-ADNT & 4-ADNT; Half-life: ~100 days |
| PETN | Intact PETN (trace amounts) | Thermal degradation compounds | Thermally labile; fragments in GC-EI-MS analysis |
| RDX | Unexploded RDX crystals | Nitramine derivatives | Distributed via smoke plume; follows inverse-square law |
| TATP | TATP vapor, DADP | Acetone, hydrogen peroxide | High vapor pressure; sublimes readily at crime scenes |
Analytical Challenges in Residue Analysis
The forensic detection and identification of post-blast residues present multiple technical obstacles that complicate analysis and interpretation.
Fundamental Limitations
Analysts encounter several inherent difficulties when working with post-blast evidence:
- Trace Concentration: Explosive residues persist in minimal quantities following detonation, often at nanogram to picogram levels, necessitating highly sensitive detection methods and efficient concentration techniques [1] [6].
- Matrix Complexity: Residues typically distribute across heterogeneous surfaces and absorb into porous materials like soil, introducing substantial chemical interference that complicates extraction and analysis [1]. Soil samples particularly challenge analysts due to the presence of humic acids, inorganic salts, and organic contaminants that co-extract with target analytes [1].
- Analyte Instability: Many explosive compounds demonstrate limited environmental persistence or thermal lability. PETN undergoes extensive fragmentation in standard GC-EI-MS systems due to thermal decomposition in the injection port and ion source, while TATP sublimes readily at ambient temperatures, creating significant vapor-phase losses [1] [2].
Methodological Considerations
The analytical process introduces additional complications that require careful methodological control:
- Recovery Efficiency: Extraction techniques exhibit varying efficiencies for different explosive compounds. Soxhlet warm extraction methods recover approximately 60% of nitrotoluenes from soil matrices, highlighting significant analyte losses during sample preparation [1].
- Detection Specificity: Complex residue mixtures challenge the discrimination capabilities of analytical systems, particularly when distinguishing between structurally similar compounds or decomposition products with nearly identical mass spectra or retention times [1] [3].
Experimental Protocols for Residue Analysis
Standardized methodologies ensure reliable and reproducible analysis of post-blast residues across different laboratory environments.
Sample Collection and Extraction
Systematic approaches to sample handling form the foundation of effective residue analysis:
- Artificial Explosion Protocol: For method development and validation, researchers employ standardized procedures where trace amounts of investigated explosives (ranging from milligrams to nanograms) are individually mixed with 100g of sand or soil. These controlled samples undergo extraction optimization to establish filtration, clean-up, and concentration protocols before analysis [1].
- Solid-Phase Extraction (SPE): Following collection, residue extracts undergo SPE clean-up to remove matrix interferences and concentrate target analytes. This step is particularly crucial for soil samples where co-extracted compounds can suppress analyte response in chromatographic systems [1].
- Headspace Sampling for Volatiles: For highly volatile explosives like TATP, passive headspace sampling and solid-phase microextraction (SPME) techniques effectively capture vapor-phase analytes while minimizing matrix contributions. These approaches essentially concentrate the sample and provide superior detection limits compared to direct solvent extraction [2].
Analytical Detection Methods
A multi-technique approach leveraging complementary analytical platforms provides the most comprehensive residue characterization:
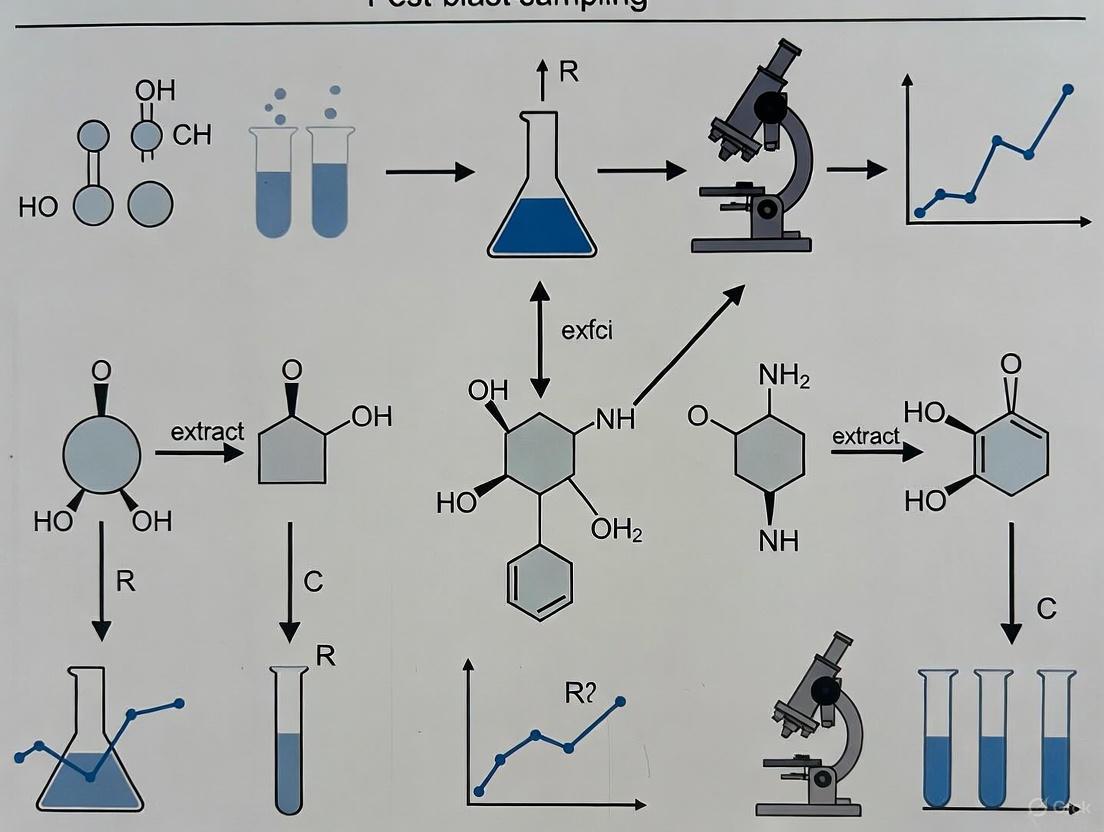
- Color Tests and TLC Screening: Traditional spot tests using reagents including diphenylamine in sulfuric acid, brucine sulfate, and Griess reagent provide rapid screening for explosive residues [1]. Subsequent Thin Layer Chromatography employing solvent systems like trichloroethylene-acetone (4:1) separates compounds including TNT, PETN, and tetryl, with visualization enhanced by spraying plates with sodium hydroxide followed by Griess reagent after heating [1].
- Chromatographic-Spectrometric Analysis: Gas Chromatography-Mass Spectrometry (GC-MS) with electron impact (EI) or chemical ionization (CI) sources provides confirmatory testing for organic explosives, though method optimization is required for thermally labile compounds like PETN [1] [2]. Ion Mobility Spectrometry (ITMS) offers rapid analysis with excellent mass detection limits and minimal sample requirements, making it particularly valuable for screening post-detonation debris [1].
- Spectroscopic Techniques: Fourier Transform Infrared (FTIR) Spectroscopy enables identification of explosive materials through molecular fingerprinting, requiring minimal sample material and providing high sensitivity and selectivity. When combined with machine learning classification techniques like LDA-PCA, FTIR can successfully identify explosive materials even in high-order explosion remnants [3].
Diagram 1: Analytical Workflow and Key Challenges
Quantitative Analysis of Post-Blast Residues
Method validation requires establishing performance characteristics for each analytical technique to ensure reliable quantification.
Table 2: Detection Limits and Method Performance for Explosive Residue Analysis
| Analytical Technique | Target Analytes | Typical LOD | Key Applications | Limitations |
|---|---|---|---|---|
| Ion Mobility Spectrometry (IMS) | Organic explosives | pg–ng | Rapid screening of debris, minimal sample requirement | Medium specificity, matrix effects |
| GC-MS (EI/CI) | PETN, TNT, TATP, degradation products | pg–ng (varies by compound) | Confirmatory testing, structural identification | Thermal decomposition of labile compounds |
| LC-MS | Thermally labile explosives, precursors | pg–ng | Analysis of compounds unsuitable for GC | Requires appropriate ionization techniques |
| FTIR Spectroscopy | Functional group identification | μg range | Molecular fingerprinting, minimal sample prep | Limited sensitivity vs. MS methods |
| SPME-GC-MS (TATP) | Volatile peroxide explosives | 0.6 ng (CI), 0.9 ng (EI) | Headspace sampling, minimal interference | Specialized sampling equipment required |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful residue analysis requires specific chemical reagents and reference materials tailored to explosive compounds.
Table 3: Essential Research Reagent Solutions for Explosive Residue Analysis
| Reagent/Material | Composition/Type | Primary Function | Application Notes |
|---|---|---|---|
| Griess Reagent | Sulfanilamide, N-(1-naphthyl)ethylenediamine | Detection of nitrite compounds from nitrate ester degradation | Enhanced visualization after NaOH spray and heating |
| Diphenylamine Reagent | 1 mg diphenylamine in 10 mL conc. H₂SO₄ | Screening test for oxidizing agents | Traditional color test for explosives |
| Brucine Sulfate Reagent | 0.5 g brucine sulfate in 10 mL conc. H₂SO₄ | Nitrate and nitrite compound detection | Spot test application |
| Alcoholic KOH | 1 g KOH pellets in 10 mL absolute alcohol | Hydrolysis agent for explosive compounds | TLC visualization reagent |
| Trichloroethylene-Acetone | 4:1 ratio mixture | Mobile phase for TLC separation | Separates TNT, PETN, and tetryl |
| SPME Fibers | Various coatings (e.g., PDMS, CAR/PDMS) | Headspace concentration of volatile analytes | Optimal for TATP sampling |
| SPE Cartridges | C18, silica, specialized sorbents | Sample clean-up and concentration | Reduces matrix interference before analysis |
Advanced Technical Approaches
Recent methodological innovations have significantly enhanced capabilities for detecting and characterizing challenging residue compounds.
Enhanced Detection of Problematic Analytes
Specific explosive compounds require specialized analytical approaches to overcome their inherent chemical properties:
- PETN Analysis: The development of GC-EI-MS methods without additional external tools or special manipulation of regular settings enables more reliable PETN detection. Supplemental techniques include Nitrate ester derivatization for generating structurally informative ions and GC/VUV (vacuum ultraviolet) spectroscopy to characterize thermal degradation products including nitric oxide, carbon monoxide, and formaldehyde [1].
- TATP Capture: The high vapor pressure and sublimation tendency of TATP necessitates specialized sampling approaches. Passive headspace sampling techniques coupled with GC-MS analysis effectively concentrate this volatile analyte from post-blast debris. Both electron impact (EI) and chemical ionization (CI) methods demonstrate similar detection limits (0.6-0.9 ng), providing flexibility in analytical configuration [2].
Data Analysis Innovations
Advanced computational methods augment traditional analytical techniques:
- Machine Learning Integration: The application of multivariate statistical analysis and machine learning classification techniques like the hybrid LDA-PCA method to FTIR spectral data enables successful identification of explosive materials in high-order explosion residues where visual spectrum interpretation proves inadequate [3].
- Aging and Degradation Modeling: Quantitative studies of residue transformation kinetics, such as the established 40-day half-life for 2,4-DNT and 100-day half-life for TNT in soil matrices, facilitate more accurate temporal assessment of blast events and residue evolution [4].
Diagram 2: Advanced Techniques for Specific Challenges
The definitive characterization of post-blast residues represents a multifaceted analytical challenge requiring integrated approaches that span sample collection, preparation, and instrumental analysis. The complex nature of these residues—encompassing both unexploded materials and decomposition products—demands sophisticated methodological frameworks that address the issues of trace concentration, matrix complexity, and analyte instability. Current research demonstrates that the combination of classical techniques like color tests and TLC with advanced instrumental methods including GC-MS, IMS, and FTIR spectroscopy provides the most comprehensive analytical coverage. Furthermore, the integration of machine learning algorithms for data interpretation and the development of compound-specific solutions for challenging analytes like PETN and TATP significantly enhance forensic capabilities. As sampling and concentration techniques continue to evolve within this research domain, the systematic approach outlined in this guide establishes a foundational protocol for reliable residue analysis that supports both forensic investigations and broader security initiatives.
Residue analysis represents a critical frontier in forensic science and public health safety, particularly in the context of post-blast investigations and food safety monitoring. This technical guide examines the three fundamental challenges that analysts face: detecting substances at trace levels, mitigating complex matrix effects, and preventing cross-contamination during sampling and analysis. The ability to overcome these hurdles directly impacts the accuracy of forensic attribution in security investigations and the reliability of safety assessments in food production chains. Recent advancements in analytical technologies and method standardization have significantly improved capabilities, yet these core challenges persist, demanding continued innovation and rigorous protocol development. This whitepaper synthesizes current methodologies and emerging solutions, providing a comprehensive resource for researchers and forensic professionals engaged in this high-stakes field.
Core Analytical Challenges
The fundamental obstacles in residue analysis manifest across various stages of the analytical workflow, from initial sampling to final quantification. The table below summarizes these primary challenges and their implications for analysis.
Table 1: Key Challenges in Residue Analysis
| Challenge | Impact on Analysis | Common Matrices Affected |
|---|---|---|
| Trace Amounts | Limits detection, requires highly sensitive instruments and preconcentration techniques [2] [3]. | Post-blast debris, low-level pesticide residues in food, environmental samples [2] [7]. |
| Matrix Interference | Causes signal suppression/enhancement, reduces analytical accuracy and precision [7] [8]. | High-fat foods, animal products, soil, complex post-blast debris [7] [9]. |
| Contamination Risks | Leads to false positives or introduction of external contaminants, compromising results [2] [3]. | Forensic evidence during collection/storage, laboratory samples during preparation [3]. |
The Problem of Trace Amounts
In post-blast scenarios, the target analytes are often present in nanogram or picogram quantities, demanding extreme analytical sensitivity. For instance, triacetone triperoxide (TATP), a common homemade explosive, is highly volatile and sublimes readily, leaving minimal trace evidence after an explosion [2]. Similarly, monitoring pesticide residues for food safety requires detecting concentrations that often must be below maximum residue limits (MRLs) set by regulatory bodies, sometimes in the parts-per-billion range [7] [8]. The minute quantities present a significant risk of failing to detect hazardous substances, with serious public health and security consequences. This challenge necessitates sophisticated instrumentation and often involves a sample pre-concentration step to boost the analyte signal above the instrument's detection limit.
Matrix Interference Complexities
Matrix effects represent one of the most persistent problems in residue analysis, where co-extracted compounds from the sample itself interfere with the detection and accurate quantification of the target analyte. In food analysis, animal-derived matrices with high fat and protein content are notoriously difficult, as lipids can cause significant signal suppression or enhancement in chromatographic systems [7]. In post-blast investigations, residues are collected from surfaces like plastic, fabric, or wood, which can leach interfering compounds during extraction [9] [3]. These interferences can obscure the target signal, alter retention times, or lead to incorrect compound identification. Without effective sample cleanup and analytical techniques that can correct for these effects, the reliability of quantitative results is severely compromised.
Contamination Risks
The risk of contamination permeates the entire analytical process, from field sampling to laboratory reporting. In forensic settings, cross-contamination at a crime scene can falsely link evidence or misdirect an investigation [3]. During the analysis of volatile explosives like TATP, their tendency to sublime poses an internal cross-contamination risk within laboratory instrumentation if not properly managed [2]. Furthermore, the use of solvents, reagents, and laboratory equipment can introduce contaminants such as plasticizers or previous analyte carryover, leading to false positives. Stringent quality control, appropriate sample storage, and the use of procedural blanks are essential, yet not foolproof, defenses against this ever-present challenge.
Advanced Detection and Analytical Techniques
Overcoming the core challenges requires a sophisticated arsenal of analytical instruments and methods. The selection of a technique depends on the analyte's chemical properties, the complexity of the matrix, and the required sensitivity.
Chromatographic and Mass Spectrometric Methods
Gas Chromatography (GC) and Liquid Chromatography (LC) coupled to Mass Spectrometry (MS) are cornerstone techniques for separating, identifying, and quantifying residues in complex mixtures.
- Gas Chromatography-Mass Spectrometry (GC-MS): This technique is ideal for volatile and thermally stable compounds. It is the benchmark for analyzing explosive residues like TATP, where methods have been developed using both electron impact (EI) and chemical ionization (CI) to achieve limits of detection in the sub-nanogram range [2]. GC-MS is also widely used for multi-residue pesticide analysis, particularly for GC-amenable pesticides [7].
- Liquid Chromatography-Mass Spectrometry (LC-MS): LC-MS, especially tandem mass spectrometry (LC-MS/MS) and high-resolution mass spectrometry (HRMS), has become indispensable for analyzing non-volatile, thermally labile, and polar compounds. It is the dominant technique for comprehensive pesticide screening in food, enabling the detection of hundreds of analytes in a single run [10] [7]. HRMS is particularly valuable for non-targeted screening and exposomics, as it allows for the retrospective analysis of data to identify unexpected residues or transformation products [7].
Table 2: Comparison of Key Analytical Techniques for Residue Analysis
| Technique | Best For | Typical LOD | Advantages | Limitations |
|---|---|---|---|---|
| GC-MS | Volatile explosives (TATP), non-polar pesticides [2] [7]. | ~0.6 ng (for TATP) [2]. | Robust, extensive libraries, high resolution. | Limited to volatile/stable compounds; derivatization often needed. |
| LC-MS/MS | Non-volatile pesticides, metabolites, polar compounds [7] [8]. | Low ppt-ppb levels [7]. | High sensitivity & selectivity; broad analyte coverage. | Matrix effects can be severe; instrument cost is high. |
| FTIR Spectroscopy | Functional group identification, post-blast residue screening [9] [3]. | Varies by compound and matrix. | Non-destructive, minimal sample prep, molecular fingerprinting. | Lower sensitivity than MS; complex mixture analysis is difficult. |
| Ion Mobility Spectrometry (IMS) | Rapid screening, separating isobaric ions [7]. | Varies by compound. | Adds a separation dimension to LC-MS; portable units available. | Limited resolving power; requires reference standards. |
Spectroscopic and Spectrometric Techniques
- Fourier-Transform Infrared (FTIR) Spectroscopy: FTIR provides a molecular "fingerprint" based on vibrational energy transitions. Its utility in post-blast investigations has been demonstrated for identifying unreacted explosives like C-4, PETN, and TNT on various surfaces after both high- and low-order explosions [3]. While its sensitivity is generally lower than MS, it requires minimal sample preparation and is non-destructive, allowing for further analysis.
- Ion Mobility Spectrometry (IMS): Often coupled with LC-HRMS, IMS separates ions based on their size, shape, and charge as they drift through a gas. This adds an orthogonal separation dimension, helping to resolve isobaric and isomeric interferences that are common in complex matrices like food extracts, thereby improving identification confidence [7].
- Single-Particle Mass Spectrometry: An emerging technique for real-time detection and identification of post-blast explosive particles, offering insights for forensic attribution [11].
Experimental Protocols and Workflows
Standardized and robust experimental protocols are essential for generating reliable, reproducible, and legally defensible data in residue analysis. The following section details key methodologies cited in recent literature.
Protocol 1: Passive Headspace Sampling and GC-MS for Post-Blast TATP
This protocol, adapted from Bain et al. (2025), is designed to capture the volatile explosive TATP from post-blast debris [2].
- Principle: Exploits the high vapor pressure of TATP to passively concentrate analyte vapors from the headspace of a sealed container onto a solid-phase microextraction (SPME) fiber or into a sorbent tube.
- Sample Preparation: Post-blast debris is collected and sealed in a headspace vial. For passive headspace sampling, a sorbent tube or SPME fiber is suspended in the vial's headspace.
- Extraction: The sealed vial is heated to 60°C for a defined period (e.g., 30 minutes) to facilitate the transfer of TATP vapor from the matrix to the headspace and onto the SPME fiber/sorbent.
- Analysis: The SPME fiber is thermally desorbed in the GC inlet, or the sorbent tube is thermally desorbed, and analytes are separated on a GC column. Detection is achieved via MS using either electron impact (EI; monitoring m/z 59) or chemical ionization (CI; monitoring m/z 240) [2].
- Key Advantage: This method is highly effective for concentrating the volatile analyte, provides less matrix interference compared to direct solvent extraction, and achieves excellent limits of detection (LOD of 0.6 ng with CI and 0.9 ng with EI) [2].
Diagram 1: Workflow for Passive Headspace Sampling of TATP.
Protocol 2: QuEChERS Extraction for Multi-Residue Pesticide Analysis
The QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) method is a standard approach for extracting pesticides from diverse food matrices [7] [8].
- Principle: Involves liquid-liquid partitioning using acetonitrile and salt-induced separation, followed by a dispersive solid-phase extraction (d-SPE) clean-up to remove interfering matrix components.
- Sample Preparation: A homogenized food sample (e.g., date fruit) is weighed into a centrifuge tube.
- Extraction: Acetonitrile is added, and the tube is shaken. Anhydrous magnesium sulfate (MgSO₄) is added to remove water, and salts like sodium chloride (NaCl) are added to induce partitioning. The tube is shaken vigorously and centrifuged.
- Clean-up: An aliquot of the upper acetonitrile layer is transferred to a d-SPE tube containing sorbents (e.g., primary secondary amine (PSA) for removing fatty acids, C18 for removing non-polar interferences, and MgSO₄). The tube is shaken and centrifuged.
- Analysis: The purified extract can be analyzed directly or after concentration by both LC-MS/MS and GC-MS/MS to achieve broad coverage of pesticides with varying polarities and volatilities. Reported recoveries for many pesticides in date fruit using this method range from 77% to 119% [7].
Diagram 2: QuEChERS Extraction and Clean-up Workflow.
Protocol 3: FTIR Spectroscopy with Machine Learning for Post-Blast Residue Classification
This protocol, based on work published in Molecules (2023), uses FTIR spectroscopy combined with chemometrics to classify explosive residues [3].
- Principle: FTIR spectra serve as unique molecular fingerprints for explosives. Machine learning models are trained to recognize patterns in these spectra, enabling classification even when the signal is weak or mixed with interferences.
- Sample Preparation: Post-blast residues are collected from witness materials (e.g., metal, plastic) using acetone rinsing or direct sampling. The residue is mixed with potassium bromide (KBr) and pressed into a pellet for transmission mode analysis.
- Spectral Acquisition: FTIR spectra are collected in the mid-infrared range (4000–400 cm⁻¹) at a high spectral resolution (e.g., 4 cm⁻¹). Multiple scans are averaged to improve the signal-to-noise ratio.
- Data Pre-treatment and Modeling: The exported spectral data is processed in a statistical environment like R. Pre-treatment steps include baseline correction and normalization. Machine learning techniques, such as a hybrid Linear Discriminant Analysis - Principal Component Analysis (LDA-PCA) model, are then trained to classify the residues (e.g., as C-4, PETN, or TNT) based on their spectral features [3].
- Key Advantage: This method is non-destructive and, when combined with machine learning, provides a powerful tool for the automated and objective identification of complex post-blast residues.
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful residue analysis relies on a suite of specialized reagents and materials designed to optimize extraction, clean-up, and detection.
Table 3: Essential Reagents and Materials for Residue Analysis
| Item | Function | Application Example |
|---|---|---|
| SPME Fibers | Concentrates volatile & semi-volatile analytes from headspace or liquid [2]. | Passive headspace sampling of TATP in post-blast debris [2]. |
| QuEChERS Kits | Provides pre-measured salts & sorbents for standardized sample prep [7] [8]. | Multi-residue pesticide extraction from fruits and vegetables [7]. |
| d-SPE Sorbents (PSA, C18, GCB) | Removes specific matrix interferences (acids, lipids, pigments) during clean-up [7]. | Cleaning up fatty acids from fruit extracts or pigments from leafy vegetables [7]. |
| Deuterated Internal Standards | Corrects for matrix effects & losses during sample prep; improves quantification [7]. | Used in LC-MS/MS and GC-MS analysis of pesticides and explosives for accurate results. |
| High-Purity Solvents (Acetonitrile, Acetone) | Extracts analytes from solid matrices without introducing interfering contaminants [3]. | Solvent extraction of explosives from swabs or pesticides from food [7] [3]. |
| KBr (Potassium Bromide) | Transparent to IR light; used to create pellets for FTIR spectroscopic analysis [3]. | Preparing solid samples for FTIR analysis of post-blast residues [3]. |
The field of residue analysis remains a dynamic and challenging discipline, continuously evolving to meet the demands of detecting ever-smaller quantities of analytes in increasingly complex matrices. While techniques like HRMS, passive headspace sampling, and advanced chemometrics have dramatically improved capabilities, the fundamental challenges of trace detection, matrix interference, and contamination control persist. The future path points toward greater integration of automation for reproducibility, the application of machine learning for data interpretation, and the development of green chemistry principles in sample preparation. Furthermore, harmonizing methodologies and data reporting standards across laboratories will be crucial for building comparable and reliable datasets. For researchers and forensic professionals, a deep understanding of these core challenges and the available tools to overcome them is paramount to ensuring accurate results that uphold public safety and security.
The forensic investigation of explosive incidents relies heavily on the successful recovery and identification of post-blast residues. These chemical traces provide critical evidence for attributing the device to a specific manufacturer or supplier, understanding the perpetrator's capabilities, and supporting legal proceedings. However, the integrity of this evidence is profoundly influenced by environmental conditions, with rainfall representing a primary factor in residue degradation and loss. This technical guide examines the impact of rainfall on explosive residue retention and detection, framing the discussion within the broader context of post-blast residue sampling and concentration techniques. For researchers and forensic professionals, understanding these dynamics is essential for developing robust analytical protocols that can overcome environmental challenges and yield forensically significant results even in suboptimal conditions.
Rainfall-Induced Challenges for Post-Blast Residue Analysis
Rainfall affects explosive residues through multiple mechanisms that complicate detection and analysis. The dissolution and transport of water-soluble explosive compounds, such as ammonium nitrate and inorganic oxidizers, can rapidly deplete residue concentrations at the crime scene. Even for less soluble organic explosives, hydrodynamic displacement can physically remove particulate evidence from surfaces. Furthermore, the introduction of moisture can accelerate chemical degradation processes; for instance, organic peroxide homemade explosives (HMEs) are known to degrade rapidly, and water exposure can hydrolyze certain functional groups critical for identification [12].
The complexity of post-blast samples is further exacerbated by environmental matrix interference. Rainfall introduces a multitude of environmental contaminants and other congeners that can mask target analytes during instrumental analysis [12]. This matrix interference is particularly problematic for techniques like ion mobility spectrometry (IMS), where environmental contaminants can reduce discriminating power [12]. The presence of water also creates practical challenges for sample collection and preservation, potentially promoting microbial activity that can biologically degrade explosive compounds if samples are not properly stabilized.
Analytical Techniques for Challenging Environmental Samples
Technique Selection Criteria
When analyzing rainfall-affected residues, the selection of analytical methods must be guided by three principal criteria: sensitivity, selectivity, and specificity. Sensitivity is crucial because rainfall dilutes residues to trace levels, requiring detection capabilities in the parts-per-million (ppm) to parts-per-billion (ppb) range [13]. Selectivity allows the instrument to respond to target analytes in complex mixtures containing environmental interferents, while specificity enables unambiguous identification—a forensic necessity for legal proceedings [13].
Advanced Instrumental Approaches
Chromatography-Mass Spectrometry Platforms: Gas chromatography coupled with vacuum ultraviolet spectroscopy (GC-VUV) has emerged as a powerful tool for explosives analysis. GC-VUV can achieve detection in the low ppm range while effectively discriminating interferents based on distinct VUV absorption patterns [13]. Similarly, liquid chromatography quadrupole time-of-flight mass spectrometry (LC-QToF-MS) provides high sensitivity and selectivity for organic explosives and propellants [12].
Ambient Ionization Techniques: Direct Analysis in Real Time mass spectrometry (DART-MS) represents a significant advancement for analyzing environmentally challenged samples. DART-MS eliminates extensive sample processing by directly thermally desorbing and ionizing substances from specimens or swabs [12]. This ambient ionization technique is particularly valuable for moist samples, as the thermal desorption process can help overcome moisture-related issues.
Spectroscopic Methods with Multivariate Analysis: Fourier Transform Infrared (FTIR) spectroscopy requires minimal sample and provides molecular fingerprinting capabilities. When combined with machine learning classification techniques like the hybrid LDA-PCA approach, FTIR can successfully identify explosive materials in residues after both high- and low-order explosions [3]. This statistical approach is particularly valuable for complex samples where visual spectrum interpretation is inadequate.
Table 1: Comparative Analysis of Techniques for Rainfall-Affected Residues
| Analytical Technique | Sensitivity | Key Advantage for Wet Samples | Limitations |
|---|---|---|---|
| GC-VUV | Low ppm range | Selective filtering cancels interferents | Requires method development for new analytes |
| DART-MS | High (picogram) | Minimal sample preparation; direct analysis | Method validation for ISO17025 requirements needed |
| LC-QToF-MS | High | Confirmatory analysis with forensic identification | Laborious sample processing typically required |
| FTIR with Machine Learning | Varies by compound | Identifies materials despite complex backgrounds | Requires reference spectral database |
Experimental Protocols for Rainfall Impact Assessment
Simulated Rainfall and Residue Recovery Studies
To systematically evaluate rainfall effects on residue retention, controlled studies employing simulated rainfall conditions provide valuable quantitative data. The following protocol outlines a standardized approach:
Sample Preparation: Deposit standardized quantities of target explosives (e.g., C-4, TNT, PETN, ANFO) onto representative substrates commonly encountered at crime scenes, including glass, steel, plastic, wood, and fabric [3]. Substrates should be cleaned and characterized prior to explosive application to establish baseline conditions.
Rainfall Simulation: Expose samples to simulated rainfall of varying intensities (e.g., 5-50 mm/hr) and durations (0.5-24 hours) using calibrated rainfall simulators. Collect runoff water at timed intervals to quantify residue wash-off rates. Control samples protected from rainfall provide reference data.
Sample Collection and Extraction: Following rainfall exposure, collect residues using standardized swabbing techniques with both dry swabs and swabs moistened with solvents such as acetone or acetonitrile [12] [3]. For porous substrates, consider excision of material followed by solvent extraction. Transfer swab extracts or solvent extracts to appropriate vials for analysis.
Analysis and Quantification: Analyze extracts using GC-VUV, DART-MS, or LC-MS/MS depending on target analytes. Employ internal standards to correct for recovery variations. Compare results against control samples to calculate percentage recovery under different rainfall scenarios.
Data Interpretation Guidelines
Statistical analysis should focus on identifying significant correlations between rainfall parameters (intensity, duration, total accumulation) and residue recovery rates. Multivariate approaches can help disentangle the effects of rainfall from other environmental factors. Establishing regression relationships between rainfall exposure and analyte recovery provides predictive models for estimating initial explosive quantities based on residual amounts detected after rainfall events.
Research Reagent Solutions for Environmental Challenge Mitigation
Table 2: Essential Research Reagents for Rainfall-Affected Residue Analysis
| Reagent/Solution | Function | Application Notes |
|---|---|---|
| Acetone (>99.7%) | Solvent for residue extraction from swabs and substrates | Effective for a wide range of organic explosives; use ACS grade or better [12] |
| Acetonitrile (≥99.9%) | Alternative extraction solvent, particularly for LC-MS analysis | Higher selectivity for certain polar explosives; compatible with various detection methods [12] |
| Potassium Bromide (KBr) | Matrix for FTIR pellet preparation | Mix with residue in ~1:100 ratio; ensures proper transmission mode analysis [3] |
| Internal Standards (deuterated analogs) | Quantification standards for mass spectrometry | Correct for recovery variations; essential for accurate quantification in complex matrices |
| Cotton Swabs | Sample collection from surfaces | Use dry or solvent-moistened; validated for compatibility with analytical methods [12] |
Workflow for Post-Blast Investigation in Rainfall Conditions
The following diagram illustrates a systematic workflow for investigating post-blast scenes exposed to rainfall, integrating specialized sampling and analytical approaches to overcome environmental challenges.
Rainfall significantly complicates the detection and analysis of post-blast explosive residues through multiple physical and chemical mechanisms. Successful forensic investigation under these conditions requires specialized sampling strategies that prioritize protected surfaces, analytical techniques with sufficient sensitivity and selectivity to detect trace residues amid environmental interferents, and data interpretation that accounts for rainfall-induced losses. Future research should focus on quantifying compound-specific washoff rates across different substrate types, developing more effective concentration techniques for dilute aqueous samples, and establishing probabilistic models that back-calculate initial charge weights from residual amounts detected after rainfall exposure. Advancements in these areas will enhance the reliability of forensic conclusions and contribute to more effective investigation of security incidents occurring in diverse environmental conditions.
In the meticulous realm of post-blast investigation, the integrity of the entire analytical process hinges upon the initial, critical phase of sample collection. The complex and contaminated environment of a blast site presents significant challenges, where the amount of explosive residue remaining after detonation is often minimal and useful chemical signatures are prone to degradation [13]. Within this context, proper debris collection emerges as the primary defense against false positives—erroneous results that can misdirect investigations, incriminate innocent persons, or compromise national security. This technical guide, framed within broader research on post-blast residue sampling and concentration techniques, examines the foundational principles and methodologies that ensure analytical accuracy from the crime scene to the laboratory.
The Analytical Chain: Why Collection Integrity Dictates Outcomes
Forensic analysis of explosive residues relies on instrumental methods that must demonstrate exceptional sensitivity (response to low analyte levels), selectivity (ability to respond to an analyte in a complex mixture), and specificity (unambiguous identification of the analyte) [13]. The vulnerability of this process to false positives is intrinsic:
- Sensitivity and Contaminants: Highly sensitive instruments, capable of detecting parts-per-million or even parts-per-billion concentrations, cannot distinguish between residue originating from the explosive device and incidental environmental contaminants introduced via poor collection technique [13]. For example, a tool used to collect multiple samples without decontamination can transfer trace materials, creating a false associative link.
- Selectivity and Interference: Complex sample matrices can contain compounds that mask the target analyte or produce overlapping signals during analysis. Proper collection minimizes the introduction of such interferents, allowing the instrument to isolate the signal of interest effectively [13].
- Specificity and Ambiguity: Unambiguous identification is crucial for legal proceedings. Cross-contamination during collection can introduce compounds with similar structures to target explosives, leading to ambiguous results that fail to meet the standard for scientific and legal opinion [13].
The following workflow diagram illustrates how proper and improper collection protocols directly influence analytical outcomes:
Key Analytical Figures of Merit in Post-Blast Analysis
The performance of any analytical method used in explosives investigation is quantified by specific figures of merit. Understanding these metrics is essential for evaluating the impact of sample quality.
Table 1: Key Analytical Figures of Merit and Impact of Sample Integrity
| Figure of Merit | Definition | Role in Preventing False Positives | Impact of Poor Sample Collection |
|---|---|---|---|
| Sensitivity [13] | The instrument's response to low levels of an analyte; ability to avoid false negatives. | Allows detection of authentic trace residues without need for pre-concentration, which can introduce contaminants. | Inability to distinguish true analyte from introduced contaminants; increased false positive risk. |
| Selectivity [13] | The ability to respond to a specific analyte in a complex mixture of similar compounds. | Enables analysis of complex post-blast samples without extensive clean-up steps that can alter signatures. | Introduction of interferents that mask the analyte or produce similar signals, leading to misidentification. |
| Specificity [13] | The ability to unambiguously identify a specific analyte, often based on unique structural features. | Provides reliable, court-admissible evidence through unambiguous compound identification (e.g., nitroglycerin). | Introduction of compounds with similar structures creates ambiguity, undermining evidential value. |
Advanced Analytical Techniques and Their Reliance on Quality Samples
The reliance of advanced analytical techniques on pristine sample collection is exemplified by two NIJ-funded research projects focused on the reconstruction phase of explosives investigations.
Gas Chromatography-Vacuum Ultraviolet Spectroscopy (GC-VUV)
This analytical couples a gas chromatograph (GC), which separates complex mixtures, with a vacuum ultraviolet (VUV) spectrometer that detects analytes based on their unique absorption spectra in the 100-200 nm range [13].
- Methodology: A sample extract is vaporized and passed through a GC column, where components separate based on their affinity for the column walls. Each separated compound then flows into a VUV flow cell, and its spectrum is measured in real-time. The resulting VUV spectrum acts as a unique fingerprint [13].
- Dependence on Sample Purity: The selectivity of GC-VUV relies on certain functional groups absorbing in distinct spectral regions. A contaminated sample can introduce overlapping spectral signals, preventing the instrument from canceling out interferences and leading to false identifications [13]. Furthermore, the specificity of the method, achieved through statistical analysis of complex spectral data, can be compromised by contaminants that alter the spectral profile [13].
Isotopic Signature Analysis for Source Attribution
This technique investigates whether isotopic and chemical signatures that link an explosive to its manufacturing source remain preserved after detonation and can be recovered from a blast site [13].
- Methodology: Researchers conduct field detonations of explosives like RDX, TNT, and AN-AL in open environments. Post-blast residues are collected via swabbing surfaces and extracting residues from soil. These samples are processed and analyzed using mass spectrometry to measure isotopic ratios, which are then statistically compared to pre-blast signatures [13].
- Dependence on Sample Purity: The core of this method is linking a minuscule amount of post-blast residue to a pre-blast source. Any contamination during the swabbing or soil collection process introduces foreign isotopic signatures, which can create a false association (false positive) or obscure a true one. As noted in the research, recoverable amounts of high-order explosives like RDX and TNT are already minimal, making the sample's purity paramount [13].
The Scientist's Toolkit: Essential Materials for Reliable Sample Collection
The following reagents and materials are critical for maintaining sample integrity from collection to analysis.
Table 2: Essential Research Reagent Solutions and Materials for Post-Blast Residue Collection
| Item | Function & Importance |
|---|---|
| Sterile Swabs | Primary tool for collecting trace residues from surfaces. Must be sterile and made of material (e.g., cotton, polyester) that will not interfere with subsequent chemical analysis. |
| Solvent Kits | Pre-packaged, high-purity solvents (e.g., methanol, acetone) for wetting swabs or performing initial extractions on-site. Purity is critical to prevent contamination. |
| Separate Containers | Inert, airtight containers (e.g, glass vials, metal cans) for storing each sample individually. This is a primary defense against cross-contamination. |
| Clean Soil Extraction Tools | Tools like clean trowels or spatulas used for collecting soil samples from beneath the blast epicenter. Tools must be decontaminated or replaced between each sample. |
| Personal Protective Equipment (PPE) | Gloves, masks, and disposable coveralls. Prevents the introduction of contaminants from the collector's person (e.g., skin cells, fibers) into the sample. |
| Chain of Custody Documentation | Pre-printed forms and labels for recording sample location, time, and collector. Maintains legal integrity and sample identification, preventing mix-ups. |
The path to a conclusive and accurate forensic finding in a post-blast investigation is a continuous chain of interdependencies. As this guide has detailed, even the most sophisticated analytical technologies, such as GC-VUV and isotopic analysis, cannot compensate for samples compromised at the point of collection. The principles of sensitivity, selectivity, and specificity that underpin reliable laboratory results are directly supported or undermined by the rigor of debris collection protocols. Preventing false positives is not merely a function of laboratory procedure but is fundamentally determined at the blast site, through the disciplined application of sterile, systematic, and contamination-aware evidence gathering. Future research in concentration techniques must continue to be developed in tandem with improved, non-destructive collection methodologies to further advance the capabilities of forensic science.
A Practical Workflow: From Scene Sampling to Laboratory Analysis
The forensic investigation of explosive incidents is critical for reconstructing events, identifying explosive formulations, and supporting judicial processes [14]. The reliability of these investigations hinges on the initial steps of evidence collection and processing. This guide details standardized protocols for the systematic collection, sub-sampling, and handling of complex evidence, such as oversized exhibits, within the context of post-blast residue sampling and concentration techniques. These protocols are designed to maximize residue recovery, minimize contamination, and ensure the analytical reliability required for robust scientific and legal outcomes.
Core Principles of Evidence Collection
Accurate post-explosion sample collection is a fundamental step upon which all subsequent analysis depends. Even the most advanced analytical methods yield unreliable results if improper materials are collected at the crime scene [1]. Two overarching principles must guide the entire process:
- Quality Control for Minimizing Contamination: Proper debris collection helps avoid contamination and false positives, which are critical challenges when dealing with complex sample matrices [1].
- Adaptation to Sample Matrix: The process of detecting post-blast residues is inherently difficult due to the minimal quantities of unreacted materials present. Methods must be robust enough to handle trace amounts of analytes mixed with various contaminants [1].
Protocols for Swabbing and Sub-sampling
Sub-sampling for Efficient Analysis
Sub-sampling allows for the preservation of original evidence while enabling rapid or repeated analysis. A proven method, adapted from forensic biology for use with explosive swabs, involves transferring a portion of material from a primary swab to a secondary, mini-swab.
Detailed Experimental Protocol for Sub-sampling [15]:
- Primary Sample Collection: Biological material (e.g., 10 µL or 150 µL of blood for method validation) is collected on a standard, regular-sized flocked swab (e.g., 4N6FLOQSwabs code 4504 C).
- Sub-sampling Technique: A mini flocked swab with a rigid head (e.g., 4N6FLOQSwabs Subungual Shape code 40U022D) is moistened with 15 µL of ultra-pure DNA-free water. The mini-swab is then applied by rolling and tapping on the original, stained swab until the entire surface of the mini-swab head is visibly stained.
- Application to Explosives Residues: While the cited study used blood, this protocol is directly adaptable for explosive residues. The moistening solvent can be replaced with a suitable solvent like acetone or ether for specific organic explosives, allowing a portion of a primary swab sample to be used for rapid on-site screening (e.g., with ion mobility spectrometry) while preserving the main sample for confirmatory laboratory analysis.
This approach provides a flexible solution to the limitation of analytical systems that consume the entire sample [15].
Swabbing Oversized and Fragmented Exhibits
Oversized exhibits, such as vehicle fragments, concrete blocks, and metallic debris, present significant challenges due to uneven residue distribution [16] [14]. A sequential swabbing technique is recommended for these heterogeneous substrates.
Detailed Protocol for Sequential Swabbing of Oversized Exhibits [14]:
- Materials: Surgical syringe (10 ml capacity), absorbent cotton wool, solvents (Diethyl Ether, Acetone, Demineralized (DM) Water, Sodium Hydroxide solution, Pyridine), and syringe filters (0.22 µm).
- Procedure:
- The same exhibit area is swabbed sequentially with different solvents to recover both organic and inorganic explosive traces.
- The sequence is typically: Ether (for non-polar organics like diesel fuel) -> Acetone (for organic high explosives) -> Water (for inorganic ions like nitrates) -> Sodium Hydroxide (for other inorganics) -> Pyridine (for elemental sulfur).
- Complete swabbing is performed for each solvent using fresh cotton for each step.
- The extracts collected from the swabs are filtered using a 0.22 µm syringe filter to remove solid impurities.
- The filtrate is collected in a beaker and concentrated to 2-5 ml by evaporation at room temperature for subsequent analysis.
This integrated workflow, which combines targeted swabbing, sequential solvent extraction, and syringe filtration, has been proven highly effective for investigating oversized post-blast exhibits [16] [14].
Handling and Analysis of Oversized Exhibits
Challenges and Strategic Workflow
Conventional extraction techniques, optimized for small, homogeneous samples, are often insufficient for large-scale or heterogeneous exhibits, yielding low recovery rates and reduced sensitivity [14]. The primary challenge lies in the uneven residue distribution and contamination gradients across large surfaces [16]. A robust, integrated workflow is essential for managing this complexity. The following diagram illustrates the strategic approach to processing oversized evidence, from collection through analysis.
Efficacy of Syringe Filtration
A key step in the processing of extracts from oversized exhibits is filtration. Research demonstrates that syringe filtration produced the highest recovery yield by minimizing background interference [16]. Filtering the solvent extract through a 0.22 µm nylon syringe filter prior to concentration and instrumental analysis is critical for removing fine particulate matter that could otherwise interfere with analytical instrumentation and result in false negatives or inaccurate quantification [14].
Quantitative Data and Environmental Considerations
Impact of Environmental Factors on Nitrate Detection
The persistence of explosive residues, particularly inorganic ions, is significantly affected by environmental conditions. Nitrate ions, a common component of many explosives like ANFO and black powder, are highly water-soluble. A 2025 study systematically investigated the impact of rainfall on the retention and detectability of nitrate ions in post-blast soil samples [17].
Experimental Protocol for Simulating Rainfall Effects [17]:
- Pit Construction: Two experimental soil pits were constructed on a sloped surface, each 12 inches in diameter and 6 inches deep. One was a simulated blast pit, the other a control.
- Spiking and Rainfall Exposure: The simulated pit was spiked with 1 gram of potassium nitrate (KNO₃). Both pits were filled to 75% of their volume with water and subjected to natural drizzling rainfall for one week. Water levels were replenished with natural rainwater to maintain real environmental conditions.
- Sample Collection and Preparation: After drying, soil samples were collected from the top (SPT) and bottom (SPB) layers of the simulated pit, and from the top (CPT) and bottom (CPB) layers of the control pit. Nitrate ions were extracted from the soil using demineralized water, and the extracts were filtered, evaporated to dryness, reconstituted, and diluted for Ion Chromatography (IC) analysis.
Results of Nitrate Ion Concentration After Rainfall [17]: Table: Quantification of nitrate ions reveals clear vertical leaching.
| Sample Description | Nitrate Ion Concentration (ppm) |
|---|---|
| Standard Potassium Nitrate (1g) | 842.522 |
| Simulated Pit - Top Layer (SPT) | 38.242 |
| Simulated Pit - Bottom Layer (SPB) | 24.971 |
| Control Pit - Top Layer (CPT) | 14.316 |
| Control Pit - Bottom Layer (CPB) | 8.381 |
The data shows a clear vertical leaching effect, with higher nitrate concentrations detected in the top layer of the simulated pit compared to the bottom layer. This underscores the critical importance of stratified sampling (collecting from both surface and subsurface layers) at blast sites exposed to rain, as relying on a single surface sample greatly increases the risk of false negatives [17].
Integrated Analytical Workflow for Complex Evidence
The full analytical journey for a piece of evidence, from collection to identification, requires the integration of multiple techniques. The workflow below maps out the standard operating procedures for analyzing both organic and inorganic components of explosive residues, illustrating how different analytical methods are applied to various extracts.
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key reagents, solvents, and materials essential for conducting forensic explosive residue analysis as described in the protocols, along with their specific functions [1] [17] [14].
Table: Essential materials and reagents for explosive residue analysis.
| Item & Description | Primary Function in Analysis |
|---|---|
| Flocked Swabs (e.g., 4N6FLOQSwabs regular and mini) | Efficient collection and sub-sampling of residues from various surfaces. |
| Solvents: Diethyl Ether, Acetone, Demineralized (DM) Water | Sequential extraction of organic (non-polar and polar) and inorganic explosive residues. |
| Syringe Filters (0.22 µm pore size, Nylon) | Removal of fine particulate matter from extracts to prevent instrumental interference. |
| TLC Plates (Silica gel 60G F254, 20x20 cm) | Separation of complex mixtures of organic explosives for preliminary identification. |
| Chemical Reagents:• Griess Reagent• Diphenylamine (5% in ethanol)• Sodium Hydroxide solution | Visualization agents for colorimetric spot tests and TLC development to identify specific explosive compound classes (e.g., nitramines, nitrate esters). |
| IC Eluent (e.g., 8.0 mM Na₂CO₃ / 0.25 mM NaHCO₃) | Mobile phase for the separation and quantification of inorganic anions (e.g., nitrate, chlorate) via Ion Chromatography. |
The forensic analysis of post-blast residues represents one of the most analytically challenging scenarios in modern forensic science. Investigators are frequently presented with complex sample matrices containing trace amounts of target explosive analytes amidst a background of co-extracted interferents. The recovery and concentration of these residues are critical for subsequent identification and confirmation, processes that form the foundational evidence in legal proceedings related to terrorism, sabotage, and other criminal activities. This technical guide provides an in-depth evaluation of three core techniques—solvent extraction, solid-phase extraction (SPE), and syringe filtration—systematizing their application within a robust post-blast analysis workflow. The ultimate goal is to equip researchers and forensic scientists with the knowledge to select and optimize methods that maximize analyte recovery, minimize matrix effects, and ensure the analytical integrity of results, thereby strengthening the chain of evidence from the crime scene to the courtroom.
Core Techniques and Comparative Analysis
The selection of an appropriate extraction and concentration strategy is dictated by the sample matrix, the target analytes, and the required sensitivity. The following techniques are most commonly employed in tandem to meet these challenges.
Solvent Extraction
Solvent extraction is the fundamental first step for isolating organic explosive residues from solid matrices such as post-blast debris or hand swabs. The principle involves using an organic solvent, with acetone being widely used, to dissolve and extract the target analytes from the solid sample. However, this non-selective process invariably leads to the co-extraction of unwanted materials, which are often present in far greater quantities than the explosive residues themselves [18] [19]. This necessitates subsequent cleanup steps to remove interfering compounds that can obscure detection and damage analytical instrumentation.
Solid-Phase Extraction (SPE)
Solid-Phase Extraction (SPE) is a pivotal cleanup and concentration technique used following initial solvent extraction. It functions by passing the solvent extract through a cartridge or column containing a sorbent material, which selectively retains the target analytes while allowing impurities to pass through. The analytes are then eluted with a stronger solvent, resulting in a purified and concentrated sample.
A comparative study of solvent extract cleanup procedures demonstrated that the Oasis HLB cartridge significantly outperformed other materials, including adsorbent resins like Chromosorb-104 and traditional silica, with analyte recoveries approaching 95% [18]. The study highlighted that adsorbent resins offered a higher degree of matrix rejection, leading to reduced background noise in chromatographic analysis. The optimization of SPE parameters—including solution pH, solvent type and concentration, and elution volume—is critical for achieving high recovery rates, as demonstrated in methods developed for pharmaceutical contaminants which achieved recoveries of 70% to 94.61% for target analytes using HLB cartridges [20].
Table 1: Comparison of Solid-Phase Extraction (SPE) Cleanup Procedures for Organic Explosives
| Cleanup Procedure | Reported Analyte Recovery | Key Advantages | Key Limitations |
|---|---|---|---|
| Oasis HLB Cartridge | Approaching 95% [18] | High recovery efficiency; suitable for broad range of analytes | Requires optimization of parameters (pH, solvent) [20] |
| Adsorbent Resins (e.g., Chromosorb-104) | Not specified, but showed high matrix rejection [18] | High degree of matrix rejection; reduces background noise | Lower overall recoveries compared to HLB [18] |
| Amberlite XAD-7 Procedure | Lowest overall recoveries [18] | Information not available in search results | Low analyte recovery |
| SPE with C-X Columns | Used for NMR-grade purity [19] | Provides extremely pure extracts for sensitive techniques like NMR | Potentially more complex protocol |
Syringe Filtration
Syringe filtration is a final preparatory step used to remove fine particulate matter from a liquid sample immediately before instrumental analysis. This process is crucial for protecting expensive chromatographic columns and instrumentation from clogging and damage. Syringe filters are characterized by their membrane material and pore size, which must be selected based on chemical compatibility with the sample and the required level of particulate removal [21] [22].
Filters with a 0.22 µm or 0.45 µm pore size are standard for ensuring the removal of fine particles and microorganisms. The choice of membrane material is critical: PTFE is highly resistant to aggressive chemicals and solvents, Nylon offers broad chemical compatibility for general applications, and PVDF or PES are preferred for biological samples due to their low protein binding characteristics [22]. An alternative to standalone syringe filters is the use of filter vials, which integrate the filtration membrane and the analysis vial into a single unit, streamlining the workflow and reducing the risk of contamination for high-throughput laboratories [23].
Table 2: Syringe Filter Membrane Selection Guide
| Membrane Material | Chemical Resistance | Protein Binding | Ideal Application |
|---|---|---|---|
| Polytetrafluoroethylene (PTFE) | Excellent (acids, bases, solvents) [22] | Low [22] | Aggressive organic solvents, harsh chemicals [22] [23] |
| Nylon | Good (except strong acids/bases) [22] | Moderate [22] | Aqueous-organic mixtures; general laboratory use [22] [23] |
| Polyvinylidene Fluoride (PVDF) | Excellent [22] | Low [22] | Biological samples, pharmaceuticals [22] |
| Polyethersulfone (PES) | Excellent [22] | Low [22] | Sterile filtration, biological samples [22] [23] |
| Polypropylene (PP) | Good (for aqueous solutions) [22] | Low [22] | Aqueous solutions, weak acids/bases [22] |
| Regenerated Cellulose (RC) | Good (for many solvents) [22] | Low [22] | Biological samples, HPLC [22] |
Workflow Integration and Experimental Protocols
The individual techniques must be integrated into a logical sequence to form a complete analytical workflow. The following diagram and associated protocols outline this process for the analysis of a post-blast soil sample.
Workflow for Post-Blast Residue Analysis
Detailed Experimental Protocols
Protocol 1: Extraction of Explosives from Post-Blast Soil via Solvent Extraction and SPE
This protocol is adapted from methods used for the analysis of organic explosives like RDX, TNT, and PETN from post-blast soil samples [19] [1].
- Sample Preparation: Collect approximately 100 g of soil from the post-blast scene. Dry the sample at room temperature and homogenize it using a mortar and pestle.
- Solvent Extraction: Transfer a sub-sample of the soil to a conical flask. Add a sufficient volume of an organic solvent, typically acetone, to fully immerse the soil. Sonicate or shake the mixture for a defined period (e.g., 30 minutes) to facilitate the dissolution of explosive residues.
- Initial Filtration: Decant or filter the solvent extract through filter paper (e.g., Whatman No. 1) to remove gross soil particles. Collect the filtrate in a clean flask.
- Solid-Phase Extraction Cleanup: a. Conditioning: Pre-condition an Oasis HLB SPE cartridge (60 mg/3 mL) by passing 3 mL of acetone followed by 3 mL of acetonitrile, 3 mL of methanol, and finally 3 mL of reverse osmosis water [19]. b. Loading: Load the filtered solvent extract onto the conditioned SPE cartridge at a controlled flow rate (e.g., 1-2 mL/min). c. Washing: Wash the cartridge with 5 mL of a 10% methanol/water solution to remove weakly retained interferents. d. Elution: Elute the retained explosive analytes using 4-6 mL of 100% methanol into a clean collection tube [20].
- Concentration: Evaporate the eluate to dryness under a gentle stream of nitrogen gas at 50°C.
- Reconstitution: Reconstitute the dried residue in 1 mL of methanol suitable for instrumental analysis.
Protocol 2: Verification of Syringe Filtration Effectiveness
Ensuring the syringe filter does not adsorb analytes is critical for quantitative accuracy. The following method can be used for verification [21].
- Particle Counting Method:
a. Measure the particle count in a defined volume of the sample using a particle counter.
b. Filter an identical volume of the sample through the syringe filter (e.g., Nylon, 0.22 µm).
c. Measure the particle count in the filtered liquid.
d. Calculate filtration efficiency:
Filtration Efficiency = (Particle Count Before - Particle Count After) / Particle Count Before × 100%. - Chromatography Method: a. Analyze a standard solution of the target analyte using High-Performance Liquid Chromatography (HPLC). b. Filter an identical standard solution through the syringe filter. c. Analyze the filtered standard using the same HPLC method. d. Compare the chromatographic peak areas of the pre- and post-filtration samples. A significant reduction in peak area indicates analyte adsorption by the filter membrane, necessitating a change of membrane material.
The Scientist's Toolkit: Essential Research Reagents and Materials
A successful forensic analysis relies on the selection of appropriate materials and reagents. The following table details key components used in the extraction and concentration workflow for post-blast residues.
Table 3: Essential Research Reagents and Materials for Post-Blast Residue Analysis
| Item | Function/Application | Exemplary Specifications |
|---|---|---|
| Oasis HLB SPE Cartridge | Cleanup and concentration of a wide range of organic explosives from liquid extracts. | 60 mg/3 mL bed volume; Hydrophilic-Lipophilic Balanced copolymer [18] [20]. |
| Acetone (HPLC Grade) | Primary solvent for the extraction of organic explosive residues from solid matrices like soil or swabs. | High purity to minimize background interference during analysis [19] [1]. |
| Methanol (HPLC Grade) | Elution solvent for SPE; reconstitution solvent for dried extracts. | 100% concentration for efficient elution of analytes from HLB cartridges [20]. |
| Syringe Filters | Final particulate removal from samples prior to instrumental analysis to protect columns and detectors. | Membrane: PTFE for solvents, Nylon for general use, PES/PVDF for biological mixes. Pore Size: 0.22 µm for sterile/fine filtration, 0.45 µm for standard HPLC [22] [23]. |
| Nitrogen Evaporator | Gentle concentration of eluates following SPE by evaporating the solvent under a stream of inert nitrogen gas. | Equipped with a heating block (temp. range up to 50-60°C) and adjustable gas flow [20]. |
| Ion Chromatography (IC) System | Detection and quantification of inorganic explosive residues (e.g., nitrate, chlorate ions) in post-blast samples. | Metrosep A Supp column; Na₂CO₃/NaHCO₃ eluent [17]. |
The forensic investigation of explosive events demands a meticulous and multi-staged approach to sample preparation. While initial solvent extraction is indispensable for liberating residues from complex matrices, it is the subsequent application of SPE that provides the necessary purification and concentration to achieve detectable analyte levels. The final step of syringe filtration safeguards the integrity of analytical instrumentation. The quantitative data clearly establishes that SPE, particularly with Oasis HLB cartridges, is the most effective standalone cleanup technique, with recoveries up to 95%. The integration of these techniques into a coherent workflow, as detailed in this guide, ensures that trace levels of explosive residues can be reliably recovered, concentrated, and prepared for definitive identification, thereby providing robust scientific evidence crucial for legal and security outcomes.
In the advanced landscape of forensic chemistry, the analysis of post-blast residues represents a significant challenge, requiring robust protocols to isolate and identify trace explosive materials within complex, contaminated matrices. While modern instrumental techniques provide definitive confirmation, the initial screening of samples relies heavily on classical methods. This whitepaper details the enduring role of color tests and Thin-Layer Chromatography (TLC) within integrated analytical workflows for post-blast residue analysis. We outline standardized experimental protocols, provide quantitative performance data, and demonstrate how these cost-effective, rapid techniques serve as indispensable prescreening tools that guide subsequent confirmatory analysis, thereby enhancing the overall efficiency and reliability of forensic investigations.
The forensic investigation of an explosion site necessitates the chemical characterization of trace explosive residues deposited on fragmented and heterogeneous substrates such as soil, metal, and plastic [1] [24]. These residues are often present in nanogram to microgram quantities, mixed with debris, and have undergone partial decomposition, making their analysis inherently complex [1]. A systematic approach to sampling, extraction, and concentration is fundamental to any successful analysis, as improper collection can lead to unreliable results even with the most advanced instrumentation [1].
Within this context, a holistic analytical strategy employs a combination of techniques. Classical methods, particularly color tests and TLC, provide rapid, presumptive information that is crucial for triaging samples and guiding the application of more resource-intensive confirmatory methods like Gas Chromatography-Mass Spectrometry (GC-MS) and Ion Trap Mobility Spectrometry (ITMS) [1] [25]. This integrated approach, which combines classical and advanced techniques, ensures comprehensive coverage of potential analytes and mitigates challenges posed by complex sample matrices [1].
Experimental Protocols for Post-Blast Residue Analysis
Sample Collection and Extraction
The efficacy of any analytical technique is contingent upon proper sample preparation. For post-blast soil and debris, a modified extraction and clean-up protocol is critical.
- Evidence Collection: Oversized exhibits from blast scenes require sequential swabbing with solvents of varying polarity (e.g., ether, acetone, water, sodium hydroxide, pyridine) to ensure representative recovery of both organic and inorganic residues from irregular surfaces [24].
- Extraction and Clean-up: Soil or swab extracts undergo filtration and concentration. Solid-Phase Extraction (SPE) is employed to remove interferences, and syringe filtration (0.22 µm) has been shown to maximize recovery yield by minimizing background interference [1] [24]. For soil samples, warm extraction techniques like Soxhlet Warm Extraction (SWE) can be applied, though recovery rates vary; for example, recovery for nitrotoluenes is approximately 60% with SWE [1].
Color Tests: Presumptive Spot Analysis
Color tests, or spot tests, are foundational presumptive assays used for the initial identification of explosive compound classes. They are performed by applying a chemical reagent to a small sample and observing the resulting color change.
- Key Reagents and Procedures: Common reagents and their target explosive groups include [1]:
- Diphenylamine (1 mg in 10 mL concentrated H₂SO₄): Used for nitrate esters and nitro compounds.
- Brucine sulfate (0.5 g in 10 mL concentrated H₂SO₄): Often used for nitrates.
- Thymol (1 crystal in 10 mL concentrated H₂SO₄): A reagent for nitrocellulose.
- Alcoholic Potassium Hydroxide (1 g KOH in 10 mL absolute alcohol): Used for TNT and other nitroaromatics.
- Griess Reagent: Specifically detects nitrite compounds, which are common decomposition products of nitrate esters and nitramines.
- Methodology: A small aliquot of the extracted residue is spotted on a white tile or filter paper. A drop of the reagent is added, and the immediate color formation is noted and compared to a reference standard [1].
Thin-Layer Chromatography (TLC): Separation and Preliminary Identification
TLC provides a higher degree of separation and specificity than color tests alone, allowing for the resolution of complex mixtures from post-blast extracts.
- TLC Plate Preparation: Pre-coated silica gel 60 F254 plates (200 µm thickness) are used. Plates are often activated in an air oven at 110°C for 30 minutes prior to use [24].
- Spotting and Development: The concentrated extract is spotted onto the baseline of the TLC plate. The plate is developed in a saturated chamber containing an appropriate mobile phase [1] [24].
- Mobile Phases: The choice of mobile phase is critical for achieving optimal separation. Research has proposed novel phases for explosives, including:
- Visualization Techniques: After development and drying, the plate is visualized using a multi-step process:
- Spraying with Sodium Hydroxide: The plate is lightly sprayed with a 10% NaOH solution and heated at 100°C for 10 minutes [1].
- Application of Griess Reagent: Following NaOH, Griess reagent is sprayed to enhance the visualization of spots [1].
- Alternative Developers: A 5% ethanolic diphenylamine solution can also be used, followed by observation under UV light (254 nm) and subsequent spraying with concentrated sulfuric acid to record the colors produced [24].
Diagram 1: Integrated Analytical Workflow for Post-Blast Residues, illustrating the role of classical techniques within a broader strategy.
Technical Data and Research Findings
Performance of Color Tests and TLC
The following tables summarize key experimental data from recent studies on the application of these classical techniques.
Table 1: Minimum Detection Limits (MDL) for Common Explosives via Integrated Analytical Methods [25]
| Explosive Compound | MDL (ng) | Analytical Technique for MDL Determination |
|---|---|---|
| TNT | 2 ng | Integrated Protocol (TLC, GC-MS, ITMS) |
| RDX | 3 ng | Integrated Protocol (TLC, GC-MS, ITMS) |
| NG (Nitroglycerin) | 3 ng | Integrated Protocol (TLC, GC-MS, ITMS) |
| PETN | 4 ng | Integrated Protocol (TLC, GC-MS, ITMS) |
Table 2: TLC Rf Values for Explosive Standards and Post-Blast Residues [1]
| Explosive Compound | Rf Value (Trichloroethylene-Acetone, 4:1) | Visualization Method |
|---|---|---|
| TNT (Standard) | 0.69 | NaOH / Griess Reagent |
| TNT (Post-Blast) | 0.69 (matched) | NaOH / Griess Reagent |
| PETN (Standard) | 0.74 | NaOH / Griess Reagent |
| PETN (Post-Blast) | 0.74 (matched) | NaOH / Griess Reagent |
| Tetryl (Standard) | 0.56 | NaOH / Griess Reagent |
| Tetryl (Post-Blast) | 0.56 (matched) | NaOH / Griess Reagent |
| Nitrocellulose (NC) | 0.00 (remains at start) | NaOH / Griess Reagent |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Explosive Residue Analysis [1] [24]
| Reagent/Solution | Composition | Function in Analysis |
|---|---|---|
| Diphenylamine Reagent | 1 mg diphenylamine in 10 mL conc. H₂SO₄ [1] | Color test for nitrate esters and nitro compounds. |
| Brucine Sulfate Reagent | 0.5 g brucine sulfate in 10 mL conc. H₂SO₄ [1] | Color test for nitrate ions. |
| Griess Reagent | Commercially available or prepared formulation [1] | Detection of nitrite compounds, crucial for visualizing nitrate ester and nitramine decomposition on TLC plates. |
| Alcoholic KOH | 1 g KOH pellets in 10 mL absolute alcohol [1] | Color test for TNT and other nitroaromatics. |
| TLC Mobile Phase A | Trichloroethylene-Acetone (4:1 v/v) [1] | Solvent system for separating TNT, PETN, and Tetryl. |
| TLC Mobile Phase B | Chloroform-Acetone (1:1 v/v) [24] | General solvent system for separating organic explosives. |
| TLC Mobile Phase C | Toluene-Cyclohexane (7:3 v/v) [24] | General solvent system for separating organic explosives. |
| TLC Visualization Spray | 5% Diphenylamine in 95% Ethanol [24] | Developing agent for TLC plates, produces characteristic colors with different explosives. |
Diagram 2: Thin-Layer Chromatography (TLC) Workflow, detailing the key steps from spotting to visualization.
Discussion: Integration with Modern Analytical Techniques
The value of color tests and TLC is not as standalone identification tools, but as integral components of a hierarchical analytical strategy. Their presumptive nature necessitates confirmation by more specific techniques [25]. For instance, a study on oversized post-blast exhibits successfully used TLC and chemical spot tests to triage samples, which were subsequently confirmed by GC-MS for organic components (e.g., diesel-range hydrocarbons in ANFO) and FTIR spectroscopy for inorganic ions (e.g., nitrate) [24].
Furthermore, advancements in instrumental methods have addressed limitations of classical techniques. For example, the analysis of PETN is notoriously challenging due to its thermal lability in standard GC-EI-MS systems. Recent research has developed new GC-EI-MS methods that overcome decomposition issues, allowing for reliable confirmatory analysis following TLC prescreening [1] [25]. Similarly, Ion Trap Mobility Spectrometry (ITMS) offers rapid, sensitive analysis for field detection and persistence studies, complementing the initial information gained from classical screens [1] [25].
In the demanding field of post-blast forensic analysis, classical screening techniques have demonstrated remarkable endurance. Color tests and TLC provide rapid, cost-effective, and reliable presumptive analysis that is essential for efficient sample triage and guiding analytical workflows. Their continued relevance is secured by their integration with advanced confirmatory techniques like GC-MS and ITMS, creating a robust, multi-tiered framework for the definitive identification of explosive residues. As the complexity of explosive formulations and substrates evolves, the optimized protocols for these classical methods, as detailed in this guide, will remain a cornerstone of effective forensic science practice.
The forensic investigation of explosive events relies heavily on the precise chemical analysis of post-blast residues to identify the materials involved and reconstruct the incident. These residues present a significant analytical challenge due to their complex matrix interference, trace concentration levels, and heterogeneous distribution across blast scenes [26] [1]. This technical guide provides an in-depth examination of three cornerstone instrumental techniques—Gas Chromatography-Mass Spectrometry (GC-MS), Ion Chromatography (IC), and Fourier Transform Infrared (FTIR) Spectroscopy—within the context of post-blast residue analysis. The integration of these methods enables comprehensive characterization of both organic and inorganic explosive components, forming a complete analytical workflow from sample collection to data interpretation [1] [16] [9]. This whitepaper details standardized protocols, performance benchmarks, and advanced chemometric approaches that collectively enhance detection sensitivity, selectivity, and forensic reliability for researchers and forensic professionals.
Core Principles and Forensic Applications
Fourier Transform Infrared (FTIR) Spectroscopy operates on the principle of measuring molecular vibrational excitations induced by infrared radiation, generating unique spectral fingerprints specific to individual explosive compounds [26]. FTIR is particularly valuable for its non-destructive analysis capability and minimal sample preparation requirements. In forensic applications, FTIR successfully identifies functional groups and molecular structures in both pure explosives and complex post-blast residues, including C-4, PETN, and TNT, based on their characteristic absorption patterns [26] [27]. Advanced synchrotron-radiation-based FTIR demonstrates particular effectiveness for trace analysis due to significantly enhanced sensitivity [26].
Gas Chromatography-Mass Spectrometry (GC-MS) combines the superior separation power of gas chromatography with the precise identification capability of mass spectrometry. This technique is indispensable for analyzing volatile and semi-volatile organic explosives in complex mixtures [1] [9]. GC-MS separates individual components from sample matrices before mass spectrometry provides structural identification through characteristic fragmentation patterns. The technique effectively identifies nitramine explosives (e.g., RDX), nitroaromatics (e.g., TNT), and nitrate esters (e.g., PETN), though thermally labile compounds may require specialized derivatization or lower inlet temperatures to prevent decomposition [1] [28].
Ion Chromatography (IC) specializes in separating and detecting ionic species, making it ideally suited for analyzing inorganic explosive residues. IC plays a critical role in identifying anions like nitrate (NO₃⁻) and perchlorate (ClO₄⁻) from inorganic explosives and pyrotechnics [29]. In post-blast investigations, IC provides quantitative data on ionic composition that helps identify explosive formulations and distinguish them from environmental background contamination through comparative analysis with control samples [29].
Performance Characteristics Comparison
Table 1: Performance Metrics of Key Analytical Techniques for Explosive Residue Analysis
| Analytical Technique | Target Analytes | Typical Detection Limits | Key Strengths | Primary Limitations |
|---|---|---|---|---|
| FTIR Spectroscopy | Organic explosives (C-4, PETN, TNT, RDX) | Low ppm range | Non-destructive; minimal sample preparation; molecular fingerprinting | Interference from environmental contaminants; requires reference spectra |
| GC-MS | Organic explosives (TNT, RDX, PETN, NG) | Low ppm to ppb range | High sensitivity; selective separation; library matching | Thermal degradation of labile compounds; requires sample derivatization for some analytes |
| Ion Chromatography (IC) | Inorganic ions (NO₃⁻, ClO₄⁻, NH₄⁺) | ppb range | Excellent for inorganic explosives; quantitative analysis; minimal interference | Limited to ionic species; requires sample dissolution |
Experimental Protocols
Standardized Workflow for Post-Blast Residue Analysis
The following workflow diagram illustrates the integrated analytical approach for comprehensive post-blast residue characterization:
Figure 1: Integrated Analytical Workflow for Post-Blast Residue Analysis
FTIR Spectroscopy Protocol
Sample Preparation for Solid Residues: Gently remove visible particles from debris surfaces and mix with potassium bromide (KBr) in an approximate 1:100 weight ratio. Homogenize the mixture using an agate mortar and pestle. Pelletize the homogeneous powder using a hydraulic press with a clamping force of 80 kN to form 13 mm diameter transparent pellets [27].
Instrumental Parameters:
- Spectral Range: 4000–400 cm⁻¹
- Resolution: 4 cm⁻¹
- Scanning Speed: 67 scans per minute (approximately 1 minute acquisition time)
- Detector: Mercury Cadmium Telluride (MCT) cooled to 77 K
- Beam Source: Synchrotron radiation for enhanced sensitivity [26]
- Sample Chamber: Evacuated to 3 mbar to eliminate atmospheric water vapor interference [27]
Analysis Procedure: Place the KBr pellet in the sample holder and insert it into the evacuated sample chamber. Collect background spectrum with an empty evacuated chamber. Acquire sample spectra and process using appropriate baseline correction and normalization algorithms. Compare resulting spectra against reference databases of known explosive materials [26] [27].
GC-MS Protocol for Organic Explosives
Sample Extraction: Swab suspected surfaces with cotton swabs moistened with acetone or rinse debris with acetone solvent. Transfer the solvent to a clean vial and concentrate using gentle nitrogen evaporation. For complex matrices, employ solid-phase extraction (SPE) clean-up to remove interfering compounds [1] [16].
GC-MS Parameters:
- Column: Medium-polarity fused silica capillary column (e.g., DB-35ms, 30m × 0.25mm × 0.25μm)
- Inlet Temperature: 175°C (lowered for thermally labile compounds like PETN)
- Oven Program: 40°C (hold 2 min), ramp to 300°C at 15°C/min, final hold 5 min
- Carrier Gas: Helium, constant flow 1.2 mL/min
- Transfer Line Temperature: 280°C
- Ionization Mode: Electron Impact (EI) at 70 eV
- Mass Range: 35-550 Da [1] [28]
Data Analysis: Identify compounds by comparing retention times and mass spectra with certified reference standards. For challenging analytes like PETN, employ chemical ionization or derivatization techniques to enhance detection. Use internal standards for quantitative analysis [1].
Ion Chromatography Protocol for Inorganic Residues
Sample Preparation: Collect control soil samples from at least 5 meters away from the blast epicenter for comparative analysis. Homogenize pit soil samples from blast epicenter and extract soluble ions using deionized water with mechanical agitation for 30 minutes. Filter extracts through 0.45μm membrane filters to remove particulate matter [29].
IC Parameters:
- Column: Anion exchange column (e.g., AS14A or equivalent)
- Eluent: Carbonate/bicarbonate buffer (3.5mM Na₂CO₃/1.0mM NaHCO₃)
- Flow Rate: 1.0 mL/min
- Injection Volume: 25μL
- Detection: Suppressed conductivity detection
- Run Time: 15-20 minutes [29]
Quantitative Analysis: Prepare calibration curves using certified anion standards. Compare nitrate and other relevant anion concentrations in blast samples against control samples to identify significant elevations indicative of explosive residues [29].
Advanced Applications and Data Analysis
Chemometric Integration with FTIR Spectroscopy
Advanced statistical analysis significantly enhances the interpretation of FTIR spectral data from post-blast residues. Machine learning techniques applied to spectral datasets enable precise classification of explosive materials even after high-order detonations that consume most of the original charge [27].
Data Pre-treatment: Before analysis, implement spectral preprocessing including baseline correction, normalization, and derivative spectroscopy to reduce scattering effects and enhance spectral features [27].
Multivariate Classification: Employ Principal Component Analysis (PCA) to reduce spectral dimensionality and identify key discriminating variables. Apply Linear Discriminant Analysis (LDA) to maximize separation between explosive classes. The hybrid LDA-PCA approach demonstrates particularly effective classification of high explosives like C-4, TNT, and PETN in post-blast residues [27].
Table 2: Research Reagent Solutions for Explosive Residue Analysis
| Reagent/Material | Application | Function | Technical Specifications |
|---|---|---|---|
| Potassium Bromide (KBr) | FTIR sample preparation | Matrix for pellet formation | FTIR-grade, 1:100 sample-to-KBr ratio |
| Acetone | Solvent extraction | Organic residue extraction | HPLC-grade, low background contamination |
| Diphenylamine Reagent | Colorimetric screening | Nitrate ester detection | 1mg in 10mL concentrated H₂SO₄ |
| Griess Reagent | TLC visualization | Nitroaromatic detection | Spray reagent after separation |
| Carbonate/Bicarbonate Buffer | Ion Chromatography | Mobile phase eluent | 3.5mM Na₂CO₃/1.0mM NaHCO₃ |
| Deionized Water | Aqueous extraction | Inorganic ion extraction | 18.2 MΩ·cm resistivity |
Enhanced GC-MS Detection Strategies
The analysis of thermally labile explosives requires specialized approaches to prevent decomposition during analysis. For PETN detection, implement reduced inlet temperatures (175°C) and consider alternative ionization techniques [1]. Gas Chromatography with Vacuum Ultraviolet detection (GC-VUV) provides complementary data through characteristic absorption spectra in the 125-240 nm range, offering additional confirmation of compound identity [28].
The following diagram illustrates the decision process for analyzing challenging explosive compounds:
Figure 2: GC-MS Method Selection for Challenging Explosive Compounds
Isotopic and Impurity Profiling
Advanced applications extend beyond simple identification to source attribution through isotopic signature analysis and impurity profiling. Isotopic ratios (e.g., ¹⁵N/¹⁴N, ¹⁸O/¹⁶O) preserved in post-blast residues can potentially link explosive materials to their manufacturing sources [28]. Similarly, trace impurity patterns provide characteristic signatures that differentiate between production batches and manufacturers [9] [6].
The integrated application of GC-MS, IC, and FTIR spectroscopy provides a comprehensive analytical framework for post-blast residue investigation. GC-MS delivers superior sensitivity for organic explosive compounds, IC specifically targets inorganic ionic species, and FTIR offers rapid, non-destructive molecular fingerprinting. The synergy between these techniques enables complete characterization of complex post-blast residues. Future advancements will focus on enhancing portability for field deployment, improving sensitivity for trace-level detection, and refining chemometric integration for automated data interpretation. The continued development of these analytical methodologies remains essential for advancing forensic capabilities in explosives investigation and strengthening security responses to explosive threats.
Post-blast forensic investigations present significant analytical challenges due to the complex nature of evidence. Oversized exhibits, such as vehicle fragments, concrete blocks, and metallic debris, are often contaminated with mixed explosive formulations and exhibit heterogeneous residue distribution across their surfaces [24] [16]. These challenging substrates require specialized approaches beyond conventional methods optimized for small, homogeneous samples. The inherent complexity is further compounded by environmental contamination and non-uniform residue deposition, which can obscure analytical signatures and reduce detection sensitivity [24]. This technical guide outlines integrated analytical workflows specifically designed to address these challenges through systematic residue recovery, concentration, and complementary analysis techniques, providing a robust framework for forensic reconstruction of detonation events within broader research on post-blast residue sampling and concentration techniques.
Integrated Workflow Design and Principles
The fundamental principle underlying effective post-blast analysis involves integrating multiple sampling and analytical techniques to overcome the limitations of single-method approaches. Traditional single-technique methods, such as direct solvent rinsing or simple cotton swabbing, often provide incomplete recovery from irregular or large fragments, resulting in reduced analytical sensitivity and potential false negatives [24]. The integrated workflow emphasizes sequential recovery of both organic and inorganic explosive residues through systematic solvent extraction, followed by orthogonal analytical confirmation using complementary instrumental techniques [24] [25].
This approach is particularly crucial for investigating mixed explosive detonations, where overlapping signals or partial combustion can obscure analytical signatures. For example, ammonium nitrate fuel oil (ANFO) residues typically consist of both nitrate salts and hydrocarbon components, which exhibit different binding affinities toward metallic, soil, and polymeric substrates [24]. The integrated workflow described in this guide addresses this heterogeneity through tailored extraction and analysis protocols that maximize residue recovery and strengthen evidentiary interpretation for both researchers and forensic practitioners.
Experimental Protocols: Methodologies for Complex Evidence
Evidence Collection and Spatial Subsampling
The initial evidence collection phase requires strategic approaches to preserve residue integrity and distribution patterns:
Collection of Heterogeneous Exhibits: Metallic spades, large and small metal fragments, nails, plastic containers, wires, and structural components should be collected alongside soil samples from both the blast epicenter and control areas [24]. These materials represent the complex and composite matrices typically encountered in post-blast forensic investigations.
Spatial Subsampling: For oversized fragments, dividing the exhibit into logical sections based on proximity to the suspected blast origin, surface characteristics, or visible residue patterns enhances detection sensitivity by accounting for non-uniform residue deposition [24]. This approach recognizes that residue distribution is rarely homogeneous across large, fragmented surfaces.
Environmental Controls: Soil samples from control areas away from the blast epicenter are essential for distinguishing explosive residues from environmental background contamination [24].
Sequential Swabbing and Extraction Protocol
A multi-stage sequential swabbing protocol applied to all exhibits ensures comprehensive recovery of a wide range of explosive residues while minimizing sample loss:
Table 1: Sequential Swabbing Protocol for Explosive Residue Recovery
| Step | Solvent | Target Residues | Subsequent Analysis |
|---|---|---|---|
| 1 | Diethyl ether | Non-polar organic compounds (hydrocarbons, fuel oils) | GC-MS for ANFO identification [24] |
| 2 | Acetone | Polar organic explosives (TNT, RDX, PETN) | TLC and GC-MS for high explosives [24] [25] |
| 3 | Demineralized water | Water-soluble inorganic ions (nitrate, chloride, sulfate) | Chemical spot tests and FTIR [24] |
| 4 | Sodium hydroxide solution | Basic inorganic compounds | FTIR and chemical tests [24] |
| 5 | Pyridine | Elemental sulfur and related residues | Specific identification tests [24] |
Procedure:
- Gently clean oversized and irregular fragments of loose debris prior to extraction [24].
- Saturate absorbent cotton with the specified solvent in the sequence listed in Table 1.
- Swab the entire surface area of each exhibit systematically with the solvent-saturated cotton.
- Filter all extracts through 0.22 μm nylon syringe filters to remove particulate matter [24].
- Concentrate extracts to approximately 2-5 mL by evaporation at room temperature [24].
- Store extracts appropriately for subsequent analysis.
Filtration and Cleanup Procedures
Syringe filtration represents a critical step in the workflow, with studies demonstrating that it produces the highest recovery yield by minimizing background interference and enhancing analytical signal clarity [24] [25]. The use of 0.22 μm nylon syringe filters effectively removes particulate matter that could interfere with subsequent instrumental analysis while retaining the target explosive residues in the filtrate [24]. For highly contaminated samples, additional cleanup steps such as solid-phase extraction (SPE) may be incorporated, with stacked cartridges combining graphitized carbon with weak anion exchange proving effective for isolating target analytes like PFAS while minimizing background interference in complex matrices [30].
Analytical Techniques for Residue Characterization
Organic Residue Analysis
Gas Chromatography-Mass Spectrometry (GC-MS) provides high sensitivity for organic explosive components, particularly petroleum hydrocarbons in ANFO formulations [24]. The analysis of ether extracts confirms the presence of high-boiling petroleum hydrocarbons consistent with diesel fractions, with characteristic compounds such as hexadecane identified through library matching [24]. For challenging compounds like PETN, which often undergo thermal degradation in standard GC systems, optimized GC/EI-MS methods have been developed to enable reliable detection [25].
Thin Layer Chromatography (TLC) serves as a complementary technique for preliminary screening of organic high explosives. The protocol involves:
- Using pre-coated Silica gel 60G F254 Plates (200 μm thickness, 20 × 20 cm) activated at 110°C for 30 minutes [24].
- Preparing solvent systems such as chloroform:acetone (1:1) and toluene:cyclohexane (7:3) in saturated developing chambers [24].
- Spotting concentrated acetone extracts alongside reference standards.
- Developing the plate until the solvent front rises 10 cm from the origin.
- Detecting compounds by spraying with 5% diphenylamine in 95% ethanol, observing under UV light (254 nm), and subsequently spraying with concentrated sulfuric acid [24].
- Comparing resulting colors and Rf values with reference standards for identification [24].
Recent advances in TLC include novel mobile phases that achieve significant resolution improvements for a wide range of explosives across different groups, confirming their suitability for forensic applications [25].
Inorganic Residue Analysis
Fourier Transform Infrared (FTIR) Spectroscopy enables identification of inorganic ions through characteristic absorption patterns. Using an FTIR spectrometer with an attenuated total reflectance (ATR) accessory, samples are scanned between 4000 cm⁻¹ to 400 cm⁻¹ at a resolution of 4 cm⁻¹, with 64 scans performed to obtain characteristic spectra [24]. Spectral patterns are then compared against reference libraries to identify inorganic components such as nitrate salts [24].
Chemical spot tests provide preliminary screening for specific inorganic ions present in water and sodium hydroxide extracts. These presumptive tests, while not confirmatory, guide subsequent analytical efforts and provide rapid preliminary information about sample composition [24] [25].
Emerging and Complementary Techniques
Direct Analysis in Real Time Mass Spectrometry (DART-MS) has emerged as a powerful technique for rapid screening of explosive residues, enabling analysis with minimal sample preparation. DART-MS can directly analyze swab samples or even fragments of post-blast debris by simultaneously thermally desorbing and ionizing substances for mass spectrometric analysis [12]. This approach eliminates or reduces the need for extensive sample processing such as extraction and filtration that is typically required for chromatographic analyses [12].
Ion Trap Mobility Spectrometry (ITMS) offers a rapid approach for field detection and persistence studies, with research demonstrating that nitroglycerin (NG) exhibits higher persistence on hand swabs than TNT [25]. Studies have established minimum detection limits (MDL) of 2 ng for TNT, 3 ng for RDX and NG, and 4 ng for PETN using this technique [25].
Scanning Electron Microscopy with Energy Dispersive X-ray Spectroscopy (SEM-EDS) provides morphological and elemental characterization of inorganic residues, identifying characteristic particles such as the Pb-Ba-Sb-containing particles observed in lead styphnate-based primers [31].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Research Reagent Solutions for Post-Blast Residue Analysis
| Reagent/Material | Function/Application | Specifications |
|---|---|---|
| Diethyl ether | Extraction of non-polar organic compounds (fuel oils, hydrocarbons) | Analytical grade [24] |
| Acetone | Extraction of polar organic explosives (TNT, RDX, PETN) | Analytical grade [24] |
| Demineralized water | Extraction of water-soluble inorganic ions (nitrate, chloride) | Lab-grade purity [24] |
| Nylon syringe filters | Removal of particulate matter from extracts | 0.22 μm pore size [24] |
| Silica gel TLC plates | Separation of organic explosive compounds | 60G F254, 200 μm thickness [24] |
| Diphenylamine reagent | Visualization of nitroaromatic explosives on TLC | 5% solution in 95% ethanol [24] |
Workflow Integration and Data Interpretation
The integration of multiple analytical techniques provides orthogonal data that strengthens forensic conclusions. The workflow diagram below illustrates the comprehensive approach to post-blast residue analysis:
Integrated Workflow for Post-Blast Residue Analysis
Data interpretation requires correlating results across multiple analytical techniques to build a comprehensive picture of the explosive composition. For instance, the detection of petroleum hydrocarbons through GC-MS combined with the identification of nitrate ions through FTIR and chemical tests provides compelling evidence for ANFO explosives [24]. The analytical decision pathway guides scientists through this interpretative process:
Analytical Decision Pathway for Explosive Identification
Integrated workflows combining sequential swabbing, optimized extraction, filtration, and complementary analytical methods represent a significant advancement in forensic explosive residue analysis. This approach substantially improves residue recovery from complex and fragmented post-blast materials, enhances analytical reliability through orthogonal verification, and strengthens evidentiary interpretation [24]. The systematic integration of multiple techniques addresses the fundamental challenges of heterogeneous residue distribution and matrix interference that routinely complicate post-blast investigations. For researchers and forensic professionals, adopting this comprehensive framework provides a robust methodology for investigating complex detonation events, with applications ranging from criminal forensics to security screening and counterterrorism efforts. As analytical technologies continue to evolve, particularly in the areas of ambient ionization mass spectrometry and automated sample preparation, further refinements to these integrated workflows will continue to enhance their sensitivity, efficiency, and forensic value.
Solving Real-World Problems: Maximizing Recovery and Overcoming Analytical Hurdles
The analysis of post-blast residues presents one of the most analytically challenging scenarios in forensic chemistry, characterized by extremely low yields of target analytes distributed across complex, contaminated matrices. Following an explosion, the residual chemical signature of the original explosive material often exists at parts-per-billion concentrations or lower, creating significant detection and identification challenges for forensic investigators [13]. In this context, extraction optimization becomes paramount for successful forensic attribution. The efficiency of the extraction protocol directly determines the success of subsequent analytical techniques, including gas chromatography-vacuum UV spectroscopy (GC-VUV) and direct-analysis-in-real-time mass spectrometry (DART-MS), by determining both the quantity and quality of the analyte introduced to the instrument [13] [12].
This technical guide examines optimized extraction methodologies, with a specific focus on adapting traditional techniques like Soxhlet extraction for time-sensitive forensic investigations and evaluating modern alternatives. The strategic balance between exhaustive extraction and the practical demands of rapid turnaround times in criminal investigations frames a critical challenge for researchers and scientists engaged in method development for forensic explosives analysis.
Fundamental Principles of Soxhlet Extraction
Core Mechanism and Historical Context
Soxhlet extraction, developed in 1879 by Franz Ritter von Soxhlet, is a classical solid-liquid extraction technique that enables continuous recovery of analytes from solid matrices using a recycled solvent system [32]. The method operates on the principle of solvent reflux and siphon action, wherein heated solvent vaporizes, condenses, and percolates through a solid sample contained in a porous thimble, dissolving target compounds with each cycle [32]. Unlike simple maceration, the Soxhlet apparatus ensures that only fresh, uncontaminated solvent contacts the sample during each cycle, preventing saturation and maximizing extraction efficiency over time [32].
The fundamental process involves a cyclic sequence: (1) solvent vaporization from the heating flask, (2) vapor ascension through the distillation path, (3) condensation in the reflux condenser, (4) solvent dripping onto the sample, (5) gradual filling of the extraction chamber, (6) automatic siphoning when liquid reaches the siphon arm level, and (7) return of the analyte-enriched solvent to the heating flask [32]. This cycle repeats automatically, with each iteration extracting additional target compounds while leaving insoluble impurities in the thimble.
Advantages and Limitations in Low-Yield Contexts
Soxhlet extraction provides several significant benefits for challenging extractions, particularly its high extraction efficiency due to continuous fresh solvent contact, automated operation once configured, and proven suitability for quantitative analysis required in regulatory compliance testing [32]. However, the technique presents notable limitations for post-blast residue analysis, primarily its extended extraction times (typically 12-24 hours), which are incompatible with rapid investigation timelines [32]. Additionally, the high solvent consumption (typically hundreds of milliliters) generates significant waste and potential safety concerns, while the extraction temperature being limited to the solvent's boiling point may be insufficient for strongly bound compounds or too harsh for potential degradation products [32]. Modern instrumental validation has also demonstrated that Soxhlet results can be quantitatively incomplete for certain analytes, a critical consideration for forensic applications requiring maximal recovery [32].
Optimization Strategies for Short-Duration and Warm Soxhlet Extraction
Operational Parameter Optimization
Optimizing Soxhlet extraction for reduced processing time requires careful consideration of multiple interdependent parameters. The choice of extraction solvent significantly influences both the efficiency and selectivity of the process, with solvent polarity being a primary consideration. Research on Mentha longifolia extraction demonstrates that hydro-ethanol solutions (70% v/v) prepared with maceration and Soxhlet processes showed the ultimate rate of phenolic compounds, suggesting that mixed-solvent systems may enhance extraction efficiency for certain analyte classes [33].
Table 1: Key Optimization Parameters for Short-Duration Soxhlet Extraction
| Parameter | Optimization Strategy | Impact on Extraction Efficiency |
|---|---|---|
| Solvent Selection | Use binary solvent systems (e.g., 70% ethanol); adjust polarity for target analytes | Increases mass transfer rate; improves selectivity for target compounds [33] |
| Temperature Control | Operate at 20°C above solvent boiling point; use high-boiling-point solvents cautiously | Enhances solubility and diffusion rates; reduces viscosity and surface tension [32] |
| Sample Preparation | Reduce particle size to 0.5-2mm; dry samples with anhydrous sodium sulfate | Maximizes surface area for solvent contact; prevents solvent dilution and channeling [32] |
| Cycle Frequency | Optimize heating power to maintain consistent cycling; ensure proper condenser efficiency | Increases number of extraction cycles per unit time; maintains steady-state concentration gradient [32] |
Temperature management represents another critical optimization parameter. While the extraction temperature in traditional Soxhlet is limited by the solvent's boiling point, strategic solvent selection can enable operation at higher temperatures without specialized equipment. However, this approach requires careful consideration of analyte stability, as elevated temperatures may degrade heat-labile compounds [32]. For post-blast residues containing potentially labile organic peroxide HMEs, such as TATP, which are known to degrade rapidly, temperature control becomes particularly crucial [12].
Workflow Integration for Forensic Scenarios
Implementing optimized Soxhlet extraction within a forensic workflow requires systematic approaches that align extraction parameters with analytical objectives and constraints. The following diagram illustrates a recommended optimization workflow for method development in post-blast residue analysis:
Diagram: Soxhlet Optimization Workflow for Forensic Methods
Sample preparation emerges as a foundational step in this optimization workflow. Proper sample grinding to achieve optimal particle size (0.5-2mm) and thorough drying with anhydrous sodium sulfate are essential prerequisites for efficient extraction, as they maximize surface area for solvent contact while preventing interference from moisture [32]. In post-blast scenarios, where samples may include swabs from bomb fragments or soil extracts, this preparation becomes particularly critical due to the complex matrices involved [12].
Alternative Extraction Methods for Low-Yield Scenarios
Modern Extraction Technologies
While Soxhlet extraction remains a benchmark technique, several modern alternatives offer specific advantages for low-yield forensic applications where time, efficiency, and analyte recovery are paramount. Ultrasound-assisted extraction (UAE) employs ultrasonic energy to create cavitation bubbles in the solvent, which implode and generate localized high temperatures and pressures that disrupt sample matrices and enhance mass transfer [33]. Research on bioactive compounds demonstrates that UAE can achieve important nutritional properties with significantly reduced extraction times compared to conventional methods [33]. For thermally sensitive explosive residues, such as organic peroxide HMEs known to degrade rapidly, UAE's ability to operate at lower temperatures presents a distinct advantage [12].
Microwave-assisted extraction (MAE) utilizes microwave energy to directly heat the solvent and sample molecules simultaneously, resulting in rapid temperature increases that can significantly reduce extraction time to minutes rather than hours [32]. The technique's main advantages include reduced solvent consumption and faster extraction kinetics, though it requires specialized equipment and may not be suitable for all analyte types [32].
Accelerated solvent extraction (ASE), also known as pressurized liquid extraction, uses high pressure and temperature to achieve faster extractions with reduced solvent consumption [32]. The elevated temperatures increase analyte solubility and mass transfer rates, while the pressure keeps solvents in a liquid state above their normal boiling points. This method has gained prominence in environmental analysis for its efficiency in extracting organic pollutants from solid matrices [32].
Comparative Analysis of Extraction Techniques
Table 2: Extraction Method Comparison for Low-Yield Forensic Applications
| Method | Extraction Time | Solvent Volume | Temperature | Advantages | Limitations |
|---|---|---|---|---|---|
| Soxhlet (Optimized) | 4-8 hours (reduced) | 100-250 mL | Solvent BP | Exhaustive; established validation; quantitative [32] | High solvent use; long extraction; thermal degradation risk |
| Ultrasound-Assisted (UAE) | 20-60 minutes | 50-100 mL | 25-50°C | Rapid; low temperature; simple apparatus [33] | Potential matrix dependency; less exhaustive |
| Microwave-Assisted (MAE) | 5-30 minutes | 20-50 mL | Above solvent BP | Very fast; low solvent consumption [32] | Specialized equipment; heating uniformity issues |
| Accelerated Solvent (ASE) | 10-20 minutes | 15-40 mL | 40-200°C | Fast; automated; low solvent use [32] | High equipment cost; potential thermal degradation |
The selection of an appropriate extraction method must consider the specific requirements of the forensic investigation. For research and method development where exhaustive extraction is required for quantitative analysis, optimized Soxhlet remains valuable [32]. For rapid screening in time-sensitive investigations, UAE and MAE offer compelling alternatives with significantly reduced processing times [33]. When processing large sample batches or when solvent consumption is a concern, ASE provides an efficient though capital-intensive solution [32].
Experimental Protocols for Method Evaluation
Optimized Short-Duration Soxhlet Protocol
Materials and Equipment:
- Soxhlet extraction apparatus (borosilicate glass, 40mm ID)
- Round bottom flask (250-500mL)
- Allihn reflux condenser
- Extraction thimbles (cellulose or glass fiber)
- Heating mantle with temperature control
- Solvent: Appropriate for target explosives (e.g., acetone, acetonitrile)
- Anti-bumping granules or magnetic stir bar
Procedure:
- Sample Preparation: Reduce solid samples to 0.5-2mm particle size using a mechanical grinder. For post-blast residues (e.g., swabs, soil samples), mix thoroughly with anhydrous sodium sulfate at a 1:2 ratio to remove moisture [32].
- Apparatus Assembly: Accurately weigh 5-10 grams of prepared sample and load into an extraction thimble, avoiding overpacking. Add appropriate solvent (typically 150-200mL) to the round bottom flask with anti-bumping granules. Securely assemble the Soxhlet extractor and condenser, ensuring snug-fitting joints and proper clamping [32].
- Extraction Process: Begin heating the solvent to gentle reflux, adjusting temperature to maintain steady vapor production (typically 20°C above the solvent boiling point). Monitor initial cycles to ensure proper siphoning action occurs every 5-15 minutes. Continue extraction for a predetermined optimized time (4-8 hours for reduced duration) based on preliminary method development [32].
- Post-Extraction Processing: After the prescribed extraction time, turn off heating and allow the system to cool. Collect the extract-containing solvent from the flask. Concentrate using rotary evaporation or nitrogen blowdown to achieve appropriate analyte concentration for subsequent analysis [32].
- Analysis and Validation: Analyze concentrated extracts using appropriate analytical methods (e.g., GC-VUV, DART-MS). Validate method performance using certified reference materials and spike-recovery experiments with relevant explosive compounds [13] [12].
Ultrasound-Assisted Extraction Protocol for Rapid Screening
Materials and Equipment:
- Ultrasonic bath (40kHz frequency)
- Centrifuge tubes (50mL)
- Centrifuge
- Solvent: Appropriate for target analytes
- Temperature control system
Procedure:
- Sample Preparation: Weigh 1-2 grams of prepared sample (0.5-2mm particle size, dried) into a 50mL centrifuge tube [33].
- Solvent Addition: Add 20mL of appropriate extraction solvent (sample-to-solvent ratio of 1:20) [33].
- Sonication: Place tubes in ultrasonic bath pre-set to 25°C. Extract for 20 minutes at 40kHz frequency [33].
- Separation: Centrifuge at 1680 rpm for 10 minutes to separate solid residue [33].
- Concentration: Transfer supernatant to evaporation flask and concentrate under reduced pressure at 40°C [33].
- Analysis: Reconstitute in appropriate solvent for instrumental analysis.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Explosive Residue Extraction
| Reagent/Material | Function | Application Notes |
|---|---|---|
| Anhydrous Sodium Sulfate | Moisture removal from samples | Prevents solvent dilution; improves extraction efficiency [32] |
| Cellulose/Glass Fiber Thimbles | Sample containment during Soxhlet | Allows solvent permeation while retaining solid matrix [32] |
| Acetone (>99.7%) | Extraction solvent for organic explosives | Effective for a wide range of explosive compounds; appropriate for GC and MS analysis [12] |
| Acetonitrile (≥99.9%) | Alternative extraction solvent | Different selectivity profile; useful for polar nitroaromatics [12] |
| Anti-Bumping Granules | Prevent violent solvent boiling | Ensure smooth reflux in Soxhlet apparatus [32] |
| Deuterated Internal Standards | Quantification and quality control | Account for extraction efficiency and matrix effects [13] |
Analytical Integration and Method Validation
Coupling Extraction with Advanced Detection
The ultimate effectiveness of any extraction protocol is determined by its compatibility with subsequent analytical techniques. In post-blast investigation, advanced instrumental methods including gas chromatography-vacuum UV spectroscopy (GC-VUV) and direct-analysis-in-real-time mass spectrometry (DART-MS) provide the specificity and sensitivity required for definitive identification [13] [12]. The following diagram illustrates the complete analytical workflow from sample collection to identification:
Diagram: Post-Blast Residue Analysis Workflow
GC-VUV has emerged as a powerful tool for explosives analysis, with sensitivity in the low parts-per-million range for many explosive compounds and the ability to differentiate structurally similar analytes based on their vacuum UV absorption spectra [13]. The technique's selectivity stems from the fact that certain functional groups reliably absorb in distinct regions of the VUV spectrum, allowing chemists to cancel out interferences from complex post-blast matrices [13]. Similarly, DART-MS enables rapid analysis of explosive residues with minimal sample preparation, directly analyzing swabs or bomb fragments by proximate contact that simultaneously thermally desorbs and ionizes substances for mass spectrometric analysis [12].
Validation Framework for Forensic Applications
Method validation for forensic extraction protocols must establish specificity, accuracy, precision, and robustness using appropriate reference materials. For quantitative applications, the use of isotopically labeled internal standards is recommended to account for variable extraction efficiency and matrix effects [13]. Sensitivity requirements should reflect the challenging nature of post-blast residues, where explosive concentrations may be in the parts-per-billion range or lower [13].
Specificity must be demonstrated through analysis of blank matrices and potentially interfering substances commonly encountered in post-blast environments. Precision should be established through replicate extractions of fortified samples at relevant concentrations, with acceptance criteria defined based on the method's intended use [13] [12]. For qualitative screening methods, the focus should be on detection limits and the ability to reliably identify target explosives above established threshold concentrations.
Optimizing extraction methodologies for low-yield scenarios represents a critical challenge in advancing forensic capabilities for post-blast investigation. While traditional Soxhlet extraction can be modified for reduced processing times through strategic parameter optimization, alternative techniques including ultrasound-assisted and microwave-assisted extraction offer compelling advantages for time-sensitive applications. The selection of an appropriate extraction strategy must balance multiple factors, including required throughput, available equipment, analyte stability, and the specific demands of subsequent analytical techniques.
Future advancements in this field will likely incorporate machine learning frameworks for highly parallel multi-objective reaction optimization, similar to approaches being developed in pharmaceutical process chemistry [34]. These data-driven strategies could systematically optimize extraction parameters while balancing competing objectives such as maximized yield, minimized time, and reduced solvent consumption. Additionally, the ongoing development of greener extraction technologies aligns with broader chemical industry trends focusing on sustainability and efficiency [35]. For forensic researchers and scientists, the continued refinement of extraction protocols remains foundational to enhancing investigative capabilities and advancing the scientific rigor of post-blast explosives analysis.
The forensic identification of explosive residues in post-blast scenes presents significant analytical challenges, particularly for nitrate-based explosives such as ammonium nitrate fuel oil (ANFO), urea nitrate, and black powder in environments experiencing rainfall. Nitrate ions are of paramount forensic significance as they constitute key components of the most commonly used improvised explosives globally, yet their high water solubility makes them particularly susceptible to dilution, leaching, and complete removal from blast sites by rainwater [17]. This environmental effect substantially increases the risk of false-negative results during crime exhibit evaluation, potentially compromising legal outcomes and criminal investigations [17].
Within the broader context of post-blast residue sampling and concentration techniques research, this technical guide addresses the specific methodological adaptations required for reliable nitrate ion detection in rainy conditions. The protocols outlined herein are designed to overcome the particular difficulties posed by the high mobility of nitrate ions in wet environments, where rainwater can significantly alter the concentration and distribution of explosive residues in soil matrices [17]. By implementing specialized sampling strategies and highly sensitive analytical techniques, forensic investigators can improve detection capabilities even when environmental conditions have degraded the evidentiary value of blast scenes.
Impact of Rainfall on Nitrate Ion Distribution and Retention
Rainfall dramatically affects the fate and distribution of nitrate ions in post-blast scenes through two primary mechanisms: dissolution and translocation. The high solubility of nitrate ions in water enables rapid dissolution from explosive residues, while subsequent percolation through soil profiles leads to vertical migration and potential redistribution throughout the substrate [17]. Research demonstrates that simulated rainfall conditions cause significant movement of nitrate ions from surface layers to subsurface layers, fundamentally altering the original residue distribution pattern and complicating forensic interpretation.
Quantitative Analysis of Nitrate Redistribution
Recent forensic research investigating post-blast soil analysis in rainy environments has yielded critical quantitative data on nitrate ion retention and redistribution. The following table summarizes key findings from a controlled study examining nitrate concentration variations between surface and subsurface soil layers after exposure to natural drizzling rainfall for one week:
Table 1: Nitrate Ion Concentration in Soil Layers After Rainfall Exposure
| Sample Description | Nitrate Ion Concentration (ppm) | Percentage of Standard Concentration |
|---|---|---|
| Standard Potassium Nitrate (1g) | 842.522 | 100.0% |
| Simulated Pit Top Layer (SPT) | 38.242 | 4.5% |
| Simulated Pit Bottom Layer (SPB) | 24.971 | 3.0% |
| Control Pit Top Layer (CPT) | 14.316 | 1.7% |
| Control Pit Bottom Layer (CPB) | 8.381 | 1.0% |
Data derived from ion chromatography analysis demonstrates that even after controlled rainfall exposure, detectable nitrate concentrations persist in soil, though at significantly reduced levels compared to the original standard [17]. The higher concentration in the top layer (SPT) compared to the bottom layer (SPB) of the simulated pit suggests complex transport mechanisms, potentially including both downward percolation and surface evaporation effects. Furthermore, the detectable levels in control pit samples highlight the importance of establishing background nitrate levels when interpreting analytical results, as environmental sources such as fertilizers can contribute to baseline nitrate concentrations [17].
Analytical Technique: Ion Chromatography Methodology
Ion chromatography (IC) has emerged as the premier analytical technique for detecting trace-level nitrate ions in complex soil matrices due to its superior sensitivity, selectivity, and suitability for quantitative analysis of inorganic anions [17]. The methodology enables reliable identification and quantification of nitrate ions even at significantly reduced concentrations resulting from rainfall leaching.
Detailed Ion Chromatography Protocol
Table 2: Ion Chromatography System Configuration and Parameters
| Parameter | Specification |
|---|---|
| IC System | Metrohm Advanced Ion Chromatography system |
| Separation Column | Metrosep A Supp 19 (150 × 4.0 mm, 4.6 μm particle size) |
| Column Composition | Hydrophilic polystyrene/divinylbenzene copolymer with quaternary ammonium groups |
| Guard Column | Metrosep A SUPP 4/5 |
| Eluent Composition | 8.0 mM sodium carbonate (Na₂CO₃) and 0.25 mM sodium bicarbonate (NaHCO₃) |
| Flow Rate | 0.7 mL/min |
| Detection Method | Conductivity detection |
| Suppressor Regeneration | Sulfuric acid solution |
The analytical procedure follows a systematic workflow: First, the IC system is calibrated using standard solutions of inorganic anions (F⁻, Cl⁻, NO₂⁻, SO₄²⁻, PO₄³⁻) prepared from certified 10 ppm stock solutions [17]. Soil sample extracts are then injected into the chromatographic system, where anions are separated based on their affinity for the stationary phase. Nitrate ions are identified by matching their retention times with known standards, while quantification is achieved by comparing peak areas against the calibration curve [17]. This method provides the sensitivity required to detect nitrate ions at parts-per-million (ppm) concentrations, which is essential for analyzing rainfall-affected samples where target analyte concentrations may be substantially diminished.
Comprehensive Sample Collection and Preparation Protocol
Strategic Sample Collection in Rain-Affected Environments
Effective sample collection from post-blast scenes exposed to rainfall requires strategic adaptation of standard protocols to account for hydrological transport of water-soluble nitrate ions. Forensic investigators should collect samples from multiple locations and depths, with particular attention to:
- Crater and Perimeter Sampling: Collect 100-120 grams of soil from both the blast crater and surrounding perimeter areas [17].
- Layered Depth Sampling: Obtain separate samples from surface (0-2 cm) and subsurface (5-10 cm) layers to account for vertical migration of nitrate ions due to rainwater percolation [17].
- Control Samples: Gather control samples from adjacent unaffected areas at similar depths to establish background nitrate levels [17].
- Multiple Sampling Points: Implement grid-based sampling strategies with documentation of each sample's relative position to the epicenter to reconstruct dispersion patterns.
Post-collection, samples should be immediately transferred to clean, labeled containers, preventing cross-contamination and preserving moisture content for subsequent analysis.
Sample Preparation Workflow
Table 3: Soil Sample Preparation Protocol for Nitrate Ion Extraction
| Step | Procedure | Purpose | Critical Parameters |
|---|---|---|---|
| Initial Processing | Air-dry samples at room temperature | Stabilize samples without chemical alteration | Avoid elevated temperatures that may degrade analytes |
| Weighing | Measure predetermined sample portions | Ensure consistent analytical input | Typical portion: 10-20g representative sample |
| Extraction | Add 100ml demineralized water; agitate thoroughly | Transfer nitrate ions from soil to aqueous phase | Repeat washing (3×100ml) for complete recovery |
| Filtration | Filter through qualitative filter paper | Remove particulate soil matter | Use 0.22μm syringe filters for final filtration |
| Concentration | Evaporate combined filtrates to dryness | Pre-concentrate analytes | Reconstitute in smaller volume (e.g., 100ml) |
| Final Preparation | Dilute 1ml extract tenfold with DM water | Achieve appropriate analytical concentration | Optimize for detector linear range |
The preparation methodology emphasizes complete recovery through multiple washing stages, as a single extraction may leave significant nitrate quantities bound to soil particles [17]. The concentration step via evaporation is particularly crucial for rainfall-affected samples where analyte concentrations may be substantially diluted.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful analysis of nitrate ions in post-blast residues requires specific high-purity reagents and specialized materials to ensure analytical accuracy and prevent contamination.
Table 4: Essential Research Reagents and Materials for Nitrate Ion Analysis
| Reagent/Material | Specification | Function in Protocol |
|---|---|---|
| Demineralized Water | High-purity, residue-free | Extraction solvent for nitrate ions from soil matrix |
| Potassium Nitrate Standard | Certified reference material | Calibration standard for quantification |
| Sodium Carbonate | AR Grade (Merck, Germany) | Mobile phase component for ion chromatography |
| Sodium Bicarbonate | AR Grade (Merck, Germany) | Mobile phase component for ion chromatography |
| Syringe Filters | 0.22 μm pore size (Clairofilt) | Removal of fine particulates prior to IC analysis |
| Filter Paper | Qualitative grade (Clairofilt) | Initial filtration of soil extracts |
| Sulfuric Acid Solution | Reagent grade (Sigma Aldrich) | Suppressor regeneration in IC system |
| Anion Standards | F⁻, Cl⁻, NO₂⁻, SO₄²⁻, PO₄³⁻ from 10 ppm stock (Sigma Aldrich) | System suitability verification and calibration |
The selection of high-purity reagents is critical for maintaining low background levels and preventing interference during analysis, particularly when working with trace-level nitrate concentrations in rainfall-affected samples [17].
Quality Assurance and Method Validation
Robust quality assurance protocols are essential for generating forensically defensible data when analyzing nitrate ions in challenging environmental conditions. Key considerations include:
- Method Blank Analysis: Include demineralized water blanks processed through the entire extraction and analytical procedure to identify potential contamination sources.
- Control Samples: Analyze control pit samples to establish background nitrate levels and differentiate explosive-origin nitrates from environmental sources [17].
- Matrix Spike Recovery: Fortify control soil samples with known nitrate concentrations to determine extraction efficiency, with target recovery rates of 85-115%.
- Duplicate Analysis: Process analytical duplicates to assess method precision, with acceptable relative percent difference typically <10%.
- Continuing Calibration Verification: Analyze mid-range calibration standards after every 10-15 samples to confirm instrumental response stability.
These quality control measures are particularly crucial when analyzing samples from rainy environments where nitrate concentrations may be near detection limits, ensuring that reported results accurately reflect field conditions rather than analytical artifacts.
The reliable detection of nitrate ions in post-blast scenes affected by rainfall requires specialized approaches throughout the entire analytical process, from crime scene sampling to instrumental analysis. Based on current research, the following evidence-based recommendations emerge for forensic practitioners:
First, implement stratified sampling protocols that account for the vertical migration of water-soluble nitrate ions, systematically collecting from both surface and subsurface layers to maximize detection probability [17]. Second, employ concentration techniques during sample preparation to counteract the dilution effects of rainfall, utilizing multiple extraction cycles and evaporation steps to improve analytical sensitivity [17]. Third, utilize ion chromatography as the primary analytical technique due to its demonstrated capability to detect trace-level nitrate ions in complex soil matrices, with rigorous quality control measures to ensure result reliability [17].
Future research directions should focus on developing more efficient extraction methodologies for water-soluble residues from soil matrices, establishing statistical models to estimate original explosive quantities based on residual nitrate patterns in rain-affected scenes, and exploring complementary analytical techniques that may provide orthogonal confirmation of explosive residues. Through the systematic implementation of these specialized protocols, forensic investigators can significantly improve the evidentiary value of post-blast residue analysis, even when environmental conditions have compromised the crime scene.
The analysis of trace-level target compounds, such as explosive residues or pesticides, within complex matrices is a fundamental challenge in analytical chemistry. These matrices—ranging from post-blast debris to food and environmental samples—contain numerous co-extracted substances that can interfere with instrumental analysis, a phenomenon known as the matrix effect [36]. Matrix effects can adversely affect quantification and qualification by suppressing or enhancing the analyte signal, leading to inaccurate results [36]. In the critical context of post-blast residue sampling, where rapid and accurate identification of explosive fillers can provide key forensic intelligence for an investigation, ensuring reliable data is paramount [12]. This guide details established and emerging clean-up procedures designed to minimize this background interference, thereby ensuring the accuracy, sensitivity, and reliability of analytical results.
The Core Challenge: Matrix Effects in Complex Samples
A matrix effect is defined as the influence of one or more undetected components from a sample on the measurement of an analyte's concentration or mass [36]. In techniques like Gas Chromatography tandem Mass Spectrometry (GC-MS/MS) or Liquid Chromatography tandem Mass Spectrometry (LC-MS/MS), these effects can manifest as:
- Signal Suppression or Enhancement: Co-extracted matrix components can compete with analytes for ionization or interact with active sites in the analytical system, altering the detected signal [36].
- Reduced Analytical Performance: Matrix effects can increase background noise, raise detection limits, and cause poor reproducibility [12].
The complexity is magnified in matrices such as dried herbs, fruits, and post-blast residues, which contain high levels of interfering substances like lipids, pigments, flavonoids, essential oils, and inorganic materials [36] [12]. Consequently, a robust clean-up procedure is not optional but essential for valid data.
Established Clean-up Techniques and Protocols
Several physical and chemical strategies are employed to purify samples before instrumental analysis. The following are key techniques with detailed methodologies.
Solid-Phase Extraction (SPE)
SPE is a widely used method for selective cleanup. It involves passing the sample extract through a cartridge or column containing a sorbent that selectively retains either the target analytes or the interfering compounds.
Experimental Protocol: Selective Cleanup with MgO Microspheres This protocol is adapted from a method developed for the selective isolation of Benzo[a]pyrene (BaP) from complex samples like sediments, soils, and animal tissue [37].
- Sorbent Preparation: Pack a solid-phase extraction column with magnesium oxide (MgO) microspheres as the sorbent.
- Column Conditioning: Condition the MgO sorbent with an appropriate organic solvent (e.g., n-hexane).
- Sample Loading: Load the prepared sample extract onto the column using an optimized loading solvent.
- Rinsing: Rinse the column with a defined volume of a solvent mixture to remove impurities such as lipids, sulphur, pigments, halobenzenes, and dioxins without eluting the target analyte.
- Elution: Elute the purified target analyte (BaP) with a specific volume of a stronger elution solvent.
- Analysis: Concentrate the eluent and reconstitute it for analysis by GC-MS or LC-MS.
This method has demonstrated high recovery rates (85.1–100.8%) and superior selectivity compared to traditional sorbents like silica gel or florisil, while also reducing organic solvent consumption [37].
The QuEChERS Method
While the core QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) method involves extraction and partitioning, a dispersive-SPE (d-SPE) clean-up step is integral to its effectiveness for complex matrices.
Experimental Protocol: d-SPE Clean-up for Pesticides in Dried Herbs This protocol is derived from a study on minimizing matrix effects in the analysis of 236 pesticides in dried herbs and fruits using GC-MS/MS [36].
- Extraction: Extract the homogenized sample with acetonitrile.
- Partitioning: Add a salt mixture (e.g., MgSO₄, NaCl, trisodium citrate dihydrate) to induce phase separation.
- d-SPE Clean-up: Transfer an aliquot of the acetonitrile extract into a tube containing a d-SPE sorbent mixture. Common sorbents include:
- PSA (Primary-Secondary Amine): Removes fatty acids and other polar organic acids.
- ENVI-Carb (Graphitized Carbon Black): Removes pigments and sterols.
- MgSO₄: Removes residual water.
- Vortex and Centrifuge: Vortex the mixture vigorously, then centrifuge it to pellet the sorbents and adsorbed impurities.
- Analysis: Transfer the purified supernatant for analysis.
Micro-SPE Pipette Tips
For small-volume samples and trace-level analysis, micro-SPE pipette tips offer a miniaturized and efficient clean-up solution.
Experimental Protocol: ZipTip Clean-up for Oligonucleotides or Proteins This protocol is standardized for cleaning up samples for Mass Spectrometry (MS) analysis [38].
- Pre-wet: Attach the ZipTip (with C18 resin for peptides, C4 for large proteins) to a pipettor. Aspirate and dispense 50% acetonitrile in water to waste to wet the sorbent bed. Repeat.
- Equilibrate: Wash the tip 3 times with 10 µL of an equilibration solution (e.g., 0.1 M TEAA for oligonucleotides) to prepare the sorbent for binding.
- Bind Sample: Aspirate and dispense the sample solution (up to 10 µL) 5-10 times to allow the analyte to bind to the sorbent.
- Wash: Wash the tip 3 times with 10 µL of a wash solution (e.g., 0.1 M TEAA followed by water) to remove salts and impurities.
- Elute: Elute the purified analyte by aspirating and dispensing a small volume (1-10 µL) of an elution solvent (e.g., 50% acetonitrile/50% water) 3-10 times.
Table 1: Comparison of Established Clean-up Techniques
| Technique | Principle | Best For | Advantages | Limitations |
|---|---|---|---|---|
| Solid-Phase Extraction (SPE) | Selective adsorption onto a sorbent | Broad-range purification; high-interference samples [37] | High selectivity; effective impurity removal; can be automated [37] | Can be time-consuming; requires optimization; sorbent cost |
| QuEChERS (d-SPE) | Dispersive binding of impurities | Multi-residue analysis in food and environmental samples [36] | Quick and easy; low solvent use; high throughput | May be insufficient for extremely complex matrices |
| Micro-SPE (ZipTip) | Miniaturized solid-phase extraction | Small-volume samples; trace-level analytes for MS [38] | Minimal sample loss; ideal for MS analysis; easy to use | Limited binding capacity; for small volumes only |
Advanced and Complementary Strategies
Beyond physical clean-up, chemical and procedural strategies are critical for counteracting matrix effects that persist after sample preparation.
Matrix-Matched Calibration
This approach involves preparing calibration standards in a blank, but otherwise identical, sample matrix. The matrix-matched standards experience the same matrix effects as the real samples, thus compensating for them during quantification [36]. The main challenge is sourcing a truly uncontaminated matrix.
Analyte Protectants (APs)
Analyte protectants are compounds (e.g., ethyl glycerol, gulonolactone) added to the sample or injected into the system to interact with active sites in the GC inlet and column. By "blocking" these active sites, APs prevent the adsorption and degradation of target analytes, thereby minimizing the matrix-induced enhancement effect [36].
Application Methods:
- Addition to Extract: The mixture of APs is added directly to every sample extract before injection [36].
- Injection at Sequence Start: A mixture of APs is injected at the beginning of the analytical sequence to pre-condition the GC system, a newer approach that has shown success in minimizing matrix effects for a wide range of pesticides [36].
Internal Standards
The use of internal standards, particularly isotope-labeled analogues of the target analytes, is a highly effective strategy. Because the internal standard is chemically identical to the analyte and is subjected to the same extraction, clean-up, and matrix effects, any signal variation affects both equally. This allows for precise correction of analyte response during quantification [36].
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Clean-up of Complex Matrices
| Reagent / Material | Function in Clean-up |
|---|---|
| PSA (Primary-Secondary Amine) | d-SPE sorbent; removes fatty acids, organic acids, and some sugars [36]. |
| ENVI-Carb / GCB | d-SPE sorbent; removes pigments (e.g., chlorophyll) and sterols [36]. |
| C18 | Reverse-phase sorbent for SPE/micro-SPE; retains non-polar compounds; widely used for peptide analysis [38]. |
| MgO Microspheres | SPE sorbent; offers selective isolation of target analytes and removal of various impurities like lipids and pigments [37]. |
| Analyte Protectants (e.g., ethyl glycerol) | Chemical masking agents; block active sites in the GC system to minimize matrix effects [36]. |
| Isotope-Labeled Internal Standards | Correction standard; accounts for variability in extraction efficiency and matrix effects during quantification [36]. |
Visualizing the Strategic Workflow
The following diagram illustrates the logical decision-making process for selecting and applying clean-up procedures within an analytical workflow, particularly for a post-blast analysis context.
Diagram 1: A strategic workflow for selecting clean-up procedures to minimize background interference.
The accurate analysis of target compounds in complex matrices is a non-trivial endeavor, demanding a systematic approach to clean-up. As demonstrated, a combination of physical clean-up techniques—such as selective SPE, QuEChERS, and micro-SPE—with advanced chemical strategies—like analyte protectants and isotope dilution—provides a powerful toolkit to combat background interference. This is especially critical in fields like forensic explosive analysis, where the integrity of results can have significant investigative and legal implications. The choice of method must be guided by the nature of the matrix, the analytes of interest, and the required sensitivity. By rigorously applying these procedures, researchers and scientists can ensure their data is both reliable and definitive.
The analysis of post-blast residues presents a significant forensic challenge, requiring the detection and identification of trace amounts of unreacted explosives within complex environmental matrices. Pentaerythritol tetranitrate (PETN), a high explosive common in military applications and improvised explosive devices (IEDs), is particularly problematic due to its intrinsic thermal lability. During standard gas chromatography-mass spectrometry (GC-MS) analysis, PETN undergoes thermal decomposition in the heated injection port and chromatographic system, leading to extensive fragmentation and the absence of a molecular ion under conventional electron impact (EI) ionization conditions. This decomposition complicates identification, introduces analytical ambiguities, and reduces method sensitivity, especially at the trace levels encountered in post-blast debris [1] [39].
The accurate characterization of post-blast residues is a crucial step in forensic investigations, helping to identify explosive materials and uncover terrorist activities. Overcoming the analytical challenges associated with thermally sensitive compounds like PETN is therefore not merely a technical exercise but a necessity for supporting law enforcement and ensuring public safety. This guide details specialized GC-MS setups and methodologies developed to enable the reliable detection of intact PETN, with direct application within a broader research context focused on optimizing post-blast residue sampling and concentration techniques [1] [19].
Technical Solutions and Methodologies
Optimization of the GC Inlet and Temperature Program
The primary point of degradation for thermally labile compounds is often the hot injection port. Specialized inlet systems and carefully controlled temperature programming are critical to vaporize the analyte without causing its thermal breakdown.
Programmable Temperature Vaporization (PTV) Inlet: The use of a PTV inlet is a cornerstone technique for analyzing labile compounds. Unlike a standard isothermal inlet, a PTV allows the sample to be introduced at a low temperature, which is then rapidly increased at a controlled rate to transfer the volatilized analyte onto the column.
- Optimal Ramp Rates: Research on the analysis of BDNPA/F, another thermally labile energetic material, demonstrated that a fast PTV ramp rate of 60 °C/min from an initial 60 °C to a final 325 °C significantly reduced thermal decomposition compared to a standard isothermal inlet held at 325 °C [40].
- Inlet Temperature: Maintaining a lower inlet temperature is also viable. One study confirmed that intact PETN could be analyzed with a GC inlet temperature of 175 °C, observing a single chromatographic peak without signs of decomposition [1].
GC Oven Program: A suitably designed column temperature program further mitigates on-column degradation. While specific oven programs for PETN vary, a common strategy involves a moderate temperature ramp to achieve efficient separation while minimizing the time the analyte spends at high temperatures.
Alternative Ionization Techniques
The standard 70 eV Electron Impact (EI) ionization causes extensive fragmentation of the PETN molecule, complicating its identification. Alternative ionization methods can provide more informative spectral data.
- Flowing Atmospheric-Pressure Afterglow (FAPA): This ambient ionization technique, when coupled to a high-resolution mass spectrometer (HRMS), has been used to definitively identify intact PETN by observing its molecular adduct ions, such as
[M]⁻and[M+NO₃]⁻, which are not typically seen in EI [39]. - Chemical Ionization (CI): Both Negative Chemical Ionization (NCI) and Positive Chemical Ionization (PCI) are softer techniques that produce less fragmentation than EI, often yielding
[M-H]⁻or[M+adduct]⁺ions that are crucial for confirming the molecular weight of an unknown compound [40]. - Derivatization: PETN can be chemically derivatized before analysis to form a more stable and volatile compound that is more amenable to GC-MS analysis. Nitrate ester derivatization has been applied to generate structurally informative ions for PETN via GC-MS [1].
Supporting Analytical Techniques
While GC-MS is a powerful confirmatory tool, other techniques play a vital role in the initial screening and detection of explosive residues.
- Color Tests and Thin-Layer Chromatography (TLC): Classical spot tests and TLC remain useful for initial screening of post-blast extracts. For TLC visualization of PETN, a developed plate can be lightly sprayed with sodium hydroxide, heated at 100 °C for 10 minutes, and then treated with Griess reagent to produce a visible spot [1].
- Ion Mobility Spectrometry (IMS): IMS provides excellent mass detection limits, rapid analysis time, and small sample requirements, making it attractive for the forensic analysis of explosive materials in post-detonation debris [1].
- Nuclear Magnetic Resonance (NMR) Spectroscopy: As a non-destructive technique,
^1HNMR is highly selective and sensitive for identifying explosives and elucidating their structure directly from post-blast debris, requiring simple sample preparation [19].
Experimental Protocols and Workflows
Sample Preparation from Soil Matrices
The extraction of unburnt explosive residues from post-blast soil samples is a critical first step. Solid-Phase Extraction (SPE) is highly effective for cleaning up samples and concentrating analytes.
- Conditioning: Condition a Hypersep Retain C-X 60 mg SPE column with 3 mL of acetone, followed by 3 mL of acetonitrile, 3 mL of methanol, and finally 3 mL of distilled water [19].
- Loading: Load the soil extract (in acetone) onto the conditioned SPE column [19].
- Washing: Wash the column with 3 mL of distilled water to remove interfering polar compounds [19].
- Drying: Dry the column by applying a vacuum for 5 minutes [19].
- Elution: Elute the target explosive residues using 3 mL of acetone into a clean test tube [19].
- Concentration: Concentrate the eluent to dryness under a gentle stream of nitrogen gas and reconstitute in a suitable solvent for instrumental analysis [19].
Optimized GC-HRMS Method for Labile Compounds
The following protocol, adapted from work on BDNPA/F, outlines an optimized GC-HRMS method suitable for thermally labile compounds [40].
Injection:
- Inlet: PTV
- Mode: Splitless
- Injection Volume: 1 μL
- PTV Program: Start at 60 °C, then ramp at 60 °C/min to a final temperature of 325 °C.
GC Separation:
- Column: Rxi-5 ms, 30 m × 0.25 mm i.d. × 0.25 μm d.f. (or equivalent)
- Carrier Gas: Helium, constant flow of 1.4 mL/min.
- Oven Program: Hold at 40 °C for 2.25 min, then ramp at 10 °C/min to the final temperature (e.g., 325 °C).
Mass Spectrometry:
- Transfer Line Temperature: Set to match the final PTV temperature (e.g., 325 °C).
- Ionization: Electron Impact (EI) at 70 eV.
- Ion Source Temperature: 250 °C.
- Mass Range: 35–500 m/z.
- Solvent Delay: 200 s.
Workflow for Post-Blast Residue Analysis
The following diagram illustrates the integrated workflow for analyzing PETN in post-blast samples, from collection to identification.
Diagram 1: Integrated analytical workflow for PETN identification in post-blast residues.
Data Presentation and Analysis
Key Research Reagent Solutions
Table 1: Essential reagents and materials for the analysis of PETN and other explosives.
| Item | Function/Description | Example Application |
|---|---|---|
| Solid-Phase Extraction (SPE) Column | For clean-up and concentration of trace explosive residues from complex soil extracts. Removes interferences like oils and plasticizers. | Hypersep Retain C-X 60 mg columns used to purify soil extracts for NMR and other analyses [19]. |
| SPME Fiber | For headspace sampling or direct deposition. A non-exhaustive, solvent-less extraction technique ideal for volatile and semi-volatile analytes. | 65 μm PDMS/DVB fibers used for headspace sampling of explosive evidence or direct deposition of standards [41]. |
| Griess Reagent | A colorimetric reagent used for the detection of nitrite, which is formed when nitrate esters (like PETN) are degraded or reduced. | Used as a spray for visualizing PETN and other nitrate esters on Thin-Layer Chromatography (TLC) plates [1]. |
| Sodium Hydroxide (NaOH) Solution | Used as a developing agent in TLC to hydrolyze nitrate esters, facilitating their visualization with Griess reagent. | A 10% NaOH solution sprayed on TLC plates to enable detection of PETN [1]. |
| GC Capillary Column | The stationary phase for chromatographic separation. A non-polar or low-polarity phase is standard for explosive analysis. | Restek MXT-5 (5% diphenyl / 95% dimethyl polysiloxane) used for fast separation of explosive standards [41]. |
Comparison of Ionization Techniques for PETN
Table 2: A comparison of mass spectrometric ionization techniques used in the analysis of PETN.
| Ionization Technique | Key Characteristics for PETN | Advantages | Disadvantages |
|---|---|---|---|
| Electron Impact (EI) | Causes extensive fragmentation; molecular ion ([M]⁺) is typically absent; spectrum is dominated by NO₂⁺ and other fragment ions [1]. |
- Standard, widely available- Extensive library searchable databases | - Difficult to identify parent compound due to fragmentation- Not ideal for confirmation at trace levels |
| Chemical Ionization (CI) | Softer ionization; can yield adduct ions like [M+NO₃]⁻ in negative mode or [M+adduct]⁺ in positive mode, providing molecular weight information [40]. |
- Less fragmentation than EI- Provides molecular weight information- Increased specificity | - Requires tuning and reagent gas- Library databases are less extensive than for EI |
| Flowing Atmospheric-Pressure Afterglow (FAPA) | An ambient ionization technique that can be coupled to GC effluent; observes [M]⁻ and [M+NO₃]⁻ ions, confirming intact PETN [39]. |
- Minimal fragmentation- Definitive identification of molecular ion via high mass accuracy | - Not a standard configuration on most commercial GC-MS systems |
The reliable GC-MS analysis of thermally labile explosives like PETN in post-blast residues is achievable through a systematic approach that addresses the key points of analyte degradation. The integration of specialized hardware, particularly PTV inlets with optimized temperature programs, is fundamental to preserving the analyte's integrity during vaporization. Coupling this with softer ionization techniques such as CI or advanced methods like FAPA-HRMS overcomes the limitations of traditional EI and provides the confirmatory data necessary for definitive identification. These technical solutions, embedded within a robust sample preparation and analytical workflow, significantly enhance the capabilities of forensic scientists. By enabling the accurate detection of PETN at trace levels, these methodologies provide critical intelligence for post-blast investigations and contribute powerfully to a broader research thesis aimed at advancing the science of explosive residue analysis.
Post-blast forensic investigations frequently involve the analysis of oversized and fragmented evidence, such as vehicle fragments, concrete blocks, and metallic debris, which are often contaminated with mixed explosive formulations. These heterogeneous substrates present significant challenges for residue recovery and analysis when conventional methods, optimized for small, homogeneous samples, are applied. Within the broader context of research on post-blast residue sampling and concentration techniques, the development of robust forensic strategies tailored to such complex evidence is paramount. Spatial subsampling has emerged as a critical methodology for enhancing detection sensitivity and analytical reliability in these scenarios, providing a systematic framework for investigating complex detonation events [16]. This technical guide explores the integrated workflows, experimental protocols, and analytical techniques that constitute effective spatial subsampling approaches for heterogeneous evidence in forensic investigations.
Core Principles and Methodological Framework
The Challenge of Heterogeneous Evidence
Oversized exhibits from explosive detonations represent particularly challenging substrates for forensic analysis due to their irregular surfaces, fragmented nature, and heterogeneous contamination patterns. Traditional sampling methods often fail to adequately address the spatial distribution of residues across these complex surfaces, potentially missing critical evidence or producing unrepresentative analytical results. The fundamental principle underlying spatial subsampling is the systematic division of large, heterogeneous exhibits into logically defined spatial regions for individual analysis, thereby increasing the probability of detecting low-abundance residues and enabling the mapping of contamination patterns across the evidence surface [16].
Spatial Subsampling Conceptual Framework
Spatial subsampling operates on the premise that explosive residues are not uniformly distributed across oversized exhibits following detonation. Instead, residue deposition follows physically determined patterns influenced by factors including exhibit geometry, material composition, and orientation relative to the blast epicenter. The subsampling process involves two primary approaches:
- Bounding Box Query: Defines rectangular regions of interest based on minimum and maximum coordinates within a specified coordinate system, particularly effective for preliminary screening of large exhibits [42].
- Polygon Query: Utilizes irregular polygonal shapes to target specific morphological features or areas of suspected high residue concentration based on visual inspection or prior intelligence [42].
The application of these spatial query methods ensures that transformations are preserved throughout the analytical process, maintaining the spatial context of recovered residues relative to the original exhibit configuration [42].
Experimental Protocols and Workflows
Integrated Analytical Workflow
A comprehensive workflow for spatial subsampling of oversized exhibits incorporates sequential processing stages from evidence collection through analytical characterization. The following diagram illustrates this integrated approach:
Detailed Spatial Subsampling Protocol
Evidence Documentation and Coordinate System Establishment
Prior to subsampling, comprehensive documentation of the oversized exhibit is essential:
- Photograph the exhibit from multiple angles under consistent lighting conditions
- Create a detailed diagram noting physical features, damage patterns, and potential residue deposition zones
- Establish a coordinate system relative to a fixed reference point on the exhibit
- Divide the exhibit surface into logical sectors based on morphology and suspected blast effects
Spatial Query Implementation
For bounding box queries:
- Define minimum and maximum coordinates (xmin, ymin, xmax, ymax) based on the established coordinate system
- Ensure the bounding box dimensions are appropriate for the analytical method (e.g., 200×200 pixel regions for GC-MS analysis)
- Record the coordinate system reference for data integration [42]
For polygonal queries:
- Define irregular shapes using multiple coordinate points to target specific morphological features
- Utilize tools such as Shapely Polygon for precise region definition
- Ensure polygon vertices are recorded in the established coordinate system [42]
Residue Recovery Techniques
Sequential swabbing with appropriate solvents is critical for effective residue recovery:
- Perform initial dry swabbing to recover particulate residues
- Follow with solvent-moistened swabbing using diethyl ether for organic residues
- Employ spatial subsampling techniques to enhance detection sensitivity [16]
Syringe filtration of extracts significantly improves recovery yield by minimizing background interference:
- Use 0.45μm syringe filters for extract clarification
- Process filters separately as they may contain additional particulate residues [16]
Analytical Characterization Methods
Table 1: Analytical Techniques for Residue Characterization
| Analytical Technique | Target Compounds | Sample Preparation | Detection Capabilities |
|---|---|---|---|
| Thin Layer Chromatography (TLC) | Organic explosives, petroleum hydrocarbons | Ether extraction, concentration | Preliminary screening, compound separation |
| Gas Chromatography-Mass Spectrometry (GC-MS) | High-boiling petroleum hydrocarbons, organic explosives | Syringe filtration, concentration | Compound identification, diesel fraction confirmation |
| Chemical Spot Tests | Inorganic ions (ammonium, nitrate) | Direct application to swab extracts | Presumptive identification of oxidizers |
| Fourier Transform Infrared (FTIR) Spectroscopy | Inorganic compounds, functional groups | Potassium bromide pellet or ATR | Molecular structure identification |
Research Reagent Solutions and Essential Materials
Table 2: Essential Materials for Spatial Subsampling and Analysis
| Category | Specific Items | Function/Application |
|---|---|---|
| Sampling Materials | Cotton swabs, nylon flocked swabs | Residue collection from exhibit surfaces |
| Sterile scalpels, tweezers | Precise sampling of fragmented materials | |
| Polypropylene containers | Evidence storage and transport | |
| Solvents | Diethyl ether, HPLC grade | Organic residue extraction |
| Methanol, acetonitrile | Alternative extraction solvents | |
| Deionized water | Inorganic residue extraction | |
| Filtration | Syringe filters (0.45μm, 0.22μm) | Extract clarification |
| Filter holders, syringes | Filtration apparatus | |
| Analytical Supplies | TLC plates (silica gel) | Preliminary organic analysis |
| GC-MS vials and inserts | Sample introduction for chromatographic analysis | |
| FTIR sample cards | Infrared spectroscopy sample preparation | |
| Reference Materials | Certified reference standards (ANFO components) | Method calibration and quantification |
| Diesel fuel samples | Petroleum hydrocarbon comparison |
Data Integration and Interpretation
Spatial Relationship Mapping
The preservation of spatial relationships throughout the analytical process enables the creation of residue distribution maps that correlate with blast dynamics. The following diagram illustrates the spatial data integration process:
Case Study: ANFO-Nitrate Detonation Analysis
In a practical application investigating ANFO-nitrate detonations, spatial subsampling of oversized exhibits revealed distinct distribution patterns:
- GC-MS analysis of ether extracts confirmed the presence of high-boiling petroleum hydrocarbons consistent with diesel fractions in ANFO
- Inorganic analyses identified ammonium and potassium nitrate, while chlorate, perchlorate, and metallic additives were absent
- Spatial subsampling enhanced detection sensitivity compared to whole-exhibit processing
- Syringe filtration produced the highest recovery yield by minimizing background interference [16]
Spatial subsampling represents a significant advancement in forensic investigation techniques for oversized and fragmented exhibits from explosive detonations. The integrated workflow combining targeted swabbing, sequential solvent extraction, syringe filtration, and complementary analytical methods has proven highly effective for complex evidence analysis. This approach not only improves residue recovery and enhances analytical reliability but also strengthens evidentiary interpretation by preserving spatial context. As post-blast investigation methodologies continue to evolve, spatial subsampling provides a robust framework for addressing the challenges presented by heterogeneous evidence, ultimately contributing to more accurate and scientifically defensible forensic conclusions.
Evaluating Analytical Efficacy: Sensitivity, Specificity, and the Role of Data Science
This technical guide benchmarks the performance of key analytical techniques—spot tests, thin-layer chromatography (TLC), gas chromatography-mass spectrometry (GC-MS), and ion trap mobility spectrometry (ITMS)—used in the analysis of post-blast residues. Focused on method detection limits (MDLs), the study provides a quantitative comparison to guide forensic chemists and researchers in method selection for identifying trace explosive materials in complex soil matrices. The data and protocols support a broader thesis on optimizing sampling and concentration techniques for reliable forensic residue analysis.
Post-blast residue analysis is one of the most challenging tasks in forensic chemistry due to the minimal quantities of unreacted materials present and complex sample matrices [1]. The forensic identification of explosive residues, such as organic compounds and ions from nitrate-based explosives, is crucial for investigating incidents [17]. A systematic approach that integrates classical and advanced analytical techniques ensures precise and reliable results across various scenarios, mitigating challenges posed by complex sample matrices [1]. This guide benchmarks the Method Detection Limits (MDLs) of spot tests, TLC, GC-MS, and ITMS, providing a structured comparison of their sensitivity and applicability within post-blast investigation protocols.
Experimental Protocols and Methodologies
Sample Collection and Preparation
Accurate post-blast sample collection is a fundamental step. Proper collection avoids contamination and false positives, which are critical when dealing with complex matrices [1].
- Artificial Explosions for MDL Determination: To determine the efficiency of the extraction method and the MDL, trace amounts of investigated explosives (ranging from milligrams to nanograms) were individually mixed with 100 g of sand. A sand sample without explosive traces served as a blank. Multiple sand samples (100 g each) were spiked with varying volumes of stock solutions to create a concentration series [1].
- Soil Extraction for Organic Explosives: A modified traditional extraction technique provided successful recovery with a relatively short duration. The process involved solid-phase extraction (SPE) for extraction, filtration, and clean-up to remove interferences. The concentrated organic extracts were then analyzed by the various techniques [1].
- Soil Preparation for Inorganic Ion Analysis (Nitrate): To simulate post-blast conditions, experimental soil pits were constructed on a sloped surface. The "exploded" pit was spiked with 1 gram of potassium nitrate (KNO₃). After exposure to natural environmental conditions, soil samples were collected from surface and subsurface layers. Nitrate ions were extracted by adding demineralized water to soil samples, followed by filtration, evaporation to dryness, and reconstitution in water for analysis [17].
Analysis Techniques and MDL Determination
- Spot Tests and TLC: Optimized concentrations of reagents were used for spot tests (e.g., 30% NaOH) and TLC visualization (e.g., 10% NaOH followed by Griess reagent) to enhance detection [1]. The minimum detection limits (MDL) for each explosive were determined to establish the sensitivity of the applied methods [1].
- Instrumental Techniques (GC-MS and ITMS): GC-EI-MS and ITMS analyses were performed according to established protocols. The minimum detection limits (MDL) for each explosive were a key objective of the analysis, providing insight into the sensitivity of the applied methods [1].
Benchmarking Detection Limits and Performance
The following table summarizes the quantitative performance and key characteristics of each technique based on experimental data.
Table 1: Comparison of Analytical Technique Performance for Explosive Residue Analysis
| Analytical Technique | Typical MDL Range | Key Advantages | Inherent Limitations |
|---|---|---|---|
| Spot Tests | ~1 mg (visual estimation) [1] | Rapid, simple, low-cost, useful for initial screening [1] | Qualitative or semi-quantitative, low sensitivity, prone to subjective interpretation [1] |
| Thin-Layer Chromatography (TLC) | Low μg to ng range (e.g., 0.04–0.1 μg for amino acids) [43] | Cost-effective, provides separation, versatile for various metabolites [43] | Requires derivatization for many compounds, less precise quantification without densitometry [43] |
| Ion Trap Mobility Spectrometry (ITMS) | Not explicitly quantified in results, but offers excellent mass detection limits and rapid analysis [1] | Excellent mass detection limits, rapid analysis time, superior resolution, small sample requirements [1] | Performance can be influenced by complex sample matrices [1] |
| Gas Chromatography-Mass Spectrometry (GC-MS) | Highest sensitivity; sub-nanogram levels for PETN confirmation [1] | High sensitivity and selectivity, provides confirmatory analysis [1] | PETN can undergo thermal degradation; may require method optimization [1] |
Essential Research Reagent Solutions
The following reagents are critical for the extraction, separation, and detection of explosive residues.
Table 2: Key Reagents and Materials for Post-Blast Residue Analysis
| Reagent/Material | Function/Application |
|---|---|
| Silica Gel 60 TLC Plates | Stationary phase for the separation of organic explosive compounds via TLC [43] |
| NaOH Solution (10%, 30%) | Used as a spot test reagent (30%) and for TLC visualization (10%) followed by Griess reagent [1] |
| Griess Reagent | Visualization agent for detecting nitroaromatics and nitrate esters after separation on TLC plates [1] |
| Ninhydrin (0.25% in Acetone) | Derivatization agent for visualizing amino acids and other amines on TLC plates [43] |
| Aniline-Diphenylamine-Phosphoric Acid Reagent | Derivatization agent for detecting monosaccharides on TLC plates with low MDL [43] |
| Solid-Phase Extraction (SPE) Disks (C18) | Used for sample clean-up and concentration of trace organic explosives from soil extracts [1] |
| Potassium Nitrate (KNO₃) | Standard compound used for simulating and calibrating the analysis of nitrate-based explosives [17] |
Analytical Workflow and Signaling Pathways
The logical workflow for analyzing post-blast residues, from sample collection to identification, integrates the techniques discussed. The following diagram visualizes this process, highlighting the complementary role of each method.
Analytical Workflow for Post-Blast Residues
This benchmarking study demonstrates that an integrated approach, combining classical techniques like spot tests and TLC with advanced instrumentation like GC-MS and ITMS, is essential for comprehensive post-blast residue analysis. While spot tests and TLC offer rapid, cost-effective screening, GC-MS provides the highest sensitivity and confirmatory power for definitive identification. The quantitative MDL data and detailed protocols provided serve as a critical reference for forensic scientists, enabling informed method selection to optimize recovery, detection, and analytical accuracy in forensic investigations. This work underscores the importance of continued protocol refinement to address challenges such as complex matrices and environmental factors.
The forensic identification of primary explosives and post-blast residues represents a significant analytical challenge, requiring techniques capable of detecting trace-level analytes within complex, contaminated matrices. In modern forensic laboratories, the convergence of multiple analytical techniques has become paramount for achieving unambiguous identification. This whitepaper examines the powerful synergy between tandem mass spectrometry (MS-MS) and scanning electron microscopy with energy-dispersive X-ray spectroscopy (SEM-EDS) for the definitive characterization of primary explosives and their post-blast residues. The evolving global security landscape, characterized by increased access to military-grade explosives and homemade explosive (HME) precursors, has further intensified the need for robust analytical methodologies [44]. Framed within broader research on post-blast residue sampling and concentration techniques, this technical guide details how these hybrid instruments provide complementary data streams—morphological and elemental information from SEM-EDS with molecular and structural data from MS-MS—to deliver a comprehensive identification platform essential for forensic investigations, security operations, and counter-terrorism initiatives.
The Analytical Challenge of Post-Blast Residue Identification
The forensic analysis of explosives after a blast event presents unique difficulties that stem from the complex nature of the samples and the demanding requirements for judicial evidence.
- Complex Matrices and Trace Levels: Post-blast debris is a chaotic mixture containing partially consumed explosive materials, reaction by-products, and environmental contamination. Analytes of interest are often present at trace levels, requiring highly sensitive techniques for detection and identification [44].
- Diverse Explosive Formulations: The advent of homemade explosives (HMEs) has dramatically increased the diversity of chemical substances that analysts may encounter. As noted in an Interpol review, restrictions on commercial and military high explosives have been circumvented by illicit production of HMEs, making universal detection protocols challenging to establish [44].
- Sample Degradation and Stability: The high-energy release during an explosion can degrade explosive compounds, creating transformation products that must be correctly identified and linked back to the original material.
- Judicial Standards of Evidence: Forensic evidence must withstand legal scrutiny, requiring analytical techniques that provide unambiguous results with a documented chain of custody and validated methodologies. The failure to properly extract analytes from difficult matrices is cited as a major reason for failure to identify post-blast residues [44].
Table 1: Key Challenges in Post-Blast Explosive Identification
| Challenge Category | Specific Issues | Impact on Analysis |
|---|---|---|
| Sample Complexity | Matrix interference, low analyte concentration, contamination | Reduced sensitivity, false positives/negatives |
| Chemical Diversity | HME formulations, evolving precursor chemicals | Need for versatile analytical approaches |
| Analytical Limitations | Inadequate extraction methods, insufficient sample collection | Failure to identify explosives in casework |
| Judicial Requirements | Accreditation standards (e.g., ISO 17025), validation demands | Slower adoption of new techniques despite potential advantages |
Scanning Electron Microscopy with Energy-Dispersive X-Ray Spectroscopy (SEM-EDS)
Fundamental Principles and Instrumentation
Scanning Electron Microscopy (SEM) is an advanced technique that enables the acquisition of detailed images of a sample's surface using a focused electron beam. When the electron beam strikes the sample, multiple interactions occur that generate various signals, including secondary electrons (SEs), backscattered electrons (BSEs), and characteristic X-rays [45]. Energy-Dispersive X-ray Spectroscopy (EDS) is integrated with SEM to analyze the elemental composition of the sample by detecting and quantifying these characteristic X-rays [45].
The operational workflow of SEM begins with the emission of electrons from a source, typically a tungsten filament or a field-emission gun (FEG). These electrons are accelerated and focused into a fine beam through electromagnetic lenses, which is then scanned across the sample surface in a raster pattern. The resulting signals are collected by specialized detectors and converted into images and spectral data [45].
SEM-EDS Methodology for Explosive Residue Analysis
The application of SEM-EDS to explosive residue analysis follows a structured methodological approach to ensure reliable and reproducible results.
Sample Collection and Preparation: The collection of gunshot residues (GSRs) shares similarities with post-blast explosive residue collection. Samples are typically collected using adhesive stubs or swabs from hands, clothing, or surfaces near the blast area [45]. For SEM analysis, non-conductive samples must be coated with a thin layer of conductive material (such as gold or carbon) to prevent the accumulation of surface charges that could distort the image [45]. Proper sample preparation is critical, as particulates must be properly adhered to the substrate without contamination.
Imaging and Analysis: Modern SEM-EDS systems often incorporate automated particle recognition software to streamline the detection and classification of characteristic explosive residue particles. This automation significantly enhances analysis throughput while maintaining consistency in particle identification [45].
Table 2: SEM Detectors and Their Applications in Explosive Residue Analysis
| Detector Type | Signals Detected | Information Provided | Application in Explosive Analysis |
|---|---|---|---|
| Secondary Electron (SE) Detector | Low-energy electrons from surface layers | High-resolution topographic images | Morphology of residue particles, surface features |
| Backscattered Electron (BSE) Detector | High-energy electrons reflected from sample | Compositional contrast based on atomic number | Discrimination of particles containing heavy elements (e.g., Pb, Ba, Sb) |
| Energy-Dispersive X-ray Spectroscopy (EDS) | Characteristic X-rays | Elemental composition and quantification | Confirmatory identification of elements associated with explosive formulations |
Figure 1: SEM-EDS Analytical Workflow for Explosive Residue Analysis
Advantages and Limitations of SEM-EDS
Advantages:
- Provides simultaneous morphological and elemental information from individual particles
- Non-destructive analysis, allowing samples to be preserved for additional testing
- High sensitivity for heavy elements commonly found in primer compositions (Pb, Ba, Sb)
- Automated particle recognition enables efficient screening of samples
- Results are generally admissible in judicial proceedings [45]
Limitations:
- Limited molecular information; cannot distinguish between different chemical compounds containing the same elements
- Lower sensitivity for light elements (e.g., H, C, N, O) that constitute organic explosives
- Requires conductive coating for non-conductive samples, potentially altering sample characteristics
- Time-consuming for manual operation, though automation has improved throughput
Tandem Mass Spectrometry (MS-MS) for Explosive Identification
Technical Foundations of Tandem MS-MS
Tandem mass spectrometry (MS-MS) employs multiple stages of mass analysis with an intermediate fragmentation step to provide detailed structural information about analyte molecules. The first mass analyzer (MS1) isolates precursor ions of a specific mass-to-charge ratio (m/z), which are then fragmented in a collision cell through processes such as collision-induced dissociation (CID). The resulting product ions are then analyzed by the second mass analyzer (MS2) to generate a fragmentation pattern that serves as a molecular fingerprint for unambiguous identification [44].
The exceptional sensitivity and selectivity of MS-MS systems make them particularly suited for identifying trace levels of organic explosives and their degradation products in complex post-blast matrices. This capability is crucial for addressing the growing challenge of homemade explosives (HMEs), where traditional military and commercial explosive signatures may be absent [44].
MS-MS Methodologies for Explosive Residues
Sample Preparation for MS-MS Analysis: Effective sample preparation is a critical prerequisite for successful MS-MS analysis. Solid-phase extraction (SPE) is widely employed for its ability to both clean up samples and concentrate trace analytes. As noted in chromatography literature, "SPE helps you concentrate trace amounts of analytes to improve the detection limits in subsequent analyses and is more effective at removing matrix interferences in LC and LC-MS analyses" [46]. Research has shown that dual sorbent SPE approaches can result in an approximate 10-fold improvement in limits of detection compared to single sorbent methods [44].
Other sample preparation techniques include:
- Liquid-Liquid Extraction (LLE): Partitioning analytes between immiscible solvents
- QuEChERS: Originally developed for pesticide analysis, this method provides a quick, easy, cheap, effective, rugged, and safe approach for sample cleanup [46]
- Derivatization: Chemical modification of analytes to improve volatility, stability, or detectability
Chromatographic Separation Coupled to MS-MS: Liquid chromatography (LC) is typically coupled with tandem MS for explosive analysis due to the relatively low volatility of many explosive compounds. The chromatographic step separates components in the sample before they enter the mass spectrometer, reducing matrix effects and simplifying spectral interpretation.
Table 3: MS-MS Analysis Modes for Explosive Identification
| MS-MS Mode | Operational Principle | Application in Explosive Analysis |
|---|---|---|
| Product Ion Scanning | MS1 fixed, MS2 scans fragment ions | Structural elucidation of target explosives |
| Precursor Ion Scanning | MS1 scans, MS2 fixed on product ion | Screening for compounds producing characteristic fragments |
| Neutral Loss Scanning | Both MS1 and MS2 scan with constant mass offset | Identifying compounds that lose specific neutral molecules |
| Multiple Reaction Monitoring (MRM) | Both MS1 and MS2 fixed on specific transitions | Highly sensitive and selective quantification of target analytes |
Advantages and Limitations of Tandem MS-MS
Advantages:
- Exceptional sensitivity and selectivity for organic explosive compounds
- Ability to identify unknown compounds through fragmentation patterns
- Capable of quantifying analytes across a wide dynamic range
- Compatible with various chromatographic separation techniques
- Can detect both parent compounds and degradation products
Limitations:
- Requires sample dissolution or extraction, which may not preserve physical evidence
- Limited direct morphological information about residue particles
- Instrumentation is complex and requires significant expertise to operate and maintain
- Can be susceptible to matrix effects that suppress or enhance ionization
The Hybrid Approach: Integrating SEM-EDS and Tandem MS-MS
Complementary Analytical Information
The true power of combining SEM-EDS and tandem MS-MS lies in the complementary nature of the information they provide. SEM-EDS delivers morphological and elemental data at the microscopic level, revealing the physical characteristics and inorganic composition of explosive residues. Meanwhile, tandem MS-MS provides molecular-level identification of organic compounds with high specificity and sensitivity. Together, these techniques create a comprehensive analytical picture that neither could achieve alone.
This hybrid approach is particularly valuable for complex post-blast samples where both inorganic and organic components may be present. For example, SEM-EDS can identify metallic particles characteristic of detonator components or casing materials, while tandem MS-MS can simultaneously identify the organic explosive matrix and its decomposition products.
Integrated Workflow for Comprehensive Analysis
A strategically designed integrated workflow maximizes the strengths of both techniques while compensating for their individual limitations.
Figure 2: Integrated SEM-EDS and Tandem MS-MS Workflow
Enhanced Forensic Capabilities
The hybrid SEM-EDS and MS-MS approach significantly enhances forensic capabilities for explosive identification in several key areas:
- Increased Confidence in Identification: Convergent findings from two fundamentally different analytical techniques provide stronger evidence for courtroom testimony.
- Broader Scope of Detectable Explosives: The combined system can identify both inorganic and organic explosive materials, including complex mixtures.
- Improved Sample Characterization: Physical characteristics (particle size, shape, morphology) from SEM-EDS combined with chemical identity from MS-MS provides a more complete forensic profile.
- Correlation of Ignition Mechanisms with Explosive Materials: SEM-EDS can identify primer components while MS-MS characterizes the main charge, helping reconstruct device construction.
Experimental Protocols and Methodologies
Sample Collection and Preservation Protocols
Proper sample collection and preservation are fundamental to successful analysis. The following protocols should be implemented:
For SEM-EDS Analysis:
- Use adhesive carbon tabs on aluminum stubs for particulate collection
- Collect control samples from adjacent areas to establish background levels
- Store samples in a desiccator to prevent moisture absorption and particle loss
- Document chain of custody for all samples
For Tandem MS-MS Analysis:
- Use solvent-moistened swabs for organic residue collection (acetonitrile or methanol are commonly used)
- Extract swabs in appropriate solvent (typically 2-5 mL) with sonication for 15-30 minutes
- Concentrate extracts under gentle nitrogen stream if needed
- Perform solid-phase extraction (SPE) cleanup using sorbents such as Oasis HLB or Isolute ENV+, which have demonstrated superior recovery for explosive compounds [44]
Instrumental Operating Conditions
SEM-EDS Typical Parameters:
- Accelerating voltage: 15-20 kV
- Working distance: 10-15 mm
- Beam current: 1-2 nA
- Detectors: SE, BSE, and EDS simultaneously active
- EDS acquisition time: 30-60 seconds live time
- Magnification: 500-10,000× depending on particle size
Tandem MS-MS Typical Parameters:
- Ionization source: Electrospray ionization (ESI) in negative or positive mode
- Nebulizer gas: 40-60 psi
- Drying gas flow: 10-12 L/min at 300-350°C
- Collision gas: Nitrogen or argon
- Collision energies: Compound-specific (typically 10-40 eV)
- MRM transitions: Optimized for target explosives and their degradation products
Quality Assurance and Validation
Robust quality assurance protocols are essential for forensic applications:
- Reference Materials: Analyze certified reference materials with each batch of samples
- Quality Control Samples: Include procedural blanks, matrix spikes, and duplicate analyses
- Instrument Calibration: Regular calibration of MS-MS using standard reference materials
- SEM-EDS Standards: Use elemental standards for EDS calibration and resolution checks
- Data Review: Implement peer review of all data and interpretations
Essential Research Reagent Solutions
Successful implementation of hybrid explosive identification methodologies requires specific research reagents and materials optimized for target analytes.
Table 4: Essential Research Reagents for Explosive Residue Analysis
| Reagent/Material | Technical Function | Application Notes |
|---|---|---|
| Oasis HLB SPE Sorbent | Mixed-mode reversed-phase and cation-exchange sorbent | Shows superior recovery for a wide range of explosive compounds; particularly effective in dual-sorbent approaches [44] |
| Isolute ENV+ SPE Sorbent | Hydroxylated polystyrene-divinylbenzene copolymer | Provides excellent recovery for polar explosive compounds; effective for environmental and complex matrices [44] |
| Acetonitrile (HPLC Grade) | Extraction solvent and LC-MS mobile phase | Effective for extracting a wide range of organic explosives; compatible with reversed-phase LC-MS analysis |
| Adhesive Carbon Tabs | Sample mounting for SEM analysis | Provides conductive surface for SEM analysis without introducing elemental interference |
| Conductive Coating Materials (Gold/Palladium, Carbon) | Surface coating for non-conductive samples | Prevents charging effects in SEM; carbon coating preferred when analyzing light elements |
| Certified Explosive Reference Standards | Quantification and method validation | Essential for creating calibration curves and verifying method performance |
| Derivatization Reagents (e.g., MSTFA, PFPA) | Chemical modification of target analytes | Improves volatility for GC analysis or enhances detection characteristics |
The integration of SEM-EDS and tandem MS-MS represents a powerful hybrid approach for the comprehensive identification of primary explosives and post-blast residues. This methodology delivers complementary data streams—morphological, elemental, and molecular information—that provide forensic scientists with an unparalleled capability to characterize explosive materials definitively. The continued evolution of these technologies, particularly through improved automation and data analysis algorithms, will further enhance their utility in forensic and security applications. As the landscape of explosive threats continues to evolve, with increasing availability of military-grade materials and innovative homemade formulations [44], the forensic community must prioritize the adoption and validation of such integrated instrumental approaches. Future developments will likely focus on streamlining the workflow between these techniques, enhancing data correlation algorithms, and expanding compound libraries to address emerging explosive threats. Through the continued refinement and implementation of these hybrid methodologies, the analytical community will be better equipped to address the complex challenges of explosive identification in both forensic and security contexts.
The forensic identification of explosives is a topic of considerable interest to all branches of homeland security due to the constant threat of increasing criminal activities. Trace residues of explosives have high evidentiary value as they represent the chemical composition of the material used in an explosion and could provide information on whether it was a homemade or commercial product [3]. In post-blast scenarios, analysts face the significant challenge of detecting and identifying minimal quantities of unreacted explosive materials amidst complex sample matrices [1]. Vibrational spectroscopy techniques, particularly Fourier Transform Infrared (FTIR) spectroscopy, have emerged as powerful tools for analyzing post-blast residues due to their high sensitivity and selectivity, requiring only tiny amounts of sample for analysis [3].
The analysis of post-blast residues by FTIR spectroscopy yields complex spectral data where visual inspection is inadequate for assessment and does not support recognition of the relationship between various chemicals and their IR fingerprints [3]. Chemometric techniques provide the solution to this challenge by enabling systematic and reproducible processing and analysis of spectral data with concurrent control of applied procedures and selected parameters [3]. This technical guide explores the integration of spectroscopic methods with multivariate statistical analysis and machine learning algorithms to create robust classification systems for identifying explosive residues, with particular focus on applications within post-blast investigation contexts.
Theoretical Foundations of Spectral Chemometrics
Spectral Data Characteristics and Pre-processing
Spectral data obtained from analytical techniques like FTIR spectroscopy possesses inherent characteristics that necessitate sophisticated computational approaches for optimal interpretation. Each FTIR spectrum represents a molecular fingerprint of the sample, with various chemical functional groups absorbing infrared light at specific wavelengths [3]. These spectra typically comprise hundreds to thousands of variables (wavenumbers), creating high-dimensional data spaces that require specialized handling.
Before applying classification algorithms, spectral data must undergo careful pre-processing to enhance signal quality and remove artifacts. Common pre-processing steps include Savitzky-Golay smoothing (using a window of 5 points with 2nd order polynomial fitting) and automatic-weighted least squares baseline correction [47]. Additional pre-processing may involve normalization procedures, though these must be carefully evaluated as they sometimes result in lower accuracies depending on the specific dataset [47]. The pre-processed spectra are typically mean-centered before multivariate analysis to improve algorithm performance [47].
Chemometric Workflow for Spectral Classification
The general workflow for chemometric analysis of spectral data follows a systematic progression from raw data to classification outcomes. Figure 1 illustrates this complete workflow, showing how raw spectral data undergoes pre-processing before feature reduction and classification:
Figure 1: Complete workflow for chemometric analysis of spectral data, from raw data to classification results.
Key Chemometric Algorithms for Spectral Classification
Feature Reduction Techniques
Principal Component Analysis (PCA)
Principal Component Analysis (PCA) stands as one of the best well-known methods for reducing variables in large volumes of spectral data [47]. This unsupervised algorithm reduces a large number of spectral variables into a smaller number of principal components (PCs), containing scores and loadings [47]. The scores reflect the variance found with regard to the samples, while the loadings show the most important variables related to the scores construction.
Mathematically, the scores and loading matrices are obtained after PCA decomposition of the pre-processed spectral matrix X as follows:
X = TPᵀ + E [47]
Where T represents the scores matrix, P represents the loading matrix, and E represents the residual matrix for total reconstruction of the pre-processed spectral matrix X [47]. As the scores represent the samples in the PC space, they can be used as input data for classification algorithms.
Successive Projections Algorithm (SPA)
The Successive Projections Algorithm (SPA) performs a discrete selection of variables, deterministically selecting the wavelengths that best differentiate groups through reduction of data multicolinearity [47]. SPA operates by minimizing variable redundancy and selecting subsets with minimal collinearity through a forward-selection approach.
SPA selects variables that best differentiate groups through the inverse of a cost function G, represented as:
G = (1/Nv) × Σgₙ [47]
Where Nv is the number of validation samples and gₙ is a function of the squared Mahalanobis distance between samples and class centers [47]. This approach ensures optimal variable selection for subsequent classification.
Classification Algorithms
Linear Discriminant Analysis (LDA)
Linear Discriminant Analysis (LDA) is a supervised classification technique that finds linear combinations of features that best separate two or more classes. The hybrid LDA-PCA technique has demonstrated exceptional performance in classifying post-blast residues, achieving the best results in studies analyzing explosive materials [3] [48]. LDA operates by maximizing between-class variance while minimizing within-class variance, creating decision boundaries in the reduced dimensional space.
Quadratic Discriminant Analysis (QDA)
Quadratic Discriminant Analysis (QDA) extends LDA by allowing non-linear decision boundaries through class-specific covariance matrices. While similar to LDA, QDA does not assume equal covariance structures across classes, making it more flexible for complex spectral datasets where different explosive materials may exhibit distinct variance patterns.
Support Vector Machines (SVM)
Support Vector Machines (SVM) represent a powerful machine learning approach for classifying highly complex data with low risk of overfitting [47]. SVM works by finding the optimal hyperplane that maximizes the margin between different classes in high-dimensional space. The PCA-SVM model has demonstrated remarkable performance in spectrochemical analysis, achieving 89% accuracy in distinguishing samples from patients with osteosarcopenia in clinical applications [47], showcasing its potential for explosive residue classification.
Algorithm Comparison and Performance
Table 1: Comparison of Chemometric Techniques for Spectral Classification
| Technique | Type | Key Characteristics | Best For | Performance Examples |
|---|---|---|---|---|
| PCA-LDA | Dimensionality reduction + Classification | Linear decision boundaries, maximizes class separation | Well-separated classes, linear problems | Best results for explosive residue classification [3] |
| PCA-QDA | Dimensionality reduction + Classification | Nonlinear decision boundaries, class-specific covariances | Classes with different covariance structures | Complex spectral datasets with distinct variance patterns |
| PCA-SVM | Dimensionality reduction + Machine Learning | Finds optimal hyperplane, handles high-dimensional data | Complex, non-linear datasets with clear margins | 89% accuracy in clinical spectral analysis [47] |
| SPA-LDA | Feature selection + Classification | Reduces multicolinearity, deterministic variable selection | Minimal feature sets, interpretable models | Discrete wavelength selection for specific compounds |
| SPA-QDA | Feature selection + Classification | Combines SPA variable selection with QDA flexibility | Non-linear problems with specific key wavelengths | Targeted analysis with specific spectral signatures |
| SPA-SVM | Feature selection + Machine Learning | Optimized feature set with powerful classification | High-dimensional data with redundant variables | Complex mixtures with identifiable key variables |
Experimental Protocols and Methodologies
Sample Preparation and Spectral Acquisition
Controlled experiments with post-blast remnants require specialized procedures to ensure representative sampling. In forensic explosive residue studies, remnants are obtained after controlled explosions performed by trained staff [3]. Various everyday objects, including glass, steel, plastic bags, plywood, chipboard, cardboard, hose pipes, towels, and fabric, should be placed near the explosion to simulate real-world conditions [3]. After detonation, the proximity of the container is initially searched as explosive residues decrease in concentration with increasing distance.
For FTIR analysis, sample preparation involves gently removing macroscopic particles from surfaces and mixing with potassium bromide (KBr) in a weight ratio of approximately 1:100 [3]. The homogeneous powder is pelletized using a hydraulic press with a clamping force of 80 kN, creating thin pellets 13 mm in diameter that are placed in the sample holder [3]. For samples surviving high-order blasts with visual damage, a cotton swab soaked in acetone can be used to clean surfaces, with the solvent transferred to an agate mortar. After acetone evaporation, the remaining products are mixed with KBr for analysis [3].
Spectral acquisition should be performed with precise instrumentation parameters. Studies utilize instruments like the FTIR IFS66v/S spectrometer with the sample chamber evacuated down to 3 mbar to eliminate water absorption lines [3]. Spectra should be collected within the 4000-400 cm⁻¹ range using a nominal spectral resolution of Δν = 4 cm⁻¹ (data spacing of 2.04 cm⁻¹), averaging 67 scans (approximately 1 minute of experiment time) to obtain good signal-to-noise ratio spectra [3]. A Mercury Cadmium Telluride (MCT) detector cooled to liquid nitrogen temperature (77 K) provides optimal detection sensitivity [3].
Data Processing and Analysis Framework
The R environment serves as an extremely versatile platform for scientific data evaluation of spectral data, with RStudio as the graphical user interface [3]. The power and flexibility of R lies in its modular structure, allowing mounting of required packages at any time [3]. Key libraries include:
- hyperSpec: For hyperspectral datasets manipulations [3]
- ggplot2: For high-quality visualizations [3]
- caret: For evaluation of classification and confusion matrices [3]
Spectral data collected using native instrument software should be exported to ASCII files through macro procedures, then imported into R software for analysis [3]. For MATLAB-based workflows, the PLS Toolbox with lab-made routines can be employed similarly [47].
The data analysis protocol should begin with dividing samples into training (70%) and test (30%) sets using algorithms like the Kennard-Stone (KS) algorithm [47]. The training samples are used for model construction and cross-validation, while testing samples serve for final model evaluation [47].
Applications in Post-Blast Residue Analysis
Classification of High and Low Order Explosion Residues
The efficiency and type of explosion are critical parameters in forensic explosive analysis. A low-order explosion occurs when the blast pressure front moves at a slow pace and displaces or distorts objects in its path, often leaving a quite high quantity of non-reacted particles from explosives on remnant surfaces as consumption is less than 100% [3]. In contrast, high-order explosions are characterized by rapid combustion of explosive materials, rapid expansion of resultant gases, and build-up of high heat and pressure, leaving only scarce amounts of post-blast residues [3].
Research demonstrates that FTIR spectroscopy combined with machine learning classification can successfully identify high explosive materials (C-4, TNT, and PETN) in residues after both high- and low-order explosions [3] [48]. In these challenging scenarios, the hybrid LDA-PCA technique has shown particularly promising results, enabling reliable identification even when minimal residue material is available [3].
Comparative Analytical Techniques
While FTIR spectroscopy with chemometrics provides valuable capabilities, forensic analysts often employ complementary techniques for comprehensive residue characterization. Table 2 compares key analytical methods used in post-blast investigation:
Table 2: Analytical Techniques for Explosive Residue Analysis
| Technique | Target Analytes | Specificity | Typical LOD | Applications in Post-Blast Analysis |
|---|---|---|---|---|
| FTIR Spectroscopy | Organic functional groups | High (molecular fingerprint) | μg range | Identification of explosive type, polymer binders [3] |
| GC-MS | Volatile and semi-volatile organics | High (mass spectrum) | pg-ng | Confirmatory analysis, trace residue detection [1] [6] |
| Raman Spectroscopy | Raman-active compounds | High (structural fingerprint) | μg/ng (SERS) | Field detection, intact explosives [2] [6] |
| IMS | Organic explosives | Medium-high | pg-ng | Rapid screening, airport security [1] [6] |
| LC-MS | Thermally labile compounds | High | pg-ng | PETN, NG, degradation products [1] [2] |
| SEM/EDS | Elements (Z > 10) | High (elements) | pg | Inorganic residues, gunshot residue [6] |
Experimental Workflow for Post-Blast Residue Classification
Figure 2 illustrates the complete experimental workflow for post-blast residue analysis using chemometric techniques:
Figure 2: Experimental workflow for post-blast residue classification using FTIR and chemometrics.
The Scientist's Toolkit: Essential Research Materials
Table 3: Essential Research Reagents and Materials for Spectral Analysis of Explosive Residues
| Item | Specification | Function/Application |
|---|---|---|
| Potassium Bromide (KBr) | Infrared grade, purity >99% | Matrix for FTIR pellet preparation; transparent to IR radiation [3] |
| Acetone | HPLC or reagent grade | Solvent for residue extraction from surfaces and debris [3] |
| Perchloric Acid | 7 M solution | Protein precipitation in biological samples; sample clean-up [47] |
| Explosive Standards | Certified reference materials (TNT, RDX, PETN, C-4) | Method calibration, quality control, reference spectra [3] [6] |
| Hydraulic Press | 80 kN clamping force | KBr pellet preparation for transmission FTIR [3] |
| ATR Crystal | Diamond crystal element | Sample platform for ATR-FTIR measurements [47] |
| MCT Detector | Liquid nitrogen cooled (77 K) | Enhanced sensitivity detection for FTIR spectroscopy [3] |
Advanced Applications and Future Directions
The integration of chemometrics with spectral analysis continues to evolve with advancements in machine learning and computational power. Deep learning approaches are emerging as powerful alternatives to traditional chemometric methods, potentially offering enhanced classification accuracy for complex spectral datasets. Furthermore, the development of portable spectroscopic instruments coupled with real-time chemometric analysis promises to transform field-based forensic investigations, allowing rapid on-site classification of explosive residues.
Recent research has also explored multi-technique data fusion, combining FTIR with Raman spectroscopy and mass spectrometric data through advanced chemometric models to achieve unprecedented classification accuracy. These approaches leverage the complementary strengths of different analytical techniques while mitigating their individual limitations, creating more robust identification systems for complex post-blast scenarios where residue composition may be highly variable and degraded.
As terrorist organizations continually adapt their explosive formulations, the forensic community must similarly advance its analytical capabilities. The combination of sophisticated spectroscopic techniques with powerful chemometric algorithms represents a critical frontier in this ongoing effort, providing forensic scientists with increasingly powerful tools for detecting and identifying explosive materials in post-blast investigations.
The forensic investigation of explosive events relies heavily on the chemical analysis of post-blast residues to determine the type of explosive used and the nature of the detonation. This process is analytically challenging due to the chaotic destruction at blast scenes and the trace amounts of unreacted material remaining, particularly from high-order explosions which consume nearly all the original material [3]. Low-order explosions, resulting from incomplete detonation due to factors like aged explosives or detonator failure, typically leave more substantial non-reacted particles on remnant surfaces [3].
Fourier Transform Infrared (FTIR) spectroscopy has emerged as a powerful technique for this analysis, providing a molecular fingerprint of samples based on the specific infrared light absorption by their chemical functional groups [3]. However, visual inspection of the complex FTIR spectra is insufficient for reliable identification. This case study details the integration of statistical multivariate analysis and machine learning classification techniques with FTIR spectroscopy to systematically and reproducibly identify high explosives in post-blast residues, framing this methodology within the critical context of post-blast residue sampling and concentration research [3].
Experimental Protocol and Methodologies
Controlled Explosions and Sample Collection
To generate representative post-blast residues, researchers conducted controlled explosions of three high-explosive materials: C-4, PETN, and TNT [3]. Both low-order and high-order explosions were performed inside a small steel container (18 by 18 by 18 cm³) [3].
- Explosive Materials: C-4 (containing RDX as its primary component), PETN, and TNT.
- Object Substrates: Various everyday objects were placed inside the container to act as residue "catchers," including glass, steel, plastic bags, plywood, chipboard, cardboard, hose pipes, towels, and fabric [3].
- Post-Blast Collection: After detonation, potentially useful objects were labeled and stored separately. Surfaces of these objects were then sampled. For visible particles, these were gently removed. For surfaces with no visible residue, a cotton swab soaked in acetone was used to rinse the surface, transferring potential trace residues into solution [3].
FTIR Spectroscopic Analysis
Collected samples underwent rigorous preparation and analysis to generate high-quality spectral data [3].
- Sample Preparation: Macroscopic particles were mixed with potassium bromide (KBr) at a weight ratio of approximately 1:100 and pressed into pellets using a hydraulic press. For acetone rinsates, the solvent was evaporated, and the remaining residue was mixed with KBr and pelletized [3].
- Instrumentation: Spectra were collected using an IFS66v/S FTIR spectrometer equipped with a Mercury Cadmium Telluride (MCT) detector cooled to 77 K [3].
- Data Acquisition Parameters:
- Spectral Range: 4000–400 cm⁻¹
- Resolution: 4 cm⁻¹
- Scans: 67 per spectrum (averaged to improve signal-to-noise ratio)
- Atmosphere: Sample chamber evacuated to 3 mbar to minimize atmospheric water interference [3].
- Data Handling: Spectra were collected using Bruker Opus software and exported as ASCII files for subsequent processing in the R environment [3].
Table 1: Key FTIR Spectroscopy Parameters for Post-Blast Residue Analysis
| Parameter | Specification | Purpose/Rationale |
|---|---|---|
| Detection Technique | Fourier Transform Infrared (FTIR) Spectroscopy | Provides molecular fingerprint based on functional group absorption [3]. |
| Spectral Range | 4000–400 cm⁻¹ | Covers fingerprint region for unique identification of organic compounds [3]. |
| Spectral Resolution | 4 cm⁻¹ | Provides sufficient detail for distinguishing subtle spectral features [3]. |
| Detector Type | Mercury Cadmium Telluride (MCT) | High sensitivity required for detecting trace amounts of residues [3]. |
| Sample Presentation | KBr Pellet | Traditional method for transmission analysis of solid powders [3]. |
| Atmosphere Control | Evacuated to 3 mbar | Removes water vapor and CO₂ to prevent interference in the infrared spectrum [3]. |
Data Pre-treatment and Machine Learning Workflow
The analysis pipeline moved from raw spectral data to a final classification using a code-driven approach in the open-source R environment, promoting reproducibility and transparency [3].
Figure 1: Machine Learning and Spectral Analysis Workflow
The process involved several critical steps:
- Data Pre-treatment: Raw spectral data often contains artifacts and non-chemical variances. Pre-processing is essential and may include:
- Baseline Correction: Removes sloping or curved baselines caused by light scattering.
- Normalization: Scales spectra to a standard intensity, correcting for variations in sample amount or thickness.
- Smoothing: Reduces high-frequency noise to improve the signal-to-noise ratio [3].
- Dimensionality Reduction: Each FTIR spectrum contains thousands of data points (wavenumbers). Principal Component Analysis (PCA) is a statistical technique used to reduce this high dimensionality by transforming the original variables into a smaller set of principal components that capture the most significant variance in the data. This simplifies the data structure without critical information loss [3].
- Model Training and Classification: The reduced dataset was used to train and test various machine learning classifiers. The study specifically highlighted the effectiveness of a hybrid LDA-PCA technique [3] [48]. Linear Discriminant Analysis (LDA) seeks to find a linear combination of features that best separates two or more classes of objects, in this case, the different explosive types.
Results and Critical Analysis
Performance of Machine Learning Classifiers
The integration of machine learning with FTIR spectroscopy proved highly successful in classifying the post-blast residues. The hybrid LDA-PCA technique yielded the best results, effectively distinguishing between C-4, PETN, and TNT residues even in the challenging context of high-order explosions where residue is scarce [3] [48]. This demonstrates that multivariate analysis can extract meaningful chemical information from complex, trace-level spectral data that would be impossible to interpret by visual inspection alone.
The Scientist's Toolkit: Essential Research Reagents and Materials
The analysis of post-blast residues requires specific materials for sample collection, preparation, and analysis. The following table details key reagents and their functions based on the cited protocols.
Table 2: Essential Research Reagents and Materials for Post-Blast Residue Analysis
| Item | Function/Application | Technical Notes |
|---|---|---|
| Potassium Bromide (KBr) | Matrix for FTIR pellet preparation; transparent to IR light. | High-purity grade required; mixed with sample at ~1:100 ratio (sample:KBr) [3]. |
| Acetone (Solvent) | Extraction of trace explosive residues from solid surfaces and debris. | Swabs soaked in solvent used to rinse surfaces; solvent is evaporated pre-analysis [3]. |
| High Explosive Standards | Reference materials for method validation and machine learning training. | Pure C-4, PETN, and TNT are essential for building a reference spectral database [3]. |
| Passive Samplers (e.g., PDMS, ACST) | Enrichment of explosive vapors or residues from air or aqueous phases. | Used for pre-blast detection and analysis of volatile explosives like TATP [49]. |
| Cotton Swabs / Wipes | Standard medium for non-destructive sampling of solid surfaces for residue. | Allows for targeted collection from complex debris and objects acting as "catchers" [3]. |
| Calibration Mixtures | For ensuring instrumental sensitivity, selectivity, and specificity. | Critical for maintaining data quality; sensitivity is key for trace (ppm-ppb) analysis [13]. |
Relationship to Sampling and Concentration Techniques
The effectiveness of the FTIR and machine learning methodology is intrinsically linked to upstream sampling and concentration techniques. As noted in research, failure to properly extract analytes from difficult matrices is a major reason for failing to identify post-blast homemade explosives [44]. The selection of a sampling method must consider the nature of the explosive and the substrate.
- Solid Phase Extraction (SPE): Research has investigated various sorbents for recovering a wide range of explosives from complex matrices like soil, cooking oil, and wastewater. A dual-sorbent SPE approach using Oasis HLB and Isolute ENV+ was found to provide low matrix effects and good recovery, improving the limit of detection approximately tenfold compared to single-sorbent methods [44].
- Passive Sampling: For volatile explosives like triacetone triperoxide (TATP), enrichment from the air or aqueous phase using passive samplers such as polydimethylsiloxane (PDMS) rods or activated carbon sampling tubes (ACST) has proven effective before analysis by techniques like Gas Chromatography-Mass Spectrometry (GC-MS) [49].
- Solvent Extraction: The use of acetone swabbing, as employed in the featured case study, remains a common and effective technique for transferring residues from solid surfaces into an analyzable form [3].
These concentration steps are vital for presenting a sufficiently rich analyte mixture to the analytical instrument, thereby enabling the sensitive detection required for successful machine learning classification.
This case study demonstrates a robust, reproducible framework for the forensic identification of explosives in post-blast residues. The core achievement lies in the successful coupling of FTIR spectroscopy with a machine learning-based classification pipeline, implemented in the open-source R environment. The hybrid LDA-PCA technique was particularly effective in classifying residues from C-4, PETN, and TNT, even after high-order detonations.
This analytical success is fundamentally dependent on rigorous sample collection and concentration techniques within the broader research context of post-blast residue analysis. Methodologies such as solvent swabbing, Solid Phase Extraction, and passive sampling are critical for isolating and enriching trace analytes from chaotic and contaminated blast scenes. The integration of advanced data analysis with rigorous forensic sampling protocols provides a powerful tool for homeland security and forensic investigations, ultimately aiding in the attribution and prosecution of bombing-related crimes.
The forensic investigation of post-blast scenes presents a unique set of analytical challenges, including the presence of complex sample matrices, environmental contamination, and the trace-level, degraded nature of explosive residues. In recent years, a significant paradigm shift has occurred, moving analysis from centralized laboratories towards field-deployable technologies capable of providing rapid, on-site intelligence. This transition necessitates a critical assessment of two foundational pillars for these portable methods: reproducibility and field-deployability. Reproducibility ensures that results are reliable, verifiable, and forensically defensible, while field-deployability determines the practical utility of a technology in the challenging and variable conditions of a real-world incident scene. This guide provides a technical framework for researchers and scientists to evaluate emerging portable technologies for post-blast explosive residue analysis, focusing on these critical attributes within the context of a broader research thesis on sampling and concentration techniques.
Analytical Technologies for Explosive Residues: A Comparative Framework
A range of analytical techniques has been adapted or developed for the detection and identification of post-blast explosive residues. The selection of an appropriate method involves balancing analytical performance with practical constraints. The table below summarizes the key technologies relevant to field application.
Table 1: Comparison of Analytical Techniques for Explosive Residue Analysis
| Analytical Technique | Key Principles | Strengths | Limitations for Field Use | Reproducibility Considerations |
|---|---|---|---|---|
| Direct Analysis in Real Time Mass Spectrometry (DART-MS) | Ambient ionization; thermally desorbs and ionizes samples directly for MS analysis [12]. | Minimal sample preparation; high specificity; confirmatory analysis [12]. | High-resolution MS preferred for confidence, which can challenge portability; requires robust calibration [12]. | Sensitivity to parameter variation (e.g., DART gas temperature); requires robust calibration and standardized protocols [12]. |
| Gas Chromatography-Vacuum Ultraviolet Spectroscopy (GC-VUV) | GC separation followed by VUV absorption detection (100-200 nm) [13]. | High selectivity for isomers; provides specific spectral fingerprints [13]. | Parts-per-million sensitivity may be insufficient for trace residues without pre-concentration; requires gas supplies [13]. | Spectral libraries and flow cell temperature can strongly influence identification specificity [13]. |
| Infrared (IR) Spectroscopy | Analysis of molecular vibrations to generate a chemical fingerprint [9]. | Non-destructive; minimal sample preparation; portable versions available (NIR, ATR-FTIR) [9]. | Can struggle with complex mixtures and spectral overlaps; portable NIR may have lower resolution [9]. | Affected by sample homogeneity and environmental contaminants; requires chemometric models for complex data [9]. |
| Ion Mobility Spectrometry (IMS) | Separation of gas-phase ions based on size, shape, and charge in a drift tube. | Rapid detection; high sensitivity; widely deployed for trace explosive detection [12]. | Susceptible to matrix interference and false positives; used for presumptive, not confirmatory, analysis [12]. | Can be affected by environmental contaminants (humidity, temperature) leading to variable results [12]. |
Assessing Reproducibility: Protocols and Metrics
Reproducibility is the cornerstone of forensic science, ensuring that results are independent of the operator, location, or specific instrument. For a method to be considered reproducible, it must demonstrate reliability across these variables.
Experimental Protocol for Reproducibility Assessment
A rigorous assessment of reproducibility should include the following steps:
- Preparation of Standardized Test Materials: Create homogeneous samples of explosive residues on relevant substrates (e.g., metal, plastic, glass). Spiked samples should cover a range of concentrations, from high to the limit of detection (LOD). For a more real-world assessment, include post-blast debris from controlled detonations [12].
- Inter-laboratory Comparison: Distribute the standardized test materials to multiple independent laboratories equipped with the same technology platform. Each laboratory should follow an identical, detailed standard operating procedure (SOP).
- Data Collection and Analysis: Each lab analyzes the samples and reports key data outputs, including:
- Identification of the explosive compound (correct/incorrect).
- Calculated concentration or peak area.
- Signal-to-noise ratio.
- Statistical Evaluation: Calculate inter-laboratory precision metrics, such as the relative standard deviation (RSD%) for quantitative measurements. For qualitative identification, report the percentage of correct identifications across all participants.
Key Reproducibility Metrics
Table 2: Key Metrics for Assessing Method Reproducibility
| Metric | Description | Target Threshold |
|---|---|---|
| Limit of Detection (LOD) | The lowest concentration that can be reliably detected. | Consistent LOD across multiple instruments and operators [44]. |
| Limit of Identification (LOI) | The lowest concentration that can be reliably identified. | Consistent LOI across multiple instruments and operators. |
| Inter-laboratory Precision | The degree of agreement between measurements made by different labs on the same sample, often expressed as RSD%. | RSD < 20% for quantitative analyses is often targeted in analytical chemistry. |
| False Positive/Negative Rate | The rate of incorrect identification when a compound is absent/present. | Minimized and consistent across all testing sites [9]. |
Evaluating Field-Deployability: Criteria and Logistics
Field-deployability assesses whether a technology can maintain its performance outside the controlled laboratory environment. This assessment is multi-faceted.
Field-Deployability Assessment Criteria
Table 3: Field-Deployability Assessment Criteria for Portable Technologies
| Criterion | Assessment Questions | Experimental Evaluation |
|---|---|---|
| Portability & Ruggedness | What are the instrument's size, weight, and power requirements (SWaP)? Is it resistant to shock, vibration, and environmental conditions? | Operate the instrument after simulated transit stress (vibration test). Measure performance using battery vs. main power. |
| Environmental Tolerance | How do temperature extremes and humidity fluctuations affect analytical performance (sensitivity, specificity)? | Conduct calibration and sample analysis in controlled environmental chambers set to extreme field-relevant conditions. |
| Ease of Use | What level of training is required? Is the workflow simple and intuitive? Can it be operated with personal protective equipment (PPE)? | Time-to-result and success rate for a novice operator following the SOP. |
| Analysis Speed | What is the total time from sample introduction to result? This includes any sample preparation and data processing. | Benchmark against the operational requirements for a bomb scene investigation (e.g., results within minutes). |
| Maintenance & Logistics | How often does the instrument require calibration and maintenance? Are consumables (gases, solvents, swabs) readily available and stable? | Track maintenance logs and consumable usage over a multi-week field trial. |
Sampling and Concentration Protocol for Field Analysis
The efficacy of any analytical technology is contingent on proper sample collection. The following protocol, adaptable for field use, is critical for ensuring representative and concentrated residues.
- Swab Selection: Use swabs compatible with the analytical technique (e.g., cotton, polyester, or specialized media like PTFE). The compatibility of dry versus solvent-wet swabs should be evaluated [12].
- Sampling: Swab the surface of post-blast debris using a consistent, firm pressure and a template pattern to ensure representative sampling. For porous or irregular surfaces, a slightly wet swab may improve recovery [12].
- Extraction/Concentration:
- For Return-to-Lab: Place the swab in a vial with a suitable solvent (e.g., acetone, acetonitrile) [12]. Sonicate or vortex to extract residues. The extract can be concentrated under a gentle stream of nitrogen if needed.
- For Field Analysis (DART-MS): The swab can be presented directly to the DART source, or a thermal desorption unit can be used to introduce the analyte into the MS [12].
- Clean-up (if needed): For complex matrices, use a solid-phase extraction (SPE) sorbent. Studies have shown that dual sorbent SPE (e.g., Oasis HLB and Isolute ENV+) can improve recovery and reduce matrix effects, lowering the LOD by approximately 10-fold [44].
A Framework for Integrated Workflow and Validation
The following diagrams and tools integrate the concepts of reproducibility and field-deployability into a practical research and validation workflow.
Experimental Workflow for Method Assessment
The diagram below outlines a logical pathway for the comprehensive assessment of a portable analytical technology.
Key Research Reagent Solutions
The following table details essential materials and reagents required for experiments in this field.
Table 4: Essential Research Reagents and Materials for Post-Blast Residue Analysis
| Item | Function/Application | Technical Notes |
|---|---|---|
| Certified Reference Standards | Calibration and method validation for target explosives (e.g., TNT, RDX, TATP, AN). | Essential for establishing reproducibility. Must be traceable to a primary standard [44]. |
| Specialized Sampling Swabs | Collection of explosive residues from post-blast debris and surfaces. | Material (cotton, polyester, PTFE) must be compatible with the analytical technique (e.g., DART-MS) [12]. |
| Solid-Phase Extraction (SPE) Sorbents | Sample clean-up and pre-concentration of residues from complex matrices (e.g., soil, wastewater). | Oasis HLB and Isolute ENV+ sorbents have shown high recovery for a range of explosives [44]. |
| Deuterated Internal Standards | Improvement of quantitative accuracy by correcting for sample loss and matrix effects during analysis. | e.g., D5-NT or 15N-AN for GC-MS or LC-MS analysis. |
| Mobile Phase Solvents | For LC-MS and extraction procedures. | High-purity, LC-MS grade solvents (e.g., acetonitrile, methanol) are required to minimize background interference. |
Validation Pathway for New Technologies
The path from a research prototype to a validated field tool requires systematic validation, as illustrated below.
Future-proofing portable technologies for post-blast analysis requires a rigorous, dual-focused approach on reproducibility and field-deployability. As the threat landscape evolves with the proliferation of homemade explosives [9] [44], the forensic community must adopt a standardized framework for evaluating new tools. This involves not only benchmarking against traditional laboratory techniques but also subjecting instruments to realistic field trials and inter-laboratory studies. By adhering to the structured assessment protocols, metrics, and workflows outlined in this guide, researchers and laboratory managers can make informed decisions, accelerate the adoption of robust technologies, and ultimately enhance the capabilities of front-line personnel in responding to and investigating explosive incidents.
Conclusion
The forensic investigation of post-blast residues necessitates a robust, multi-technique approach that intelligently integrates classical methods with advanced instrumentation. Success hinges not only on sensitive detection but also on optimized sampling and concentration protocols tailored to specific environmental and matrix challenges. The emergence of chemometrics and machine learning represents a paradigm shift, enabling more powerful data interpretation and classification from complex datasets. Future directions should focus on standardizing these integrated workflows, developing more sensitive and rugged field-deployable tools, and further harnessing artificial intelligence to automate residue identification. These advancements will significantly enhance the reliability, speed, and evidentiary value of forensic findings in both security and clinical research contexts where explosive trauma is studied.
References
- 1. Systematic analysis of post-blast organic traces in soil ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967325001244]
- 2. A multifaceted approach to capturing post-blast TATP [https://www.sciencedirect.com/science/article/abs/pii/S2468170925000566]
- 3. Classification of the Residues after High and Low Order ... [https://www.mdpi.com/1420-3049/28/5/2233]
- 4. Quantification and aging of the post-blast residue of TNT ... [https://pubmed.ncbi.nlm.nih.gov/12877289/]
- 5. The spatial distribution patterns of condensed phase post ... [https://www.sciencedirect.com/science/article/pii/S0304389416304460]
- 6. Investigation and analysis of explosive traces in public ... [https://pubs.rsc.org/en/content/articlehtml/2025/ay/d5ay00183h]
- 7. Pesticide Residue Analysis in Food: Advances, Challenges ... [https://www.chromatographyonline.com/view/pesticide-residue-analysis-in-food-advances-challenges-and-case-studies]
- 8. Innovative Analytical Approaches for Food Pesticide Residue ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12452724/]
- 9. A Review of Analytical and Chemometric Strategies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12002426/]
- 10. Pesticide residue detection techniques for increasing ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1694779/full]
- 11. Characterization of Explosives in a Controlled Blast ... [https://www.sciencedirect.com/science/article/abs/pii/S0039914025014778]
- 12. Identification of post-blast explosive residues using direct- ... [https://www.sciencedirect.com/science/article/abs/pii/S2468170919300827]
- 13. Advances in Detecting and Identifying Explosives After an Attack [https://nij.ojp.gov/topics/articles/advances-detecting-and-identifying-explosives-after-attack]
- 14. Analytical Techniques for Forensic Investigation of ... [https://www.preprints.org/manuscript/202509.0885/v1]
- 15. Subsampling Blood Swabs as an Efficient and Good ... [https://www.mdpi.com/2673-6756/4/4/47]
- 16. Analytical Techniques for Forensic Investigation of ... [https://chemrxiv.org/engage/chemrxiv/article-details/68bafd4423be8e43d64c6f80]
- 17. Impact of Rainy Environments on Nitrate Ion Detection in ... [https://www.forensicscijournal.com/journals/jfsr/jfsr-aid1092.php]
- 18. A comparison of solvent extract cleanup procedures in the ... [https://pubmed.ncbi.nlm.nih.gov/23278326/]
- 19. Identification and analysis of organic explosives from post ... [https://www.sciencedirect.com/science/article/abs/pii/S0304389420319932]
- 20. Optimization of solid phase extraction for simultaneous ... [https://www.frontiersin.org/journals/environmental-science/articles/10.3389/fenvs.2025.1539932/full]
- 21. How To Verify The Filtration Effectiveness Of Syringe Filters? [https://www.alwsci.com/news/how-to-verify-the-filtration-effectiveness-of-79411148.html]
- 22. Optimizing Filtration Efficiency: A Comparative Study of ... [https://welchlab.com/blogs/news/syringe-filter-materials-and-pore-sizes?srsltid=AfmBOopoT1MsRXJ_B6ngTMXGPJCbpI56YwASraOxnwQydQOz8GU0f_Ip]
- 23. Syringe Filters vs Filter Vials: What is the Best Method for ... [https://chromtech.com/blog/syringe-filters-vs-filter-vials-what-is-the-best-method-for-filtration/]
- 24. Integrated Analytical Approaches for Forensic ... [https://www.preprints.org/manuscript/202509.0885]
- 25. Systematic analysis of post-blast organic traces in soil ... [https://pubmed.ncbi.nlm.nih.gov/39970689/]
- 26. Post-blast detection of traces of explosives by means of ... [https://www.sciencedirect.com/science/article/abs/pii/S092420310900040X]
- 27. Classification of the Residues after High and Low Order ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10004765/]
- 28. Advances in Detecting and Identifying Explosives After an ... [https://www.forensicmag.com/611500-Advances-in-Detecting-and-Identifying-Explosives-After-an-Attack/]
- 29. Forensic Instrumental Analysis of Post-Blast Inorganic ... [https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5339911]
- 30. Chromatography 2025: Sample Preparation Goes Automated [https://www.sepscience.com/chromatography-2025-sample-preparation-goes-automated-11259]
- 31. Forensic analysis of post-blast residues from lead ... [https://pubmed.ncbi.nlm.nih.gov/40946337/]
- 32. What is Soxhlet Extraction? [https://www.organomation.com/what-is-soxhlet-extraction]
- 33. Optimization of extraction process and solvent polarities to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11933538/]
- 34. Highly parallel optimisation of chemical reactions through ... [https://www.nature.com/articles/s41467-025-61803-0]
- 35. 2025 Chemical Industry Outlook [https://www.deloitte.com/us/en/insights/industry/chemicals-and-specialty-materials/chemical-industry-outlook/2025.html]
- 36. Three approaches to minimize matrix effects in residue ... [https://www.sciencedirect.com/science/article/abs/pii/S0308814618320776]
- 37. The selective cleanup of complex matrices and ... [https://pubmed.ncbi.nlm.nih.gov/22099224/]
- 38. Sample Preparation with ZipTip ® Micropipette Tips [https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/protein-biology/protein-mass-spectrometry/ziptip-micro-spe-pipette-tips?srsltid=AfmBOor4ZjQq1YoiWBR7hBmQpqQ0A1kND_amrtytF0GQQO1726hsNJMh]
- 39. Minimizing thermal degradation in gas chromatographic ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967315003738]
- 40. Development of a Gas Chromatography with High ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10448686/]
- 41. Optimized Explosives Analysis Using Portable Gas ... [https://www.chromatographyonline.com/view/optimized-explosives-analysis-using-portable-gas-chromatography-mass-spectrometry-battlefield-forens]
- 42. Spatial query/subsampling — spatialdata - scverse [https://spatialdata.scverse.org/en/latest/tutorials/notebooks/notebooks/examples/spatial_query.html]
- 43. Chemometrics-Based TLC and GC-MS for Small Molecule ... [https://www.intechopen.com/chapters/63447]
- 44. Interpol review of the analysis and detection of explosives ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9845958/]
- 45. Scanning Electron Microscopy Techniques in the Analysis ... [https://www.mdpi.com/2076-3417/15/5/2634]
- 46. Sample Preparation Techniques [https://www.thermofisher.com/au/en/home/industrial/chromatography/chromatography-learning-center/chromatography-consumables/sample-prep.html]
- 47. Spectrochemical analysis of blood combined with ... [https://www.nature.com/articles/s41598-023-36834-6]
- 48. Classification of the Residues after High and Low Order ... [https://pubmed.ncbi.nlm.nih.gov/36903479/]
- 49. Determination of triacetone triperoxide (TATP) traces using ... [https://www.sciencedirect.com/science/article/pii/S0379073823001238]